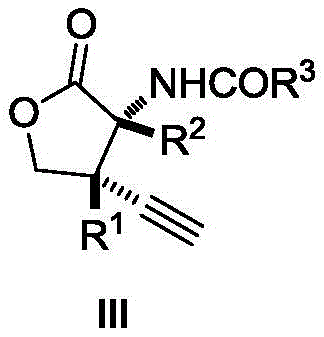
Advanced Copper-Catalyzed Synthesis of Alpha-Amino-Gamma-Butyrolactones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex heterocyclic scaffolds efficiently. Patent CN112194548B discloses a groundbreaking preparation method for α-amino-γ-butyrolactone compounds, a structural motif prevalent in bioactive natural products and drug candidates exhibiting antibacterial and analgesic properties. This technology addresses the critical challenge of constructing contiguous quaternary carbon centers, a feat often difficult to achieve with high stereocontrol using conventional chemistry. By leveraging a copper-catalyzed decarboxylative [3+2] cycloaddition strategy, this innovation enables the direct assembly of highly functionalized lactone cores from readily available azlactones and 4-ethynyl carbonates. For R&D directors and process chemists, this represents a significant leap forward in synthetic efficiency, offering a pathway to diversify lead compound libraries with unprecedented speed and precision while maintaining rigorous purity standards required for drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the α-amino-γ-butyrolactone skeleton has been plagued by significant synthetic bottlenecks that hinder large-scale production. Traditional routes often rely on the intramolecular cyclization of γ-hydroxy-α-amino acids, which necessitates tedious multi-step sequences to prepare the sensitive hydroxy-acid intermediates. Furthermore, the cyclization step itself frequently demands harsh reaction conditions that can compromise the integrity of delicate functional groups. An alternative prior art method utilizes iodine catalysis in ionic liquids like [Bmim]OH; however, this approach is economically and environmentally unsustainable due to the high cost of ionic solvents and the toxicity of elemental iodine. The requirement for extensive post-reaction washing with sodium thiosulfate to remove residual iodine adds substantial operational complexity and waste generation, making these legacy processes ill-suited for modern green manufacturing standards.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a mild copper-catalyzed system that operates efficiently at temperatures ranging from -20 to 30°C. This transformative reaction directly couples 4-ethynyl carbonates with azlactones, bypassing the need for unstable epoxide substrates or pre-functionalized hydroxy acids. The process achieves exceptional yields, reported up to 98%, with outstanding diastereoselectivity exceeding 95:5 dr in optimized cases. By eliminating the need for expensive ionic liquids and toxic halogen catalysts, this route drastically simplifies the workup procedure to a straightforward solvent evaporation and column chromatography. This shift not only enhances the safety profile of the synthesis but also opens the door for broader substrate application, allowing for the rapid generation of diverse molecular architectures essential for medicinal chemistry campaigns.

Mechanistic Insights into Copper-Catalyzed Decarboxylative Cycloaddition
The core of this technological advancement lies in the precise activation of the 4-ethynyl carbonate by the copper catalyst, which facilitates a concerted decarboxylative [3+2] cycloaddition mechanism. The copper center likely coordinates with the alkyne moiety, increasing its electrophilicity and promoting nucleophilic attack by the azlactone enolate generated in situ by the organic base. This synergistic interaction lowers the activation energy barrier for the ring-closing step, allowing the reaction to proceed smoothly under mild thermal conditions. The simultaneous extrusion of carbon dioxide serves as a thermodynamic driving force, pushing the equilibrium towards the formation of the five-membered lactone ring. This mechanistic elegance ensures that the reaction is not only fast but also highly atom-economical, minimizing the formation of byproducts that typically complicate purification in less selective cyclization protocols.
From an impurity control perspective, the high diastereoselectivity observed is attributed to the rigid transition state imposed by the copper-ligand complex during the bond-forming events. The steric environment around the metal center effectively discriminates between competing facial approaches of the substrates, ensuring the preferential formation of the desired stereoisomer with contiguous quaternary centers. This level of stereocontrol is critical for pharmaceutical applications where biological activity is often dependent on specific three-dimensional configurations. By avoiding harsh acidic or basic conditions that could lead to racemization or epimerization, this method preserves the optical purity of the starting materials and the final product. Consequently, the resulting α-amino-γ-butyrolactones exhibit a clean impurity profile, reducing the burden on downstream purification processes and ensuring consistent quality for regulatory submissions.
How to Synthesize Alpha-Amino-Gamma-Butyrolactone Efficiently
Implementing this synthesis requires careful attention to reagent quality and reaction parameters to maximize the benefits of the copper catalytic cycle. The protocol is designed to be operationally simple, utilizing standard laboratory equipment and commercially sourced chemicals without the need for specialized inert gas manifolds beyond standard Schlenk techniques. The following guide outlines the generalized procedure derived from the patent examples, serving as a foundational reference for process optimization. For detailed standardized synthesis steps and specific parameter adjustments for different substrates, please refer to the structured guide below.
- Dissolve azlactone and copper salt catalyst in an organic solvent such as acetonitrile under inert atmosphere.
- Add organic base and 4-ethynyl carbonate substrate sequentially while maintaining temperature between -20 to 30°C.
- Stir the reaction mixture until completion, then remove solvent and purify the crude residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology offers tangible strategic advantages over legacy synthetic routes. The elimination of exotic reagents like ionic liquids and elemental iodine translates directly into reduced raw material costs and simplified logistics. Since the catalyst and base are commodity chemicals available from multiple global suppliers, the risk of supply chain disruption is minimized, ensuring continuity of production even during market fluctuations. Furthermore, the mild reaction conditions reduce energy consumption associated with heating or cryogenic cooling, contributing to a lower overall carbon footprint for the manufacturing process. These factors combine to create a more resilient and cost-effective supply chain for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive ionic solvents and toxic iodine catalysts with affordable copper salts and organic bases results in significant cost savings per kilogram of product. The simplified workup procedure eliminates the need for large volumes of aqueous wash solutions and specialized waste treatment for heavy metals or halogens, further lowering operational expenditures. Additionally, the high yields achieved reduce the amount of starting material required, maximizing the return on investment for every batch produced. This economic efficiency makes the process highly competitive for commercial scale-up in cost-sensitive markets.
- Enhanced Supply Chain Reliability: The substrates used in this reaction, specifically 4-ethynyl carbonates and azlactones, are chemically stable and easier to store and transport compared to the unstable epoxides required by older methods. This stability reduces the risk of material degradation during shipping and long-term storage, ensuring consistent quality upon arrival at the manufacturing site. The use of common organic solvents like acetonitrile or dichloromethane means that solvent recovery and recycling infrastructure can be easily integrated, reducing dependency on fresh solvent supplies. This robustness ensures a steady flow of materials through the production pipeline without unexpected delays.
- Scalability and Environmental Compliance: The reaction's tolerance to a wide range of functional groups and its operation at near-ambient temperatures make it inherently scalable from gram to ton quantities without significant re-engineering. The absence of highly toxic reagents simplifies environmental compliance and waste disposal, aligning with increasingly stringent global regulations on chemical manufacturing. The high atom economy of the decarboxylative process minimizes waste generation, supporting sustainability goals and reducing the environmental impact of the facility. This scalability ensures that the technology can grow with demand, supporting everything from early-stage clinical trials to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for potential partners evaluating this route for their own pipelines. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What are the optimal reaction conditions for high diastereoselectivity?
A: The patent data indicates that using Copper(II) trifluoromethanesulfonate as the catalyst and DMAP as the base in acetonitrile at 0°C provides the best balance of yield and diastereoselectivity.
Q: How does this method compare to traditional iodine-catalyzed routes?
A: Unlike iodine-catalyzed methods requiring expensive ionic liquids and toxic reagents, this copper-catalyzed approach uses commercially available solvents and avoids hazardous waste streams, significantly simplifying downstream processing.
Q: Is the substrate scope limited to specific aryl groups?
A: The methodology demonstrates broad universality, successfully accommodating various substituents including electron-donating and electron-withdrawing groups on both the azlactone and carbonate components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amino-Gamma-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed technology in accelerating drug discovery and development programs. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the bench to the plant. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha-amino-gamma-butyrolactone intermediate meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this novel route for your specific needs, delivering high-quality intermediates that drive your research forward.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in supplying complex pharmaceutical intermediates with reliability and precision.
