Advanced Copper-Catalyzed Synthesis of Alpha-Amino-Gamma-Butyrolactone Intermediates for Commercial Scale-Up
Advanced Copper-Catalyzed Synthesis of Alpha-Amino-Gamma-Butyrolactone Intermediates for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN112194548A introduces a groundbreaking advancement in this domain by disclosing a highly efficient preparation method for alpha-amino-gamma-butyrolactone compounds. This technology leverages a copper-catalyzed decarboxylative [3+2] cycloaddition reaction between 4-ethynyl carbonates and azlactones to construct molecular architectures featuring two adjacent continuous quaternary carbon centers. Such structural motifs are notoriously difficult to synthesize yet are prevalent in numerous natural products and drug candidates exhibiting antibacterial, anticonvulsive, and analgesic activities. By enabling the rapid assembly of these complex cores under mild conditions, this patent provides a powerful tool for medicinal chemists aiming to enrich lead compound libraries and accelerate the discovery of novel therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
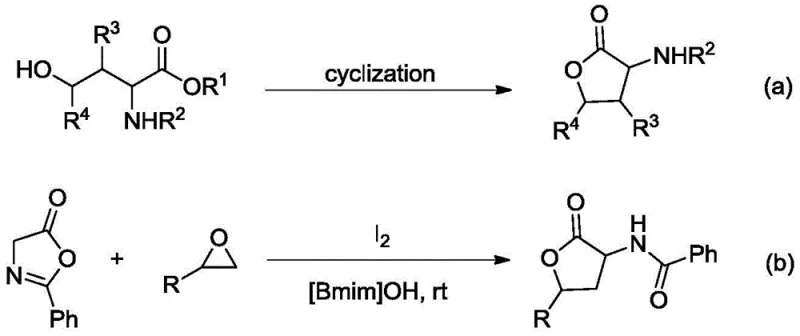
Historically, the construction of the alpha-amino-gamma-butyrolactone backbone has been fraught with significant synthetic challenges that hinder efficient commercial production. As illustrated in the prior art, traditional routes often rely on the intramolecular cyclization of gamma-hydroxy-alpha-amino acids, a process that typically necessitates multi-step sequences and harsh reaction conditions which can degrade sensitive functional groups. Alternatively, other established methods utilize iodine catalysis in ionic liquids such as [Bmim]OH to facilitate the reaction between azlactones and ethylene oxides. However, this approach suffers from severe drawbacks including the high cost and difficulty in recycling expensive ionic liquids, the toxicity and corrosivity of elemental iodine which requires extensive washing with sodium thiosulfate, and the inherent instability of ethylene oxide substrates. Furthermore, these conventional pathways often exhibit narrow substrate scope, limiting the structural diversity of the resulting lactones and complicating the subsequent functionalization required for drug development pipelines.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in CN112194548A offers a transformative solution by employing a decarboxylative [3+2] cycloaddition strategy that bypasses the need for unstable epoxides or toxic halogen catalysts. This novel route utilizes readily available 4-ethynyl carbonates and azlactones as starting materials, reacting them in the presence of a copper salt and an organic base to directly forge the desired lactone ring with exceptional precision. The process operates under remarkably mild temperatures ranging from -20 to 30°C, significantly reducing energy consumption and thermal stress on the reagents. By avoiding the use of ionic liquids and elemental iodine, the new method drastically simplifies the workup procedure, allowing for direct separation and purification of the product. This shift not only enhances the operational safety for laboratory personnel but also aligns with green chemistry principles by minimizing hazardous waste generation and improving the overall atom economy of the synthesis.

Mechanistic Insights into Copper-Catalyzed Decarboxylative Cycloaddition
The success of this synthetic transformation hinges on the precise orchestration of the copper catalyst and the organic base to facilitate a concerted decarboxylative [3+2] cycloaddition mechanism. In this catalytic cycle, the copper species likely activates the terminal alkyne of the 4-ethynyl carbonate, increasing its nucleophilicity or facilitating the formation of a reactive metallacycle intermediate. Simultaneously, the electron-deficient nature of the azlactone allows it to act as a dipolarophile or electrophilic partner in the cycloaddition event. The loss of carbon dioxide serves as a potent thermodynamic driving force, pushing the equilibrium towards the formation of the five-membered lactone ring. Crucially, the steric and electronic properties of the ligands surrounding the copper center, along with the choice of base, play a pivotal role in controlling the stereochemical outcome of the reaction. This level of control is essential for generating the two adjacent quaternary carbon centers with high diastereoselectivity, ensuring that the major product isomer is formed exclusively, which is vital for maintaining the biological activity profile of the final pharmaceutical candidate.
From an impurity control perspective, the mechanism inherently favors the desired pathway due to the irreversible nature of the decarboxylation step, which minimizes the formation of reversible side products often seen in equilibrium-driven reactions. The use of specific solvents like acetonitrile further stabilizes the transition state, preventing competing polymerization of the alkyne or hydrolysis of the azlactone. Optimization data within the patent indicates that copper trifluoromethanesulfonate paired with DMAP yields superior results compared to other metal salts or bases, suggesting a specific coordination geometry that maximizes orbital overlap during the bond-forming steps. This mechanistic understanding allows process chemists to fine-tune reaction parameters such as concentration and addition rate to suppress trace impurities, thereby ensuring that the final API intermediate meets the stringent purity specifications required by regulatory bodies for clinical trial materials.
How to Synthesize Alpha-Amino-Gamma-Butyrolactone Efficiently
To implement this high-yielding synthesis in a practical setting, operators must adhere to strict anhydrous conditions and precise stoichiometric controls to maximize the efficiency of the copper catalytic system. The general protocol involves dissolving the azlactone and copper catalyst in a dry polar aprotic solvent, followed by the sequential addition of the base and the ethynyl carbonate substrate under an inert atmosphere. Maintaining the reaction temperature at 0°C is critical during the initial mixing phase to manage the exotherm and ensure high stereoselectivity before allowing the mixture to stir to completion. Following the reaction, the removal of the solvent under reduced pressure leaves a crude residue that can be purified via standard column chromatography using a petroleum ether and ethyl acetate gradient. For detailed standardized operating procedures and specific troubleshooting tips regarding scale-up, please refer to the technical guide below.
- Dissolve the azlactone substrate and copper trifluoromethanesulfonate catalyst in dry acetonitrile under an inert argon atmosphere.
- Add the organic base (DMAP) and the 4-ethynyl carbonate reactant sequentially to the reaction mixture while maintaining the temperature at 0°C.
- Stir the reaction until completion, evaporate the solvent under reduced pressure, and purify the crude residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology represents a strategic opportunity to optimize the cost structure and reliability of the supply chain for complex heterocyclic intermediates. By eliminating the reliance on expensive and hard-to-source ionic liquids and toxic elemental iodine, the raw material costs associated with the synthesis are significantly reduced. Furthermore, the simplified purification process reduces the consumption of bulk solvents and silica gel, leading to substantial savings in waste disposal fees and processing time. The use of stable, commercially available starting materials like 4-ethynyl carbonates mitigates the risk of supply disruptions caused by the volatility of traditional reagents like ethylene oxide. This stability ensures a consistent flow of materials, allowing for better inventory planning and reduced safety stock requirements.
- Cost Reduction in Manufacturing: The elimination of costly ionic liquids and the reduction in catalyst loading to just 10 mol% directly lowers the bill of materials for each batch. Additionally, the mild reaction conditions negate the need for energy-intensive heating or cryogenic cooling systems, resulting in lower utility costs per kilogram of product. The high yield and selectivity mean less raw material is wasted on byproducts, further enhancing the overall process economics and making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Sourcing stable solid substrates rather than gaseous or unstable liquids simplifies logistics and storage requirements, reducing the risk of degradation during transit. The robustness of the reaction across a wide range of substrates means that a single manufacturing line can be adapted to produce various derivatives without extensive requalification, providing flexibility to meet changing demand from R&D teams. This adaptability ensures that the supply chain remains resilient even when specific precursor availability fluctuates.
- Scalability and Environmental Compliance: The absence of toxic iodine and corrosive reagents simplifies the environmental permitting process for manufacturing facilities, as there is no need for specialized scrubbing systems to handle hazardous off-gases. The straightforward workup procedure facilitates easier scale-up from gram to ton quantities without encountering the heat transfer or mixing issues common in viscous ionic liquid systems. This scalability ensures that the technology can seamlessly transition from pilot plant trials to full commercial production, securing long-term supply continuity for downstream API manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, derived directly from the experimental data and scope analysis provided in the patent documentation. Understanding these nuances is essential for process engineers looking to integrate this technology into existing manufacturing workflows. The answers reflect the specific findings regarding catalyst performance, substrate tolerance, and purification strategies observed during the development of this method.
Q: What are the critical reaction parameters for maximizing diastereoselectivity?
A: According to the patent data, the choice of solvent and base is critical. Acetonitrile combined with DMAP as the base and Cu(OTf)2 as the catalyst at 0°C provides the optimal balance of yield and diastereoselectivity (dr > 95:5).
Q: How does this method compare to traditional ionic liquid protocols?
A: This novel approach eliminates the need for expensive and difficult-to-remove ionic liquids like [Bmim]OH and toxic elemental iodine, significantly simplifying the downstream purification process and reducing environmental hazards.
Q: Is this synthesis suitable for large-scale production?
A: Yes, the reaction operates under mild conditions (-20 to 30°C) using commercially available catalysts and stable substrates, making it highly amenable to commercial scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amino-Gamma-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates that possess the structural complexity required for modern drug discovery. Our team of expert process chemists has extensively evaluated the copper-catalyzed decarboxylative cycloaddition technology and possesses the capability to execute this synthesis with precision and consistency. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with rigorous adherence to quality standards. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of alpha-amino-gamma-butyrolactone delivered meets the exacting requirements of the pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project timelines and budget constraints. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient methodology. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate a discussion regarding route feasibility assessments for your proprietary targets, ensuring a seamless path from benchtop innovation to commercial reality.
