Advanced Synthesis of 5-Bromo-2-Chlorobenzoic Acid for High-Purity Diabetes Drug Manufacturing
Introduction to Next-Generation SGLT2 Inhibitor Intermediates
The pharmaceutical landscape for type 2 diabetes treatment has been revolutionized by Sodium-Glucose Cotransporter 2 (SGLT2) inhibitors, creating an unprecedented demand for high-quality precursors like 5-bromo-2-chlorobenzoic acid. Patent CN113801010A introduces a groundbreaking preparation method that addresses the critical bottlenecks of legacy synthesis routes. This technology shifts the paradigm from hazardous, low-yield toluene oxidations to a sophisticated, multi-step sequence starting from 2-nitrobenzoic acid. By leveraging controlled esterification, catalytic hydrogenation, and precise halogenation, this novel approach ensures exceptional product purity exceeding 99% while maintaining mild reaction conditions. For global pharmaceutical manufacturers, adopting this methodology translates directly into enhanced supply chain reliability and reduced environmental impact, positioning it as a cornerstone technology for the next generation of hypoglycemic drug production.
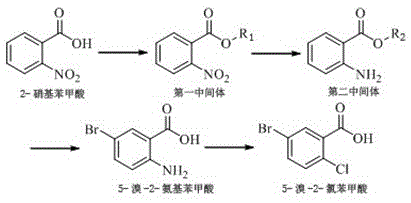
The Limitations of Conventional Methods vs. The Novel Approach
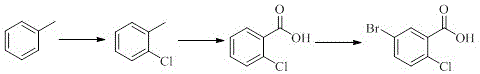
The Limitations of Conventional Methods
Historically, the industrial synthesis of 5-bromo-2-chlorobenzoic acid has relied heavily on the chlorination and oxidation of toluene derivatives, a pathway fraught with significant technical and economic inefficiencies. As illustrated in the traditional reaction schemes, the initial chlorination of toluene often leads to the formation of persistent dichlorinated impurities that are notoriously difficult to separate from the desired mono-chlorinated product. Furthermore, the subsequent oxidation of the methyl group using potassium permanganate is a major bottleneck, typically resulting in dismal yields of approximately 55%. An alternative legacy route involving ultraviolet irradiation and phosphorus pentachloride suffers from poor light penetration in solid-liquid phases, extending reaction times and capping yields at a mere 40%. These inefficiencies not only inflate production costs but also generate substantial chemical waste, posing severe challenges for environmental compliance and sustainable manufacturing practices.
The Novel Approach
In stark contrast to these archaic methods, the patented process utilizing 2-nitrobenzoic acid as the starting material offers a streamlined and highly efficient alternative that circumvents the pitfalls of radical chlorination and harsh oxidation. This innovative route begins with a high-yield esterification step, followed by a clean catalytic reduction that avoids the use of stoichiometric metal reductants which generate heavy metal waste. The strategic sequencing of bromination prior to the final chlorination via diazotization allows for precise regioselectivity, ensuring that the bromine atom is installed at the 5-position with high fidelity. By eliminating the need for aggressive oxidants like potassium permanganate and avoiding the complex purification of dichlorinated byproducts, this method drastically simplifies the downstream processing workflow. The result is a robust synthesis capable of delivering yields exceeding 85% with purity levels that meet the stringent requirements of modern API manufacturing.
Mechanistic Insights into the Four-Step Cascade Synthesis
The core of this technological advancement lies in its meticulously engineered four-step reaction cascade, which prioritizes atom economy and impurity control at every stage. The process initiates with the esterification of 2-nitrobenzoic acid, where the carboxylic acid group is protected as an alkyl ester, facilitating subsequent solubility and reactivity modifications. This is followed by a critical reduction step, preferably utilizing catalytic hydrogenation with palladium on carbon, which converts the nitro group to an amine with exceptional efficiency and minimal byproduct formation. The third stage involves a carefully controlled bromination of the amino-ester intermediate, where the amino group directs the electrophilic substitution to the para-position relative to itself. Crucially, the patent specifies the addition of a second reducing agent, such as sodium sulfite, immediately after halogenation to quench excess bromine, thereby preventing over-halogenation and ensuring the integrity of the aromatic ring before the final hydrolysis restores the carboxylic acid functionality.
The final transformation involves a classic Sandmeyer-type reaction sequence, where the 5-bromo-2-aminobenzoic acid undergoes diazotization followed by chlorination to install the final chlorine atom at the 2-position. This step is executed under mild acidic conditions using sodium nitrite and a copper chloride catalyst, avoiding the extreme temperatures and pressures associated with older methodologies. The mechanistic precision of this route ensures that the halogen atoms are introduced in a specific order—bromine first, then chlorine—which is thermodynamically and kinetically favorable for achieving the desired substitution pattern. Furthermore, the use of aqueous workups and pH adjustments throughout the process allows for the effective removal of inorganic salts and organic impurities, resulting in a final product that requires minimal recrystallization. This level of control over the reaction mechanism is what enables the consistent production of high-purity intermediates essential for the synthesis of complex SGLT2 inhibitors.
How to Synthesize 5-Bromo-2-Chlorobenzoic Acid Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters outlined in the patent to maximize yield and safety. The process is designed to be operationally simple, utilizing common solvents like dichloromethane, methanol, and ethyl acetate, which are readily available in most pharmaceutical manufacturing facilities. The detailed standard operating procedures involve precise temperature controls, particularly during the exothermic bromination and diazotization steps, to prevent thermal runaway and ensure operator safety. For a comprehensive breakdown of the specific molar ratios, reaction times, and workup protocols required to replicate these results, please refer to the standardized synthesis guide below.
- Perform esterification of 2-nitrobenzoic acid with alcohol to form the first intermediate ester.
- Conduct catalytic hydrogenation reduction on the ester to obtain the amino-ester intermediate.
- Execute bromination followed by hydrolysis to generate 5-bromo-2-aminobenzoic acid.
- Finalize with diazotization and chlorination to yield the target 5-bromo-2-chlorobenzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative advantages that extend far beyond simple chemical yield improvements. By shifting away from the volatile and inefficient toluene oxidation pathways, manufacturers can secure a more stable and predictable supply of 5-bromo-2-chlorobenzoic acid, mitigating the risks associated with batch failures and inconsistent quality. The elimination of hazardous reagents like phosphorus pentachloride and the reduction of heavy metal waste streams significantly lower the environmental compliance burden, translating into reduced operational overheads and faster regulatory approvals. Moreover, the use of inexpensive and widely available starting materials like 2-nitrobenzoic acid insulates the supply chain from the price volatility often seen with specialized petrochemical derivatives, ensuring long-term cost stability for high-volume production campaigns.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the dramatic improvement in overall yield and the simplification of purification steps. Unlike the legacy methods which suffer from yields as low as 40-55%, this new route consistently achieves yields above 85%, effectively nearly doubling the output per unit of raw material input. The avoidance of expensive oxidants like potassium permanganate and the ability to recover and reuse solvents further contribute to substantial cost savings. Additionally, the high purity of the crude product reduces the need for extensive and costly recrystallization or chromatography processes, streamlining the manufacturing timeline and reducing labor and utility costs associated with prolonged processing.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on commodity chemicals such as alcohols, sodium nitrite, and common mineral acids ensures that production is not held hostage by the availability of niche reagents. The robustness of the catalytic hydrogenation step, which uses standard palladium on carbon catalysts, means that the process can be easily scaled up in existing facilities without requiring specialized equipment for high-pressure or high-temperature reactions. This flexibility allows suppliers to respond rapidly to fluctuations in market demand for SGLT2 inhibitors, ensuring that downstream API manufacturers receive their intermediates on schedule without interruption. The simplified workflow also reduces the likelihood of production delays caused by complex troubleshooting or equipment maintenance associated with more hazardous legacy processes.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior, aligning with the increasingly stringent global regulations regarding chemical manufacturing emissions. By eliminating the generation of dichlorinated impurities and avoiding the use of toxic heavy metal oxidants, the process significantly reduces the volume of hazardous waste requiring disposal. The mild reaction conditions, typically ranging from 0 to 90 degrees Celsius, lower the energy consumption required for heating and cooling, contributing to a smaller carbon footprint. This green chemistry approach not only facilitates easier permitting for new production lines but also enhances the corporate sustainability credentials of the manufacturer, a factor that is becoming increasingly important for partnerships with major multinational pharmaceutical companies committed to ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms traditional alternatives. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the nitrobenzoic acid route superior to the traditional toluene oxidation method?
A: The traditional toluene route suffers from low yields (around 55%) during the potassium permanganate oxidation step and generates difficult-to-remove dichlorinated impurities. The new nitrobenzoic acid route achieves yields over 85% with purity exceeding 99%, avoiding harsh oxidation conditions.
Q: How does this process ensure high purity for SGLT2 inhibitor synthesis?
A: The process utilizes specific quenching agents like sodium sulfite to remove excess halogenating reagents effectively. Additionally, the stepwise purification through esterification and hydrolysis allows for the removal of organic impurities before the final critical diazotization step.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states the method uses cheap, easily obtained raw materials and solvents with mild reaction conditions (0-90°C). It avoids explosive or highly toxic intermediates, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromo-2-Chlorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior synthetic routes requires a partner with deep technical expertise and proven scalability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN113801010A are fully realized in a practical industrial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 5-bromo-2-chlorobenzoic acid meets the exacting standards required for SGLT2 inhibitor synthesis. Our commitment to quality assurance means that we can consistently deliver intermediates with impurity profiles that facilitate seamless downstream processing for our clients.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and production timelines. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance the efficiency and profitability of your diabetes drug manufacturing operations. Let us be your strategic partner in navigating the complexities of modern pharmaceutical intermediate supply.
