Scalable Synthesis of Indoline Derivatives via Palladium-Catalyzed C-H Activation for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access nitrogen-containing heterocycles, which serve as critical scaffolds for bioactive molecules. Patent CN109721523B introduces a transformative approach to synthesizing indoline derivatives, a structural motif found in numerous therapeutic agents such as physostigmine and clemastine. This technology leverages an oxamide-induced intramolecular C-N bond forming reaction, utilizing a palladium-catalyzed C-H activation strategy that significantly outperforms legacy methods in terms of atom economy and operational simplicity. By employing a readily available oxamide directing group, the process achieves high regioselectivity under relatively mild oxidative conditions. For procurement teams and R&D directors alike, this represents a pivotal shift towards more sustainable and cost-effective pharmaceutical intermediates manufacturing, addressing the perennial challenges of catalyst loading and oxidant expense.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of indoline skeletons via C-H activation has relied heavily on expensive and sometimes hazardous reagents that hinder commercial scalability. Early pioneering work by the Yu group utilized strong oxidizing agents like cerium ammonium nitrate or stoichiometric amounts of hypervalent iodine reagents such as PhI(OAc)2, which are not only costly but also generate significant amounts of iodine-containing waste. Furthermore, these traditional protocols often required high catalyst loadings, typically ranging from 10 to 15 mol% of palladium, which creates a substantial economic burden when scaling to metric ton quantities. The reliance on such expensive oxidants and high metal loading results in complex downstream processing, necessitating rigorous purification steps to remove heavy metal residues to meet stringent stringent purity specifications required for API production. Additionally, some earlier methods suffered from limited substrate scope, particularly struggling with electron-deficient or sterically hindered phenethylamine derivatives.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a highly optimized catalytic system that replaces expensive hypervalent iodine oxidants with cheap and commercially abundant organic peroxides, specifically tert-butyl peroxybenzoate (TBPB). This switch alone represents a massive leap forward in cost reduction in pharmaceutical intermediates manufacturing, as TBPB is a bulk chemical commodity compared to the niche reagent PhI(OAc)2. The process operates efficiently with a drastically reduced catalyst loading of merely 2.5 mol% palladium acetate, facilitated by the use of hexafluoroisopropanol (HFIP) as a specialized solvent that enhances the electrophilicity of the palladium species. As illustrated in the reaction scheme below, the transformation proceeds smoothly from the oxamide precursor to the cyclized indoline product with high efficiency.
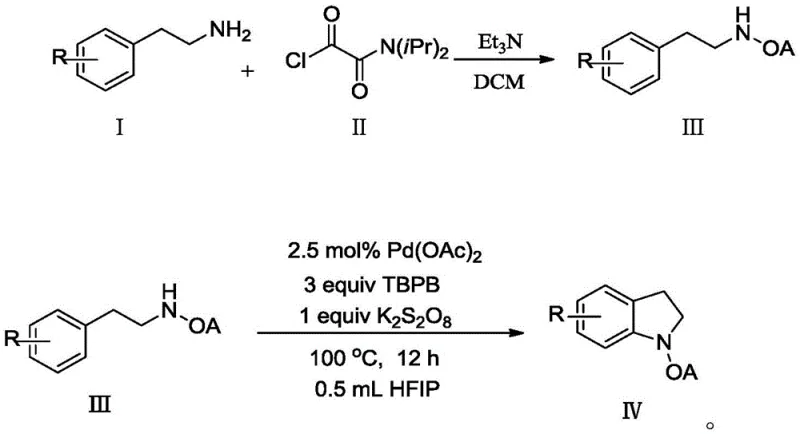
The reaction tolerates a wide array of functional groups, including halogens and alkoxy groups, making it a versatile platform for generating diverse libraries of bioactive compounds. The use of potassium persulfate as an additive further promotes the turnover of the catalytic cycle, ensuring high conversion rates without the need for excessive reagent quantities. This novel approach effectively decouples the synthesis of complex heterocycles from the constraints of expensive reagents, offering a streamlined pathway that is inherently more suitable for industrial application.
Mechanistic Insights into Pd-Catalyzed C-H Activation
The core of this technological advancement lies in the precise mechanism of the oxamide-directed C-H activation. The oxamide moiety serves as a powerful bidentate directing group that coordinates with the palladium(II) center to form a stable five-membered palladacycle intermediate. This coordination brings the metal center into close proximity with the target ortho-C-H bond on the aromatic ring, lowering the activation energy required for the C-H bond cleavage. Once the C-Pd bond is formed, the presence of the peroxide oxidant facilitates the oxidation of the palladium center to a higher valence state, likely Pd(IV), which is crucial for the subsequent reductive elimination step. This reductive elimination forms the new C-N bond, closing the five-membered indoline ring and regenerating the active Pd(II) catalyst. The choice of HFIP as a solvent is mechanistically significant, as its strong hydrogen-bond donating ability stabilizes the anionic intermediates and enhances the electrophilicity of the metal center, thereby accelerating the rate-determining C-H activation step.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based cyclization methods. Because the reaction proceeds through a well-defined organometallic cycle directed by the chelating oxamide group, the formation of random radical coupling by-products is minimized. The high selectivity ensures that the major impurity profile is dominated by unreacted starting material or simple mono-oxidized species, which are far easier to remove via standard crystallization or chromatography than complex polymeric tars often seen in non-directed radical reactions. This clean reaction profile is essential for maintaining the integrity of the supply chain, as it reduces the risk of batch failures due to difficult-to-remove impurities. The robustness of the catalytic cycle allows for consistent performance across different batches, a key requirement for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Indoline Derivatives Efficiently
The practical execution of this synthesis is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment. The process begins with the preparation of the oxamide substrate, which can be synthesized from readily available phenethylamines and oxalyl chloride derivatives. The cyclization step itself requires careful control of temperature and stoichiometry to maximize yield while minimizing side reactions. The patent specifies a preferred temperature of 100°C and a reaction time of 12 hours, conditions that balance reaction rate with thermal stability of the reagents. Detailed standardized operating procedures for this synthesis, including specific workup and purification protocols, are outlined in the technical guide below.
- Prepare the reaction mixture by combining the oxamide derivative substrate with 2.5 mol% palladium acetate catalyst, 3 equivalents of tert-butyl peroxybenzoate oxidant, and 1 equivalent of potassium persulfate additive in hexafluoroisopropanol solvent.
- Seal the reaction vessel and heat the mixture to 100°C in an oil bath, maintaining stirring for approximately 12 hours to ensure complete intramolecular C-N bond formation.
- Upon completion, cool the system to room temperature, filter any precipitated solids, remove the solvent via rotary evaporation, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain managers and procurement officers, the adoption of this synthetic route offers compelling strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for exotic and expensive oxidants like PhI(OAc)2, the process relies on commodity chemicals that are available from multiple global suppliers, thereby mitigating supply risk and enhancing negotiation leverage. This diversification of the supply base ensures enhanced supply chain reliability, protecting production schedules from the volatility often associated with niche fine chemical reagents. Furthermore, the reduced catalyst loading directly translates to lower consumption of precious metals, which not only cuts material costs but also simplifies the recovery and recycling of palladium, adding another layer of economic efficiency.
- Cost Reduction in Manufacturing: The substitution of high-cost hypervalent iodine oxidants with inexpensive organic peroxides results in a significant decrease in the bill of materials for each batch. Since the oxidant is used in stoichiometric quantities, the price differential between TBPB and PhI(OAc)2 accumulates rapidly at scale, leading to substantial overall cost savings. Additionally, the lower palladium loading reduces the capital tied up in metal inventory and decreases the cost associated with metal scavenging processes required to meet regulatory limits for residual metals in pharmaceutical products. These factors combine to create a much more economically viable process for the commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as potassium persulfate and tert-butyl peroxybenzoate ensures that production is not bottlenecked by the lead times of specialized reagents. Unlike custom-synthesized oxidants that may have long procurement cycles, these commodities can be sourced with short notice, allowing for agile response to market demand fluctuations. This availability supports reducing lead time for high-purity pharmaceutical intermediates, enabling manufacturers to maintain leaner inventory levels while ensuring continuous operation. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply chain.
- Scalability and Environmental Compliance: The reaction conditions are mild enough to be safely managed in standard stainless steel reactors without requiring specialized lining or extreme pressure ratings. The use of HFIP, while a specialized solvent, allows for efficient recycling due to its distinct physical properties, aligning with green chemistry principles by reducing solvent waste. The cleaner impurity profile reduces the burden on wastewater treatment facilities and lowers the cost of waste disposal. This environmental compatibility is increasingly critical for maintaining operational licenses and meeting the sustainability goals of modern chemical enterprises, making this route a future-proof choice for long-term manufacturing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxamide-directed cyclization technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for process development teams. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the advantages of using TBPB over PhI(OAc)2 in this synthesis?
A: The use of tert-butyl peroxybenzoate (TBPB) offers significant cost advantages over the traditionally used hypervalent iodine oxidant PhI(OAc)2. TBPB is commercially abundant and substantially cheaper, which drastically reduces the raw material costs for large-scale manufacturing while maintaining high reaction efficiency.
Q: How does the oxamide directing group improve the reaction selectivity?
A: The oxamide group acts as a robust bidentate ligand that coordinates effectively with the palladium center. This coordination directs the metal specifically to the ortho-C-H bond of the phenethylamine backbone, facilitating highly selective intramolecular cyclization and minimizing the formation of unwanted regioisomers or over-oxidized by-products.
Q: Is this process suitable for substrates containing halogen substituents?
A: Yes, the methodology demonstrates excellent functional group tolerance. The patent data confirms successful cyclization for substrates bearing bromine, fluorine, and chlorine substituents on the aromatic ring, yielding the corresponding halogenated indoline derivatives in good to excellent yields without dehalogenation side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has extensively evaluated the oxamide-directed C-H activation methodology and possesses the technical capability to optimize this process for your specific needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of indoline derivatives meets the highest standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this novel synthetic route. We encourage you to contact us to obtain specific COA data for our reference standards and to discuss route feasibility assessments tailored to your target molecules. Let us help you accelerate your development timeline and reduce your manufacturing costs with our proven expertise in heterocyclic chemistry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
