Advanced Synthesis of 5-Bromo-2-Chlorobenzoic Acid for SGLT-2 Inhibitor Manufacturing
Introduction to High-Selectivity Synthesis Technology
The global demand for SGLT-2 inhibitors, such as dapagliflozin and empagliflozin, has surged due to their efficacy in treating type 2 diabetes without the side effects associated with insulin-dependent therapies. Central to the supply chain of these life-saving medications is the key intermediate, 5-bromo-2-chlorobenzoic acid. A groundbreaking patent, CN112979448A, filed in June 2021, introduces a transformative preparation method that addresses long-standing challenges in yield, purity, and environmental impact. This novel technology utilizes dibromo-amino silica gel as a specialized bromination reagent coupled with ferric trifluoromethanesulfonate as a catalyst. By shifting away from corrosive liquid acids and non-selective brominating agents, this process achieves a product yield of greater than or equal to 95% and a purity exceeding 99.8%. For pharmaceutical manufacturers, this represents a critical advancement in securing a reliable supply of high-purity pharmaceutical intermediates while adhering to increasingly stringent green chemistry standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-bromo-2-chlorobenzoic acid has been plagued by significant technical and environmental drawbacks. Traditional routes often rely on concentrated sulfuric acid as both a solvent and catalyst, paired with N-bromosuccinimide (NBS) as the brominating source. As illustrated in prior art, this approach suffers from poor regioselectivity, leading to high levels of the unwanted 3-bromo-2-chlorobenzoic acid byproduct, which complicates downstream purification and reduces overall efficiency. Furthermore, alternative pathways involving the hydrolysis of expensive trifluoromethylbenzene derivatives or the use of elemental bromine and hydrobromic acid introduce severe safety hazards and equipment corrosion issues. These legacy methods generate substantial quantities of waste acid and hazardous byproducts, creating a heavy burden on wastewater treatment facilities and increasing the total cost of ownership for production facilities.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112979448A offers a streamlined and highly efficient alternative. The core innovation lies in the use of a solid-supported brominating agent, dibromo-amino silica gel, which facilitates a controlled transfer of bromine atoms to the aromatic ring. This system operates in a halogenated hydrocarbon solvent, such as 1-chlorobutane, at moderate temperatures between 60°C and 70°C. The reaction profile is exceptionally clean, avoiding the generation of free strong acids that typically degrade equipment and complicate workup procedures. By employing this heterogeneous catalytic system, manufacturers can achieve a dramatic improvement in process safety and operational simplicity. The ability to filter off the solid support directly after the reaction simplifies the isolation of the target molecule, marking a significant departure from the complex extraction and neutralization steps required by conventional liquid-phase bromination techniques.
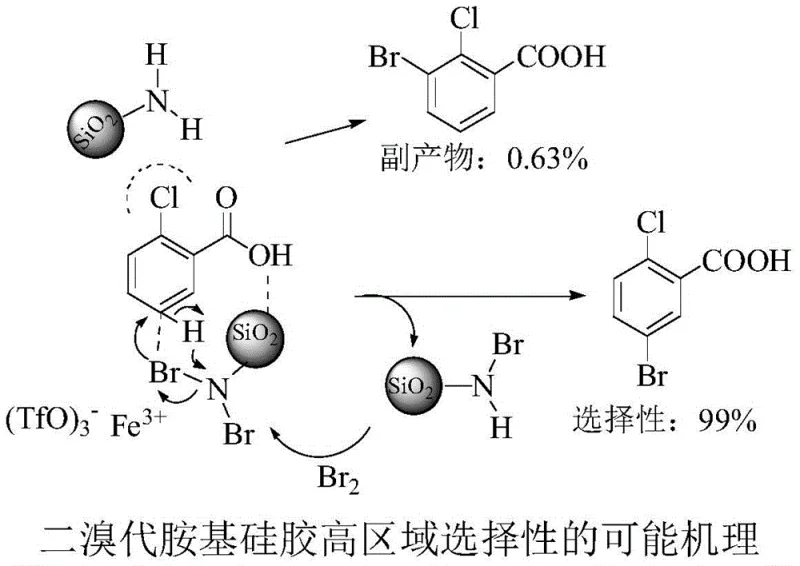
Mechanistic Insights into Fe(OTf)3-Catalyzed Regioselective Bromination
The exceptional performance of this synthesis route is rooted in a sophisticated mechanistic interaction between the catalyst, the substrate, and the solid support. The ferric trifluoromethanesulfonate catalyst plays a pivotal role not only in activating the brominating species but also in enhancing the solubility of the iron species within the organic solvent system. More critically, the dibromo-amino silica gel acts as more than just a bromine carrier; it participates in a directed molecular recognition process. The amino groups on the silica surface form hydrogen bonds with the carboxyl group of the 2-chlorobenzoic acid substrate. This specific interaction orientates the molecule such that the bromine atom is delivered preferentially to the C5 position, which is para to the carboxyl group and meta to the chlorine atom. This spatial arrangement effectively blocks substitution at the C3 position due to steric hindrance from the adjacent chlorine atom and the geometry of the hydrogen-bonded complex.
This mechanism ensures that the formation of the 3-bromo isomer is suppressed to negligible levels, typically below 0.5%, resulting in a regioselectivity of 99%. Such precision is vital for pharmaceutical applications where impurity profiles are strictly regulated. The solid support also allows for the easy separation of the spent reagent, which can be regenerated. The spent monobromo-amino silica gel byproduct can be reactivated with elemental bromine to restore its dibromo functionality. This closed-loop capability means that the reagent does not become a permanent waste stream but rather a reusable asset in the production cycle. The combination of Lewis acid catalysis and supramolecular directing effects exemplifies how modern organic synthesis can achieve both high efficiency and high selectivity through intelligent reagent design.
How to Synthesize 5-Bromo-2-Chlorobenzoic Acid Efficiently
Implementing this advanced synthesis route requires careful attention to the preparation of the specialized reagent and the optimization of reaction parameters. The process begins with the in-situ or ex-situ preparation of the dibromo-amino silica gel, followed by the catalytic bromination step under controlled thermal conditions. The workflow is designed to maximize the contact between the solid reagent and the dissolved substrate while maintaining the stability of the catalyst. Detailed operational protocols regarding stoichiometry, temperature ramps, and filtration techniques are essential for reproducing the high yields and purity reported in the patent literature. Manufacturers aiming to adopt this technology should focus on the precise control of the molar ratios between the substrate, the solid brominating agent, and the iron catalyst to ensure optimal conversion rates.
- Prepare the brominating reagent by reacting amino silica gel with bromine and potassium carbonate in dichloromethane under reflux.
- React 2-chlorobenzoic acid with the prepared dibromo-amino silica gel and ferric trifluoromethanesulfonate catalyst in a halogenated hydrocarbon solvent at 60-70°C.
- Concentrate the solvent, dissolve the product in hot toluene, filter off the silica gel support, and crystallize the pure product upon cooling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method offers compelling strategic advantages beyond mere technical superiority. The shift towards a solid-supported reagent system fundamentally alters the cost structure and risk profile of manufacturing this critical intermediate. By eliminating the need for large volumes of concentrated sulfuric acid and corrosive hydrobromic acid, the process significantly reduces the requirement for specialized corrosion-resistant equipment, thereby lowering capital expenditure and maintenance costs. Furthermore, the simplified workup procedure, which involves hot filtration and crystallization rather than complex aqueous extractions, reduces solvent consumption and energy usage. These operational efficiencies translate directly into a more robust and cost-effective supply chain, ensuring that production timelines are met without the delays often associated with hazardous waste disposal and extensive purification processes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the recyclability of the dibromo-amino silica gel. Unlike traditional reagents that are consumed entirely and generate stoichiometric amounts of waste, this solid support can be recovered and reactivated multiple times with a recovery rate reaching 98% after six cycles. This drastic reduction in reagent consumption lowers the variable cost per kilogram of the final product. Additionally, the high selectivity of the reaction minimizes the loss of raw materials to byproducts, ensuring that the expensive 2-chlorobenzoic acid starting material is converted efficiently into the desired API intermediate. The elimination of expensive heavy metal scavengers or complex chromatographic purification steps further contributes to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of hazardous raw materials and the regulatory constraints surrounding their transport and storage. This new method utilizes relatively benign halogenated hydrocarbon solvents and a stable solid reagent that can be stockpiled safely. The reduced reliance on highly corrosive acids mitigates the risk of production stoppages due to equipment failure or safety incidents. Moreover, the simplicity of the process makes it easier to scale up from pilot plant to commercial production without encountering the nonlinear engineering challenges typical of highly exothermic or corrosive liquid-phase reactions. This scalability ensures that suppliers can respond rapidly to fluctuations in market demand for SGLT-2 inhibitor precursors.
- Scalability and Environmental Compliance: In an era of increasing environmental scrutiny, the green chemistry attributes of this process provide a significant competitive edge. The reduction in three-waste emissions—specifically the avoidance of large volumes of acidic wastewater—simplifies compliance with environmental regulations and reduces the burden on effluent treatment plants. The solid waste generated, primarily the silica support, is non-hazardous and can be managed more easily than toxic sludge. This alignment with sustainable manufacturing principles not only reduces operational risks related to environmental permits but also enhances the corporate social responsibility profile of the supply chain. Companies adopting this technology can market their products as sustainably sourced, appealing to downstream pharmaceutical partners who are increasingly prioritizing green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-selectivity bromination technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent documentation. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for quality assurance teams establishing specification limits. The answers provided reflect the proven capabilities of the method as demonstrated in the patent examples, offering a realistic view of what can be achieved in a GMP-compliant manufacturing environment.
Q: What is the regioselectivity of this new bromination method?
A: The method achieves exceptional regioselectivity of 99% for the target 5-bromo-2-chlorobenzoic acid, significantly minimizing the formation of the 3-bromo isomer byproduct.
Q: Can the dibromo-amino silica gel reagent be recycled?
A: Yes, the solid-supported reagent can be recovered by filtration and reactivated with bromine, achieving a recovery rate of 98% even after six recycling cycles.
Q: How does this process compare to traditional NBS bromination?
A: Unlike traditional methods using concentrated sulfuric acid and NBS which generate large amounts of waste acid and have poor selectivity, this novel route is environmentally friendly, operates under milder conditions, and yields product purity greater than 99.8%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromo-2-Chlorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation diabetes treatments depends on the availability of high-quality intermediates produced via robust and scalable processes. Our technical team has thoroughly analyzed the potential of the dibromo-amino silica gel bromination route and is fully equipped to implement this advanced chemistry at an industrial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material regardless of market volume. Our facilities are supported by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 5-bromo-2-chlorobenzoic acid meets the exacting standards required for the synthesis of SGLT-2 inhibitors.
We invite pharmaceutical partners to collaborate with us to leverage this innovative synthesis technology for their supply chains. By working together, we can optimize the production parameters to achieve maximum efficiency and cost-effectiveness tailored to your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis and to obtain specific COA data and route feasibility assessments. Let us help you secure a sustainable and high-performance supply of this critical pharmaceutical building block.
