Advanced Two-Step HPLC Purification Strategy for High-Purity Procatide Manufacturing
Advanced Two-Step HPLC Purification Strategy for High-Purity Procatide Manufacturing
The pharmaceutical industry continuously demands higher purity standards for complex peptide therapeutics, particularly for guanylate cyclase-C receptor agonists like procatide, an analogue of uroguanylin designed for treating chronic idiopathic constipation. A significant technological breakthrough in this domain is documented in patent CN114315973A, which discloses a sophisticated method for purifying procatide that addresses the critical limitations of prior art. This innovation leverages a dual-gradient high-performance liquid chromatography (HPLC) approach, transitioning from an ammonium acetate-based mobile phase to an acetic acid-based system, thereby achieving exceptional separation efficiency. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding this purification architecture is vital, as it directly correlates to the feasibility of commercial-scale production and the economic viability of the final active pharmaceutical ingredient (API). The method described herein not only enhances the purity profile but also streamlines the downstream processing workflow, offering a robust solution for manufacturers aiming to secure a stable supply of high-quality peptide intermediates.
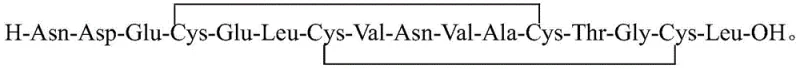
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification strategies for cyclic peptides such as procatide have historically been plagued by inefficiencies stemming from the complexity of solid-phase synthesis byproducts. Conventional single-step purification protocols often fail to adequately resolve the target peptide from structurally similar impurities, such as deletion sequences, truncated peptides, and diastereomers formed during the cyclization process. These impurities possess physicochemical properties remarkably similar to the target molecule, making their removal via standard isocratic or simple gradient elution extremely challenging and often resulting in co-elution. Consequently, the final product obtained through these legacy methods frequently exhibits suboptimal purity levels, necessitating repetitive purification cycles that drastically reduce overall yield and inflate manufacturing costs. Furthermore, the use of non-volatile buffers or harsh pH conditions in older methodologies can complicate the subsequent lyophilization steps, leading to salt contamination and extended processing times that hinder the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these outdated techniques, the novel approach detailed in the patent introduces a sequential, orthogonal purification strategy that fundamentally alters the impurity landscape. By implementing a first-stage HPLC purification using ammonium acetate adjusted to pH 8.3, the method effectively separates the bulk of hydrophobic and charge-variant impurities under mildly alkaline conditions. This is immediately followed by a second, refining HPLC step utilizing 2% acetic acid, which shifts the ionization state of the peptide and the stationary phase interactions, allowing for the resolution of closely eluting peaks that survived the first pass. This two-dimensional-like separation logic within a linear workflow ensures that the collected fractions possess a purity exceeding 98%, a benchmark that is difficult to achieve with single-pass methods. The strategic switch in mobile phase chemistry minimizes the risk of peak tailing and broadening, ensuring sharp, well-defined elution profiles that maximize the recovery of the active ingredient while rigorously excluding contaminants.
Mechanistic Insights into Dual-Gradient HPLC Purification
The core mechanism driving the success of this purification protocol lies in the precise manipulation of electrostatic and hydrophobic interactions between the procatide molecule and the octadecylsilane (C18) stationary phase. In the initial purification stage, the use of 50mmol/L ammonium acetate at pH 8.3 creates an environment where the acidic residues of the peptide, such as Aspartic acid and Glutamic acid, are largely deprotonated, altering the overall charge distribution of the molecule. This specific pH condition optimizes the selectivity of the C18 column for the target cyclic structure against linear precursors and open-chain byproducts, as the cyclic conformation presents a distinct hydrophobic surface area compared to its unfolded counterparts. The gradient elution with acetonitrile then progressively disrupts these hydrophobic interactions, eluting the target peptide at a specific retention time that is well-resolved from early-eluting polar impurities and late-eluting highly hydrophobic aggregates.
Following the collection of the first purified substance, the mechanism shifts in the second stage to exploit different selectivity parameters through the introduction of an acidic mobile phase. The transition to 2% acetic acid protonates the carboxyl groups on the peptide surface, reducing electrostatic repulsion and enhancing hydrophobic binding to the silica matrix. This change in ionization state is critical for separating diastereomers or oxidation variants that may have co-eluted in the alkaline phase. The careful control of the gradient slope in this second dimension ensures that the target procatide is eluted with maximum resolution from any remaining trace impurities. This mechanistic duality—switching from alkaline to acidic conditions—acts as a powerful filter, ensuring that the final product meets the stringent purity specifications required for clinical applications, while the subsequent rotary evaporation at controlled temperatures (30-33°C) preserves the structural integrity of the thermally sensitive peptide bonds.
How to Synthesize Procatide Efficiently
The implementation of this purification workflow requires precise adherence to chromatographic parameters to replicate the high yields and purity reported in the patent data. The process begins with the filtration of the crude cyclization solution to remove particulate matter, followed by the critical first HPLC run where fractions with purity greater than 80% are selectively pooled. These pooled fractions are then diluted with purified water to adjust the solvent strength before being subjected to the second, high-resolution purification step. The detailed operational parameters, including flow rates of 50mL/min and specific detection wavelengths of 220nm, are essential for monitoring the elution profile in real-time. For a comprehensive breakdown of the exact gradient tables and equipment setup required to execute this protocol in a GMP environment, please refer to the standardized synthesis guide below.
- Filter the cyclized procatide solution through a 0.45 μm microporous membrane to obtain the crude peptide solution.
- Perform first HPLC purification using a C18 stationary phase with ammonium acetate (pH 8.3) and acetonitrile gradient elution.
- Conduct second HPLC purification on the collected fractions using acetic acid and acetonitrile, followed by rotary evaporation and freeze-drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates into tangible strategic advantages regarding cost stability and supply continuity. The primary benefit lies in the substantial improvement in process yield, which directly impacts the cost of goods sold (COGS) by maximizing the output from expensive starting materials. Unlike traditional methods that may suffer from significant product loss during repetitive purification attempts, this streamlined two-step approach consolidates the purification into a predictable, high-efficiency workflow. This reliability reduces the risk of batch failures and minimizes the need for reprocessing, which is a common hidden cost in peptide manufacturing. By securing a method that consistently delivers high purity in fewer steps, organizations can negotiate better pricing structures with their reliable pharmaceutical intermediates supplier, as the reduced processing time and resource consumption lower the overall manufacturing burden.
- Cost Reduction in Manufacturing: The elimination of redundant purification cycles and the optimization of mobile phase usage lead to significant operational savings. By achieving high purity (>98%) in a defined two-step sequence, the method reduces the consumption of costly chromatography resins and organic solvents like acetonitrile. Furthermore, the use of volatile buffers such as ammonium acetate and acetic acid simplifies the downstream removal of salts via rotary evaporation, avoiding the need for extensive dialysis or desalting columns that add time and expense. This efficiency drives down the unit cost of the final API intermediate, making the final drug product more competitive in the market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this purification method ensures a consistent supply of high-quality procatide, mitigating the risks associated with batch-to-batch variability. The clear definition of critical process parameters, such as pH adjustment and gradient slopes, allows for easy technology transfer between manufacturing sites, ensuring that supply is not bottlenecked by a single facility's limitations. This scalability is crucial for meeting the growing global demand for gastrointestinal therapeutics, as it enables manufacturers to ramp up production volumes rapidly in response to market needs. A stable and predictable purification process means fewer delays in filling orders, providing downstream partners with the confidence to plan their own production schedules effectively.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the method offers distinct advantages due to its reliance on relatively benign mobile phase components. Ammonium acetate and acetic acid are easier to handle and dispose of compared to harsher acids or non-volatile salts often used in older peptide purification protocols. The ability to recover and recycle acetonitrile through the rotary evaporation step further aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with environmental standards facilitates smoother regulatory approvals and reduces the liability associated with hazardous waste management, making the entire supply chain more sustainable and resilient against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this procatide purification method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the operational nuances of the dual-HPLC strategy. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for large-scale commercial production.
Q: Why is a dual-mobile phase system used for procatide purification?
A: The dual-system utilizes orthogonal selectivity; the first step uses alkaline ammonium acetate (pH 8.3) to separate major impurities, while the second acidic acetic acid step refines purity to >98% by resolving closely related peptide variants.
Q: What is the expected yield of this purification method?
A: According to patent data, this optimized method consistently achieves yields between 71% and 76%, significantly outperforming traditional single-step purification techniques which often suffer from substantial product loss.
Q: What stationary phase is recommended for this process?
A: The process specifies octadecylsilane chemically bonded silica filler (C18) with dimensions of 50mm × 250 mm, providing the necessary hydrophobic interaction for effective separation of the 16-amino acid cyclic peptide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Procatide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced purification technologies in delivering high-value pharmaceutical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex peptide synthesis and purification routes like the one described in CN114315973A can be successfully translated from the lab to the plant. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of procatide meets the highest international standards. Our commitment to technical excellence ensures that our clients receive materials that are not only chemically pure but also consistent in quality, supporting their regulatory filings and clinical development timelines.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall procurement costs while maintaining superior quality. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our capacity to be your trusted partner in the efficient and reliable supply of complex peptide intermediates.
