Advanced Fragment Condensation Strategy for Commercial Scale-up of Plecanatide Manufacturing
Introduction to Advanced Plecanatide Manufacturing Technologies
The pharmaceutical landscape for gastrointestinal disorders continues to evolve, driven by the demand for high-purity peptide therapeutics such as Plecanatide. Patent CN113412272A introduces a transformative approach to the synthesis of this guanylate cyclase-C agonist, addressing critical bottlenecks inherent in traditional linear peptide assembly. This intellectual property delineates a robust non-linear solid-phase peptide synthesis (SPPS) methodology that strategically divides the 16-residue sequence into manageable fragments. By leveraging a 5+11 fragment condensation strategy, the disclosed process significantly mitigates the accumulation of deletion impurities that typically plague long-chain peptide synthesis. For R&D directors and process chemists, this represents a pivotal shift towards more predictable and controllable manufacturing workflows. The technical nuances of utilizing distinct resin systems for different peptide segments underscore a sophisticated understanding of steric effects and coupling kinetics, ultimately paving the way for cost reduction in pharmaceutical intermediates manufacturing and ensuring a reliable supply of high-quality active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional linear solid-phase peptide synthesis, while standard for shorter sequences, encounters profound challenges when applied to complex 16-residue peptides like Plecanatide. As the peptide chain elongates on the solid support, steric hindrance and intermolecular aggregation significantly impede the diffusion of reagents to the reactive sites. Specifically, in the context of Plecanatide, the coupling efficiency of amino acids from the Val8 position onwards tends to decrease markedly. This phenomenon leads to incomplete reactions at critical junctions, resulting in the formation of deletion-type impurities where one or more amino acids are missing from the sequence. These impurities are structurally similar to the target molecule, often exhibiting close elution peaks during purification, which necessitates multiple, resource-intensive chromatographic cycles. Consequently, the overall isolated yield diminishes, and the burden on downstream purification processes increases, creating a substantial bottleneck for commercial scale-up of complex pharmaceutical intermediates. Furthermore, the reliance on a single resin type for the entire sequence limits the flexibility in choosing optimal cleavage conditions, potentially compromising the integrity of sensitive side-chain protecting groups.
The Novel Approach
The innovative methodology described in the patent data circumvents these limitations through a strategic fragmentation of the synthetic route. By synthesizing the N-terminal 5-amino acid unit (Fragment A) and the C-terminal 11-amino acid unit (Fragment B) separately, the process effectively resets the steric clock, ensuring high coupling efficiency for each segment. Fragment A is constructed on 2-chlorotrityl chloride (2-ClTrt) resin, which allows for mild acid cleavage, preserving acid-labile protecting groups if necessary. Fragment B is assembled on Wang resin, providing a stable anchor for the longer sequence. The subsequent condensation of these two pre-verified, high-purity fragments in solution phase drastically reduces the probability of deletion impurities compared to stepwise linear addition. This modular approach not only enhances the purity of the crude linear peptide but also simplifies the purification profile, thereby improving the overall process mass intensity. For procurement managers, this translates to a more efficient utilization of raw materials and a reduction in the consumption of expensive chromatography media, directly impacting the cost structure of the final API.
Mechanistic Insights into Non-Linear Peptide Assembly and Oxidative Folding
The core of this improved process lies in the precise orchestration of resin chemistry and orthogonal protection strategies. The synthesis initiates with the preparation of Fragment A, typically Fmoc-Asn(Trt)-Asp(OtBu)-Glu(OtBu)-Cys(Trt)-Glu(OtBu)-OH, on 2-ClTrt resin. The use of 2-ClTrt resin is mechanistically significant because it permits cleavage under very mild acidic conditions (e.g., 1% TFA in DCM), which prevents premature removal of acid-sensitive side-chain protecting groups like Trt or OtBu during the fragment isolation stage. Conversely, Fragment B, encompassing residues 6 through 16, is anchored to Wang resin. This resin requires stronger acidic conditions for cleavage, which are reserved for the final global deprotection step after the two fragments have been coupled. The coupling of Fragment A to the amine terminus of the resin-bound Fragment B is facilitated by potent coupling reagents such as DIC-HOBt or HBTU-HOBt. This solution-phase or semi-solid phase condensation is kinetically favorable because both reacting species are relatively short and soluble, minimizing the diffusion barriers observed in long-chain linear SPPS.
Following the assembly of the full linear sequence and global deprotection using a cocktail of trifluoroacetic acid, triisopropylsilane, DODT, and water, the critical oxidative folding phase commences. The linear peptide contains four cysteine residues protected as acetamidomethyl (Acm) or trityl (Trt) derivatives, which must be selectively deprotected and oxidized to form two specific disulfide bonds. The patent discloses a sequential oxidation strategy to control the regioselectivity of this process. Initially, a monocyclic intermediate is formed using a dilute hydrogen peroxide solution, which selectively oxidizes specific free thiol groups generated during the cleavage or deprotection phase. This step creates the first disulfide bridge, constraining the peptide conformation and facilitating the subsequent formation of the second bridge. The final bicyclization is achieved using an iodine solution, which simultaneously removes the Acm protecting groups and promotes the formation of the second disulfide bond. This controlled, step-wise oxidation is crucial for avoiding the formation of scrambled disulfide isomers, which are difficult to separate and represent a significant quality risk.
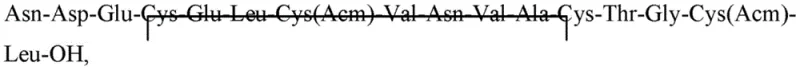

How to Synthesize Plecanatide Efficiently
The execution of this synthesis requires meticulous attention to reaction parameters, particularly regarding temperature and reagent stoichiometry, to ensure reproducibility at scale. The process begins with the independent solid-phase synthesis of the two fragments, where coupling times are optimized to be as short as 20 minutes at elevated temperatures (45-50°C) for Fragment B, demonstrating high reactivity. Once the fragments are prepared and verified, they are coupled, and the full-length peptide is cleaved from the resin. The subsequent oxidative folding steps are performed in dilute aqueous solutions to prevent intermolecular aggregation, with pH adjustments playing a critical role in controlling the reaction rate. Detailed standardized operating procedures for each reaction step, including specific solvent ratios and purification gradients, are essential for technology transfer.
- Synthesize Fragment A (5 amino acids) on 2-ClTrt resin and Fragment B (11 amino acids) on Wang resin using standard Fmoc chemistry.
- Couple Fragment A to the resin-bound Fragment B, followed by global deprotection and cleavage using TFA/TIS/DODT/water cocktail.
- Perform sequential oxidative cyclization: first monocyclization with hydrogen peroxide, followed by bicyclization with iodine solution.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this fragment condensation strategy offers substantial advantages that resonate deeply with supply chain stability and cost management objectives. The primary driver of value is the significant enhancement in crude peptide purity. By avoiding the cumulative coupling failures associated with linear synthesis, the process generates a linear precursor with a markedly reduced burden of deletion impurities. This improvement directly correlates to a streamlined purification workflow, where fewer chromatographic passes are required to achieve the stringent purity specifications demanded by regulatory bodies. For procurement managers, this efficiency translates into tangible cost reduction in pharmaceutical intermediates manufacturing, as it lowers the consumption of high-purity solvents, chromatography resins, and labor hours associated with extensive purification campaigns. Furthermore, the modular nature of the synthesis allows for parallel processing; Fragment A and Fragment B can be manufactured simultaneously in different reactors, effectively halving the production lead time for the linear precursor compared to a sequential linear approach.
Enhanced supply chain reliability is another critical benefit derived from this robust synthetic route. The use of widely available resins like 2-ClTrt and Wang, coupled with standard coupling reagents such as DIC and HOBt, ensures that the supply of raw materials is not dependent on exotic or single-source catalysts. This diversification of the supply base mitigates the risk of production stoppages due to material shortages. Additionally, the process demonstrates excellent scalability, moving seamlessly from laboratory gram-scale experiments to multi-kilogram commercial batches without fundamental changes to the chemistry. The ability to perform coupling reactions at slightly elevated temperatures (up to 50°C) further accelerates the throughput, allowing manufacturers to respond more agilely to market demand fluctuations. Regarding environmental compliance, the reduction in purification cycles inherently decreases the volume of organic waste generated, aligning with modern green chemistry principles and reducing the costs associated with waste disposal and solvent recovery.
- Cost Reduction in Manufacturing: The elimination of extensive purification steps required to remove deletion impurities significantly lowers the operational expenditure. By achieving higher crude purity through the 5+11 fragment strategy, the consumption of expensive reverse-phase chromatography media and high-grade solvents is drastically reduced. This efficiency gain allows for a more competitive pricing structure for the final API, providing a strategic advantage in cost-sensitive markets without compromising on quality standards.
- Enhanced Supply Chain Reliability: The decoupling of the synthesis into two parallel fragment streams reduces the overall critical path time for production. This parallelization capability ensures that delays in one segment do not necessarily halt the entire process, thereby enhancing the predictability of delivery schedules. Moreover, the reliance on commodity chemicals and standard resins ensures a resilient supply chain that is less vulnerable to geopolitical or logistical disruptions affecting specialized reagents.
- Scalability and Environmental Compliance: The process is designed with commercial scale-up in mind, utilizing reaction conditions that are easily manageable in large-scale stainless steel reactors. The simplified purification profile reduces the environmental footprint of the manufacturing process by minimizing solvent usage and waste generation. This alignment with sustainability goals not only reduces compliance costs but also appeals to environmentally conscious partners and stakeholders in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved Plecanatide synthesis process. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is the 5+11 fragment strategy superior to linear synthesis for Plecanatide?
A: Linear synthesis often suffers from decreased coupling efficiency and increased deletion impurities as the chain lengthens, particularly after the Val8 residue. The fragment condensation approach mitigates steric hindrance and ensures higher crude purity.
Q: What specific resins are utilized in this improved process?
A: The process employs 2-chlorotrityl chloride (2-ClTrt) resin for the N-terminal 5-amino acid fragment and Wang resin for the C-terminal 11-amino acid fragment, optimizing cleavage conditions for each segment.
Q: How are the disulfide bonds formed in this protocol?
A: The protocol utilizes a two-step oxidative folding strategy: initial monocyclization using dilute hydrogen peroxide to form the first disulfide bridge, followed by bicyclization using an iodine solution to close the second ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Plecanatide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in ensuring the consistent supply of high-value therapeutic peptides. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are equipped with state-of-the-art facilities capable of handling the specific resin chemistries and oxidative folding conditions required for Plecanatide, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality. Our commitment to excellence extends beyond mere manufacturing; we act as a strategic partner, optimizing processes to maximize yield and minimize environmental impact, thereby securing your supply chain against future volatility.
We invite you to engage with our technical procurement team to discuss how this advanced fragment condensation technology can be leveraged for your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits associated with this improved process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-purity Plecanatide that meets the exacting standards of the global pharmaceutical industry.
