Advanced Refining Technology for Prucalopride: Ensuring High Purity and Scalable Production
Advanced Refining Technology for Prucalopride: Ensuring High Purity and Scalable Production
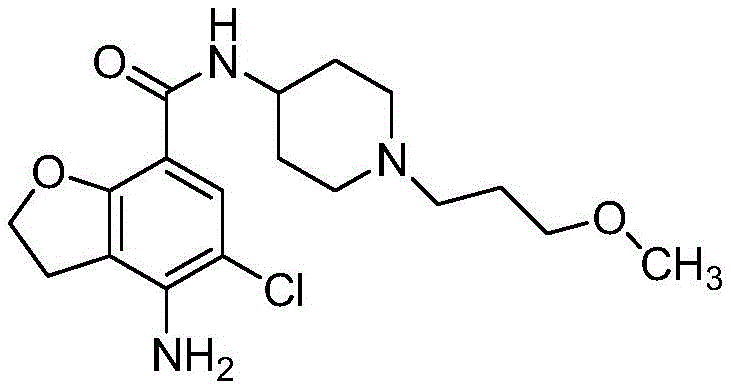
The pharmaceutical industry constantly seeks robust methodologies to ensure the highest quality of active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is detailed in Chinese patent CN108727351B, which discloses a novel refining method for Prucalopride. This compound, chemically known as 4-amino-5-chloro-2,3-dihydro-N-[1-(3-methoxypropyl)-4-piperidyl]-7-benzofuran carboxamide, serves as a critical intermediate for treating gastrointestinal motility disorders. The patent addresses a persistent challenge in the manufacturing workflow: the difficulty of recrystallizing and refining Prucalopride after it has been converted into its succinate salt form. By shifting the purification focus to the free base stage using a specific mixed solvent system, this technology offers a pathway to consistently achieve purity levels exceeding 99.8 percent.
For R&D directors and process chemists, the implications of this refinement strategy are profound. Traditional methods often struggle with impurity profiles that become locked in once the salt is formed, necessitating complex and yield-reducing reprocessing steps. The disclosed method utilizes a synergistic mixture of absolute ethyl alcohol and tetrahydrofuran, combined with activated carbon treatment, to effectively adsorb and remove colored impurities and trace by-products. This approach not only simplifies the operational workflow but also ensures that the final succinate salt, formed in subsequent steps, meets the stringent regulatory standards required for clinical applications. The ability to secure such high purity through a relatively mild and controllable crystallization process marks a substantial advancement in the production of this valuable pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for Prucalopride and its precursors have been plagued by inefficiencies that complicate large-scale manufacturing. As documented in prior art such as CN1071332C and related literature, the synthesis of the key intermediate, 1-(3-methoxypropyl)-4-aminopiperidine (Compound C), often involves costly reagents or harsh conditions. For instance, some established routes rely on the use of lithium aluminum hydride for reduction steps, which mandates strictly anhydrous and oxygen-free environments. These requirements significantly increase operational complexity and safety risks in an industrial setting. Furthermore, alternative reduction methods using ammonium formate have been reported to suffer from incomplete reactions, leading to difficult-to-remove intermediates and by-products due to the high polarity of the target compound.
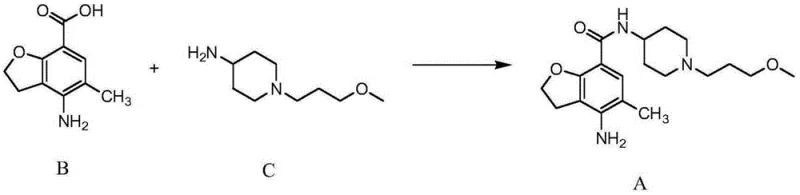
Beyond the synthesis of intermediates, the purification of the final Prucalopride molecule itself has presented a bottleneck. The prevailing conventional wisdom involved forming the succinate salt first and attempting purification thereafter. However, it was observed that Prucalopride succinate is notoriously difficult to recrystallize effectively. This limitation often results in products with suboptimal purity or requires multiple, yield-eroding recrystallization cycles. The reliance on expensive protecting groups in precursor synthesis, such as Boc or Cbz groups which require additional deprotection steps, further inflates the cost of goods and extends the production timeline. These cumulative inefficiencies create a supply chain vulnerability, where yield losses and complex purification needs can lead to inconsistent availability and higher costs for downstream drug manufacturers.
The Novel Approach
The methodology introduced in patent CN108727351B fundamentally rethinks the purification sequence by targeting the free base of Prucalopride prior to salt formation. This strategic shift allows for the exploitation of specific solubility characteristics that are not accessible once the molecule is ionized as a succinate salt. The core of this innovation lies in the use of a mixed solvent system comprising absolute ethanol and tetrahydrofuran. By dissolving the crude Prucalopride in this specific mixture at elevated temperatures (40-45 degrees Celsius) and subsequently introducing purified water as an anti-solvent, the process induces a controlled crystallization. This technique effectively separates the desired product from impurities that remain in the mother liquor.
Moreover, the integration of activated carbon adsorption during the dissolution phase provides a powerful mechanism for decolorization and the removal of organic impurities. This step is crucial for meeting the visual and chemical purity standards required for pharmaceutical grades. The process conditions are remarkably mild, operating between 30 and 40 degrees Celsius during the anti-solvent addition and cooling to just 10-16 degrees Celsius for crystallization. This contrasts sharply with the cryogenic or high-temperature conditions often needed in older synthetic routes. The result is a streamlined operation that delivers a white-like solid with yields consistently above 90 percent and HPLC purity surpassing 99.8 percent, effectively overcoming the historical defects associated with post-salt purification.
Mechanistic Insights into Solvent-Mediated Crystallization
The success of this refining method is rooted in the precise thermodynamic and kinetic control of the crystallization process. The selection of ethanol and tetrahydrofuran (THF) is not arbitrary; these solvents exhibit a unique interaction with the Prucalopride molecule. Ethanol, being a polar protic solvent, and THF, a polar aprotic solvent, create a medium where the free base of Prucalopride has high solubility at elevated temperatures but significantly reduced solubility upon the addition of water and cooling. The preferred volume ratio of ethanol to THF is approximately 3:1, which optimizes the solvation shell around the molecule. When purified water is added dropwise at 30-40 degrees Celsius, it acts as an anti-solvent, disrupting the solvation equilibrium and driving the system towards supersaturation. This controlled supersaturation is critical for promoting the growth of pure crystals rather than the precipitation of amorphous solids or oiling out.
Impurity control is further enhanced by the specific temperature profile employed. Dissolving at 40-45 degrees Celsius ensures complete solubilization of the crude material, allowing the activated carbon to access and adsorb trace impurities effectively. The subsequent gradual cooling to 10-16 degrees Celsius over a period of 4 to 6 hours allows for the orderly arrangement of molecules into the crystal lattice, excluding impurities that do not fit the crystal structure. This slow cooling rate prevents the trapping of mother liquor within the crystals, a common source of purity loss. The washing step with purified water post-filtration removes any residual solvent or surface-adhered impurities without redissolving the product, thanks to the low solubility of Prucalopride in cold water. This multi-faceted approach ensures that the final product is not only chemically pure but also possesses the physical characteristics necessary for efficient downstream processing into the succinate salt.
How to Synthesize Prucalopride Efficiently
The implementation of this refining protocol requires careful attention to solvent ratios and temperature gradients to maximize yield and purity. The process begins with the preparation of the mixed solvent system, followed by the dissolution of the crude Prucalopride under heated conditions. Activated carbon is then introduced to the solution to bind with colored impurities and organic contaminants. Following filtration to remove the carbon, purified water is added slowly to induce crystallization. The mixture is then cooled gradually to promote the formation of high-quality crystals. While the general concept is straightforward, the specific parameters regarding solvent volumes, addition rates, and cooling profiles are critical for reproducibility on a commercial scale. For a detailed, step-by-step guide including exact reagent quantities and equipment specifications, please refer to the standardized synthesis instructions provided below.
- Dissolve crude Prucalopride in a mixed solvent of absolute ethanol and tetrahydrofuran (preferably 3: 1 ratio) at 40-45 degrees Celsius.
- Add activated carbon for adsorption and decolorization, then dropwise add purified water at 30-40 degrees Celsius.
- Gradually cool the mixture to 10-16 degrees Celsius, stir for 4-6 hours, filter, wash with water, and vacuum dry to obtain high-purity solid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this refining technology offers tangible benefits that extend beyond mere technical specifications. The primary advantage lies in the drastic simplification of the purification workflow. By eliminating the need for complex recrystallization attempts on the succinate salt, manufacturers can reduce the number of processing steps, thereby lowering labor costs and equipment occupancy time. The use of common solvents like ethanol and THF, which are readily available in the global chemical market, mitigates the risk of supply disruptions associated with specialty or hazardous reagents. This reliability in raw material sourcing translates directly into enhanced supply chain stability for the final API.
- Cost Reduction in Manufacturing: The elimination of expensive reducing agents like lithium aluminum hydride in precursor synthesis, coupled with the high-efficiency refining of the free base, leads to substantial cost savings. The process avoids the need for rigorous anhydrous conditions, which reduces energy consumption and the capital expenditure required for specialized drying equipment. Furthermore, the high recovery yield minimizes raw material waste, ensuring that a greater proportion of the input material is converted into saleable product. This efficiency drives down the overall cost of goods sold, providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: The robustness of the solvent system and the mild operating conditions contribute to a more reliable production schedule. Unlike processes sensitive to minor fluctuations in temperature or moisture, this method tolerates standard industrial variations, reducing the likelihood of batch failures. The ability to consistently produce high-purity material means fewer delays caused by out-of-specification results. For supply chain managers, this predictability is invaluable, allowing for tighter inventory control and more accurate delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous exothermic reactions or toxic by-products. The solvents used are well-understood and can be recovered and recycled using standard distillation techniques, aligning with modern environmental sustainability goals. The reduction in hazardous waste generation simplifies waste disposal compliance and lowers associated environmental fees. This eco-friendly profile not only meets regulatory requirements but also enhances the corporate social responsibility standing of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical implementation and commercial viability of this refining method. They are derived from the specific technical disclosures within the patent documentation and aim to clarify the operational advantages for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: Why is refining Prucalopride before salt formation advantageous?
A: Refining the free base before forming the succinate salt overcomes the defect where the salt form is difficult to recrystallize. This pre-salt purification allows for effective removal of colored impurities and by-products using activated carbon and controlled crystallization.
Q: What represents the optimal solvent system for this purification?
A: The patent identifies a mixed solvent system of absolute ethanol and tetrahydrofuran as optimal, specifically with a volume ratio of approximately 3:1. This combination provides the necessary solubility differential to achieve high recovery yields while maintaining exceptional purity levels.
Q: How does this method impact industrial scalability?
A: The process operates under mild conditions (30-45 degrees Celsius) and utilizes common, commercially available solvents. Unlike methods requiring anhydrous environments or expensive reducing agents, this crystallization technique is straightforward to scale up for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prucalopride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the supply of pharmaceutical intermediates like Prucalopride. Our technical team has extensively analyzed the refining protocols described in CN108727351B and possesses the expertise to implement them at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to verify every batch against the highest industry standards, guaranteeing that the Prucalopride we deliver is ready for immediate salt formation and final drug formulation.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production needs. By leveraging our optimized refining processes, we can help you reduce lead times and lower overall manufacturing costs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us partner with you to secure a reliable, high-quality supply chain for your Prucalopride requirements, driving efficiency and excellence in your pharmaceutical development projects.
