Advanced Manufacturing of Prucalopride Intermediates via Green Catalytic Routes
Advanced Manufacturing of Prucalopride Intermediates via Green Catalytic Routes
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of complex active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN108976216B, which outlines a novel preparation method for Prucalopride, a selective 5-HT4 receptor agonist used for treating chronic constipation. This technical disclosure moves away from hazardous traditional chemistries, offering a streamlined process that begins with the readily available 4-nitrobenzaldehyde. By integrating protective group strategies, catalytic hydrogenation, and a final oxidative amidation step, this methodology addresses critical pain points regarding toxicity, waste generation, and operational complexity. For R&D directors and supply chain leaders, understanding this shift is vital for securing a reliable pharmaceutical intermediates supplier capable of delivering high-quality materials with reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prucalopride and its key benzofuran intermediates has relied on chemically aggressive and operationally demanding protocols. As illustrated in prior art schemes, early methods often utilized 4-aminosalicylic acid or 2-hydroxy-4-acetaminobenzoic acid methyl ester as starting materials, necessitating the use of elemental bromine and large quantities of zinc powder for cyclization. These reagents pose severe safety risks and generate substantial heavy metal waste, complicating downstream purification and environmental compliance. Furthermore, alternative routes reported in literature have depended on osmium tetroxide for oxidation and ethyl chloroformate for activation, both of which are highly toxic and require specialized handling infrastructure. Perhaps most critically, some established pathways utilize butyl lithium for lithiation steps, requiring cryogenic conditions at minus 78°C and generating isomeric by-products that demand tedious column chromatography for separation.
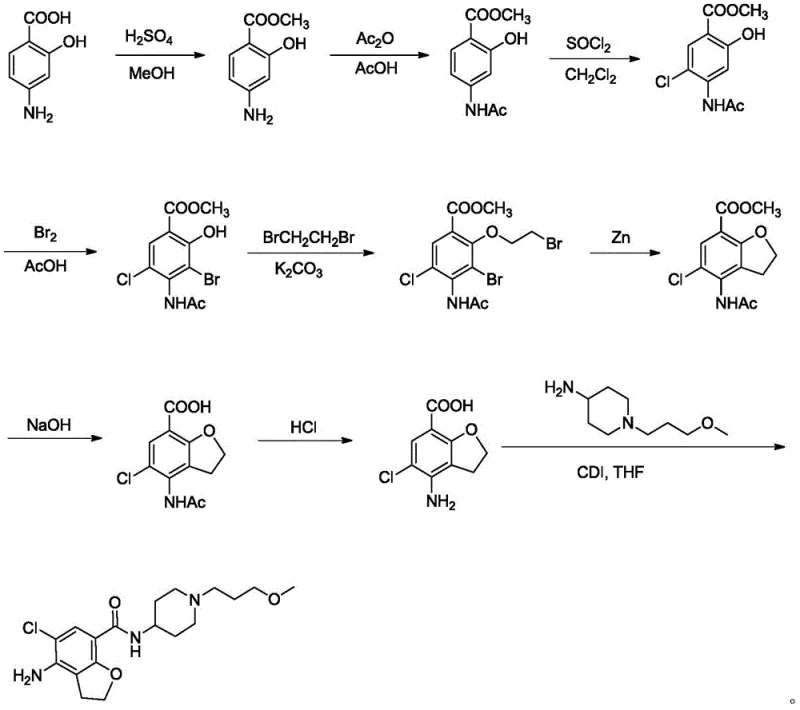
The Novel Approach
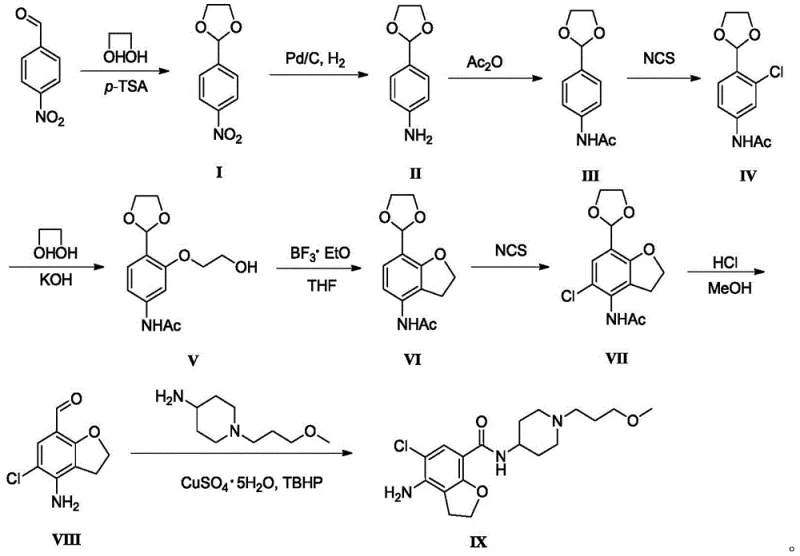
In stark contrast, the innovative route disclosed in the patent data adopts a fundamentally safer and more efficient strategy centered on 4-nitrobenzaldehyde. This approach strategically employs an aldehyde protection step to form a dioxolane derivative, which stabilizes the molecule during subsequent transformations. The process replaces dangerous halogenation and reduction agents with milder alternatives, such as N-chlorosuccinimide (NCS) for regioselective chlorination and palladium on carbon (Pd/C) for nitro reduction. The culmination of the synthesis involves a copper-catalyzed oxidative amidation, which directly couples the aldehyde intermediate with the piperidine side chain. This eliminates the need for pre-activating the carboxylic acid with hazardous coupling reagents. The overall pathway is characterized by mild reaction temperatures ranging from 70°C to 90°C, avoiding the energy-intensive cryogenic setups of the past and ensuring a cleaner reaction profile with fewer by-products.
Mechanistic Insights into Copper-Catalyzed Oxidative Amidation
The cornerstone of this synthetic advancement lies in the final coupling step, which utilizes a copper sulfate and tert-butyl hydroperoxide (TBHP) system to drive the oxidative amidation. Mechanistically, this transformation bypasses the traditional two-step sequence of oxidizing the aldehyde to a carboxylic acid followed by amide bond formation. Instead, the copper catalyst facilitates the direct activation of the aldehyde C-H bond in the presence of the amine nucleophile. The TBHP serves as a terminal oxidant, regenerating the active copper species and driving the equilibrium toward the amide product. This mechanism is particularly advantageous for industrial applications because it reduces the total number of unit operations and minimizes solvent usage. By conducting this reaction in acetonitrile at 80°C, the process achieves high conversion rates while maintaining the integrity of the sensitive dihydrobenzofuran core, ensuring that the final API intermediate meets stringent quality specifications without the need for extensive purification.
Furthermore, the regioselectivity observed during the chlorination steps is a critical factor in controlling the impurity profile. The use of N-chlorosuccinimide (NCS) in polar aprotic solvents like DMF allows for precise substitution at the ortho-position relative to the acetamido group. This specificity prevents the formation of unwanted isomers that typically plague electrophilic aromatic substitutions on activated rings. In the cyclization step, boron trifluoride diethyl etherate acts as a Lewis acid to promote the intramolecular Friedel-Crafts alkylation, closing the five-membered furan ring efficiently. The combination of these mechanistic features results in a process that is not only chemically elegant but also robust enough for commercial scale-up of complex heterocycles, providing manufacturers with a predictable and reproducible workflow.
How to Synthesize Prucalopride Intermediates Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly during the protection and cyclization phases. The initial formation of the dioxolane ring sets the stage for the entire sequence, requiring azeotropic removal of water to drive the equilibrium forward. Subsequent steps involve standard workup procedures such as aqueous washing and recrystallization, which are far more scalable than chromatographic techniques. The patent details specific molar ratios and temperature ranges that optimize yield and purity, such as maintaining the chlorination reaction between 70°C and 90°C for 4 to 7 hours. For technical teams looking to implement this route, the following standardized guide outlines the critical operational milestones required to achieve consistent results.
- Protect the aldehyde group of 4-nitrobenzaldehyde using ethylene glycol and p-TSA to form the dioxolane derivative.
- Reduce the nitro group to an amine using Pd/C catalytic hydrogenation, followed by acetylation to protect the amine.
- Perform regioselective chlorination, nucleophilic substitution with ethylene glycol, and BF3-catalyzed intramolecular cyclization to form the benzofuran core.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers transformative benefits that extend beyond simple chemical efficiency. The elimination of exotic and hazardous reagents translates directly into reduced raw material costs and lower expenses associated with hazardous waste disposal. Traditional methods relying on osmium tetroxide or butyl lithium incur significant overhead due to the need for specialized storage, handling protocols, and neutralization processes. By substituting these with commodity chemicals like ethylene glycol, acetic anhydride, and NCS, the manufacturing process becomes inherently more cost-effective and less susceptible to supply chain disruptions caused by the scarcity of specialty reagents. This shift enables cost reduction in API manufacturing by simplifying the bill of materials and reducing the regulatory burden associated with toxic substance management.
- Cost Reduction in Manufacturing: The most significant economic driver of this new process is the complete removal of column chromatography from the purification workflow. In traditional fine chemical synthesis, chromatographic separation is a major bottleneck that limits batch size and consumes vast quantities of silica gel and solvents. By designing a route where intermediates can be purified via crystallization or simple extraction, the process drastically reduces solvent consumption and processing time. Additionally, the avoidance of expensive transition metal catalysts like osmium and the use of inexpensive copper salts for the final coupling step further lowers the direct material costs, allowing for substantial margin improvements in large-scale production.
- Enhanced Supply Chain Reliability: The reliance on 4-nitrobenzaldehyde as the starting material ensures a stable and secure supply chain foundation. Unlike specialized precursors that may have limited global suppliers, 4-nitrobenzaldehyde is a bulk chemical produced by numerous vendors worldwide, mitigating the risk of single-source dependency. Furthermore, the mild reaction conditions (ambient to moderate temperatures) reduce the strain on production equipment, minimizing downtime for maintenance and allowing for faster turnaround times between batches. This operational flexibility is crucial for reducing lead time for high-purity APIs, enabling manufacturers to respond more agilely to market demand fluctuations without compromising on delivery schedules.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this route align perfectly with modern environmental, health, and safety (EHS) standards. The absence of heavy metal waste (from zinc or osmium) and the reduction of halogenated solvent usage simplify wastewater treatment and废气 (exhaust gas) management. This compliance advantage is increasingly important as global regulations tighten around pharmaceutical manufacturing emissions. The process is inherently scalable from kilogram to multi-ton scales without requiring fundamental changes to the reaction engineering, ensuring that high-purity pharmaceutical intermediates can be produced consistently regardless of batch size, thereby securing long-term supply continuity for downstream drug product manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of safety, yield, and operational simplicity.
Q: How does this new synthesis route improve safety compared to conventional methods?
A: The patented route eliminates the use of highly toxic reagents such as osmium tetroxide, bromine, and explosive butyl lithium found in traditional schemes. It operates under mild temperatures (70-90°C) rather than cryogenic conditions (-78°C), significantly reducing operational hazards and waste treatment costs.
Q: What are the key advantages for large-scale commercial production?
A: The process avoids column chromatography for purification, relying instead on crystallization and standard workups. This simplifies equipment requirements, drastically reduces solvent consumption, and enhances throughput, making it ideal for multi-ton commercial scale-up.
Q: Does this method ensure high purity for pharmaceutical applications?
A: Yes, the route is designed to minimize by-product formation and avoid isomer generation during chlorination steps. The final oxidative amidation step utilizes a copper-catalyzed system that ensures high conversion rates and clean product profiles suitable for API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prucalopride Supplier
The technical advancements detailed in patent CN108976216B represent a significant leap forward in the manufacturing of gastrointestinal therapeutics, yet translating laboratory success into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling the specific thermal and pressure requirements of oxidative amidation and Friedel-Crafts cyclizations, ensuring that every batch meets stringent purity specifications. With rigorous QC labs and a commitment to green chemistry, we guarantee that our Prucalopride intermediates are not only cost-effective but also compliant with the highest international regulatory standards.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for this critical API. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, ensuring that your project benefits from the safest, most efficient, and economically viable synthesis strategy available in the market.
