Advanced Synthesis of 5-Bromo-2-Chlorobenzoic Acid for Commercial Scale-up
The pharmaceutical industry is currently witnessing a surge in demand for SGLT2 inhibitors, such as dapagliflozin and eglliflozin, which rely heavily on high-quality intermediates for their synthesis. A pivotal breakthrough in this domain is documented in patent CN108250060B, which outlines a novel and highly efficient synthesis method for 5-bromo-2-chlorobenzoic acid. This compound serves as a critical building block not only for antidiabetic medications but also for various antiviral agents and renin inhibitors. The traditional reliance on expensive and environmentally hazardous starting materials has long been a bottleneck for manufacturers seeking to optimize their supply chains. By shifting the synthetic paradigm to utilize salicylic acid, a commodity chemical with extensive global availability, this new methodology addresses both economic and ecological concerns simultaneously. The strategic implementation of an oxidative bromination system followed by a specialized catalytic chlorination step ensures that the final product meets the rigorous purity standards required for active pharmaceutical ingredient (API) production. For R&D directors and procurement specialists alike, understanding the nuances of this patented route is essential for securing a competitive edge in the manufacturing of next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 5-bromo-2-chlorobenzoic acid has been plagued by significant technical and economic drawbacks inherent to older synthetic routes. One prevalent method involves the hydrolysis of 5-bromo-2-chlorotrifluoromethane under acidic conditions, a process that necessitates the use of extremely expensive raw materials that are difficult to source reliably on a global scale. Furthermore, this route generates substantial quantities of fluorine-containing wastewater, posing severe environmental pollution challenges that require costly treatment protocols before disposal. Another common approach utilizes 2-chlorobenzoic acid as a starting material for direct bromination; however, this reaction suffers from poor selectivity, often yielding a mixture of 5-bromo and 3-bromo isomers in ratios as unfavorable as 4:1. Even with modifications using N-bromosuccinimide systems, the yield remains suboptimal, and the starting material itself lacks the widespread availability of simpler aromatic acids. A third route involving 2-chloro-trichlorotoluene introduces additional complexities, including the generation of large amounts of waste acid and high-salt wastewater during hydrolysis, which complicates post-treatment and drives up operational expenditures. These cumulative inefficiencies render conventional methods unsuitable for modern, sustainable industrial mass production.
The Novel Approach
In stark contrast to these legacy processes, the innovative method described in the patent leverages salicylic acid as a foundational raw material, offering a pathway that is both economically viable and environmentally responsible. This two-step synthesis begins with a highly selective bromination reaction to form 2-hydroxy-5-bromo-benzoic acid, followed by a precise chlorination step to yield the target molecule. The adoption of salicylic acid eliminates the dependency on scarce fluorinated compounds or specialized chlorinated toluenes, thereby stabilizing the supply chain against raw material volatility. The process is designed to maximize atom economy and minimize waste generation, directly addressing the environmental compliance issues associated with previous routes. By carefully selecting reagents and catalysts, the new method achieves reaction yields exceeding 90 percent for both the bromination and chlorination stages, a significant improvement over the 40 to 80 percent yields typical of older techniques. This enhancement in efficiency translates directly into reduced production costs and a smaller environmental footprint, making it an ideal candidate for companies aiming to scale up production of high-purity pharmaceutical intermediates without compromising on sustainability goals.
Mechanistic Insights into Oxidative Bromination and Catalytic Chlorination
The core of this synthetic advancement lies in the sophisticated control of reaction mechanisms, particularly during the initial bromination phase where regioselectivity is paramount. The patent specifies the use of an oxidative bromination system comprising tetrabutylammonium bromide, oxygen, and sodium metavanadate, with aluminum tribromide serving as the catalyst in a 1,4-dioxane solvent. This specific combination is engineered to direct the bromine atom exclusively to the 5-position of the salicylic acid ring, achieving an impressive isomer ratio of approximately 24:1 in favor of the desired 5-bromo product over the 3-bromo impurity. The presence of oxygen and the vanadium catalyst facilitates the regeneration of the active brominating species in situ, allowing for a continuous and controlled reaction environment that minimizes side reactions. Following the isolation of the intermediate, the subsequent chlorination step employs carbon tetrachloride as the chlorinating agent alongside a molybdenum hexacarbonyl catalyst. This choice of catalyst is critical, as it effectively promotes the substitution of the hydroxyl group with chlorine while suppressing unwanted decarboxylation or phenol coupling byproducts that often plague similar transformations. The synergy between these carefully selected catalytic systems ensures that the molecular integrity of the benzoic acid backbone is maintained throughout the synthesis.
Impurity control is another vital aspect of this mechanism, ensuring that the final product meets the stringent specifications required for API intermediates. The high selectivity of the oxidative bromination system inherently reduces the formation of structural isomers, which are notoriously difficult to separate via standard crystallization techniques. In the chlorination stage, the use of molybdenum hexacarbonyl specifically mitigates the risk of forming aryl bromide byproducts or ether linkages, which could otherwise compromise the purity profile of the batch. Post-reaction processing further refines the quality, utilizing thermal dissolution in acetonitrile followed by filtration through neutral alumina to remove residual catalysts and colored impurities. The final recrystallization from an ethanol and water mixture ensures that the product achieves a purity level greater than 99.5 percent, as confirmed by HPLC analysis. This rigorous attention to mechanistic detail and purification protocol provides R&D teams with confidence in the reproducibility and robustness of the process, facilitating smoother technology transfer from the laboratory to commercial manufacturing facilities.
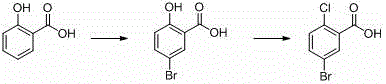
How to Synthesize 5-Bromo-2-Chlorobenzoic Acid Efficiently
Implementing this synthesis route requires precise adherence to the reaction conditions outlined in the patent to ensure optimal yield and purity. The process begins with the preparation of the oxidative bromination mixture, where salicylic acid is combined with the catalyst and solvent system under controlled temperature conditions to facilitate the selective introduction of the bromine atom. Once the intermediate 2-hydroxy-5-bromo-benzoic acid is isolated and purified, it serves as the substrate for the subsequent chlorination reaction, which demands higher temperatures and specific catalytic loading to drive the substitution to completion. Detailed standardized synthetic steps see the guide below.
- Perform oxidative bromination of salicylic acid using tetrabutylammonium bromide and oxygen with aluminum tribromide catalyst.
- Isolate 2-hydroxy-5-bromo-benzoic acid through recrystallization using ethanol and tetrahydrofuran.
- Conduct chlorination using carbon tetrachloride and molybdenum hexacarbonyl catalyst to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this novel synthesis method offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the fundamental shift to salicylic acid as a starting material, which is a commodity chemical produced in massive volumes globally, ensuring a stable and resilient supply chain不受 geopolitical disruptions that often affect specialized fluorinated reagents. This abundance translates into significant cost reduction in pharmaceutical intermediates manufacturing, as the raw material input costs are drastically lower compared to the expensive precursors required by conventional routes. Furthermore, the elimination of fluorine-containing wastewater and high-salt effluents simplifies the environmental compliance landscape, reducing the overhead costs associated with waste treatment and regulatory reporting. The high yield and selectivity of the process mean that less raw material is wasted per unit of product, enhancing overall resource efficiency and contributing to a more sustainable production model. These factors collectively create a compelling business case for adopting this technology, offering a pathway to lower cost of goods sold (COGS) while maintaining high quality standards.
- Cost Reduction in Manufacturing: The utilization of salicylic acid, a widely available and inexpensive feedstock, fundamentally alters the cost structure of producing 5-bromo-2-chlorobenzoic acid. By avoiding the need for costly fluorinated starting materials or complex chlorinated toluenes, manufacturers can achieve substantial cost savings on raw material procurement. Additionally, the high reaction yields exceeding 90 percent minimize material loss, ensuring that a greater proportion of input chemicals are converted into valuable product. The simplified post-treatment process, which avoids complex waste acid neutralization steps, further reduces operational expenditures related to utilities and waste disposal. These cumulative efficiencies allow for a more competitive pricing strategy in the global market for API intermediates.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like salicylic acid significantly de-risks the supply chain compared to sourcing niche fluorinated compounds that may have limited suppliers. The robustness of the two-step process, which utilizes standard solvents and catalysts, ensures that production can be scaled up rapidly to meet fluctuating market demands without encountering bottlenecks. The high purity of the intermediate reduces the need for extensive reprocessing or rejection of batches, leading to more predictable lead times and inventory management. This reliability is crucial for downstream API manufacturers who require consistent quality and timely delivery to maintain their own production schedules for finished dosage forms.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, avoiding the use of hazardous reagents that pose safety risks in large reactors. The absence of fluorine-containing wastewater eliminates a major environmental liability, making it easier to obtain and maintain operating permits in regions with strict environmental regulations. The use of recyclable solvents and the generation of minimal solid waste contribute to a greener manufacturing profile, aligning with corporate sustainability goals. This environmental compatibility not only reduces compliance costs but also enhances the brand reputation of the manufacturer as a responsible supplier in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial implications of this synthesis method, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance. Understanding these details is key to making informed decisions about integrating this technology into existing production workflows.
Q: What are the advantages of using salicylic acid as a starting material?
A: Salicylic acid is widely available and low-cost compared to fluorinated alternatives, significantly reducing raw material expenses and supply chain risks.
Q: How does this method improve product purity?
A: The oxidative bromination system achieves high regioselectivity (24:1 ratio), minimizing isomer impurities, while the specific chlorination catalyst prevents decarboxylation byproducts.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the method avoids hazardous fluorine-containing wastewater and uses standard solvents, making it environmentally compliant and scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromo-2-Chlorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications like dapagliflozin. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 5-bromo-2-chlorobenzoic acid meets the highest industry standards. We are committed to leveraging advanced synthetic technologies, such as the one described in CN108250060B, to deliver superior value to our partners through enhanced efficiency and reliability.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs for your key therapeutic programs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and bringing your innovations to market faster.
