Advanced Electrochemical Synthesis of Desmopressin for Commercial Scale-up and High Purity
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for the production of complex peptide therapeutics. A significant breakthrough in this domain is documented in patent CN112062813B, which outlines a novel synthesis method for Desmopressin, a critical analogue of natural argirelin used in treating central diabetes insipidus and hemophilia. This patent introduces a hybrid approach that combines the robustness of Fmoc solid-phase peptide synthesis with the precision of liquid-phase electrochemical oxidation. By integrating these technologies, the method addresses long-standing challenges regarding reaction selectivity and atom economy. The core innovation lies in the construction of the disulfide bond, a critical structural feature, using an electrochemical cell rather than traditional chemical oxidants. This shift not only enhances the purity profile of the final active pharmaceutical ingredient but also aligns with the growing global demand for green chemistry in manufacturing processes. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for evaluating future supply chain strategies and cost-reduction opportunities in peptide manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Desmopressin has relied heavily on liquid-phase or solid-phase oxidation methods utilizing chemical reagents such as iodine, hydrogen peroxide, or air oxidation. These conventional techniques suffer from significant drawbacks that impact both the economic and technical viability of the process. A primary concern is the lack of specific selectivity during the formation of the disulfide bond. When using chemical oxidants, there is a high propensity for the formation of mismatched impurities, where the cysteine residues form incorrect linkages or undergo over-oxidation to sulfonic acids. This results in a complex impurity profile that is difficult and costly to purify, often requiring multiple chromatography steps that reduce overall yield. Furthermore, the use of iodine introduces environmental hazards, necessitating rigorous waste treatment protocols to remove heavy metal residues from the final product. The atom economy of these traditional sequential coupling methods is also suboptimal, as the low utilization rate of the main chain fragments leads to substantial waste of expensive protected amino acids. These factors collectively drive up the cost of goods sold and extend the lead time for high-purity pharmaceutical intermediates.
The Novel Approach
In contrast, the method disclosed in patent CN112062813B offers a transformative solution by decoupling the backbone synthesis from the cyclization step. The novel approach employs an Fmoc/tBu strategy to assemble the linear peptide chain on a solid support, ensuring high efficiency in the coupling steps. The true innovation occurs in the final stage, where the crude linear peptide is subjected to liquid-phase electrochemical oxidation. This technique utilizes electricity to drive the formation of the disulfide bond between the mercaptopropionic acid and cysteine residues. By controlling the current and potential, the reaction avoids the indiscriminate oxidation associated with chemical reagents. This results in a much cleaner reaction profile with significantly fewer side products. The elimination of iodine and hydrogen peroxide not only simplifies the downstream purification process but also removes the need for expensive heavy metal scavenging steps. Consequently, this approach improves the utilization rate of the fragment peptide resin and the main chain fragments, leading to a more cost-effective and environmentally friendly manufacturing process that is highly attractive for commercial scale-up.
Mechanistic Insights into Electrochemical Disulfide Bond Formation
The mechanistic foundation of this synthesis relies on the precise control of electron transfer to facilitate the oxidative coupling of thiol groups. In the electrochemical cell, the crude linear peptide containing free thiol groups from the cysteine and mercaptopropionic acid residues is dissolved in a suitable electrolyte solution, such as acetonitrile containing tetrabutylammonium tetrafluoroborate. When a current is applied across platinum electrodes, the thiol groups undergo oxidation at the anode to form thiyl radicals. These radicals rapidly dimerize to form the desired disulfide bond, closing the cyclic structure of the Desmopressin molecule. The use of platinum electrodes ensures stability and prevents contamination of the product with electrode materials. The reaction conditions, typically maintained at a mild temperature of 35°C and a controlled current of 10-15mA, are critical for minimizing side reactions. This gentle oxidation environment preserves the stereochemistry of the sensitive amino acid residues, particularly the D-Arginine, which is crucial for the biological activity of the drug. The electrochemical mechanism effectively bypasses the kinetic traps that often lead to inter-chain disulfide mismatches in solution-phase chemical oxidation.
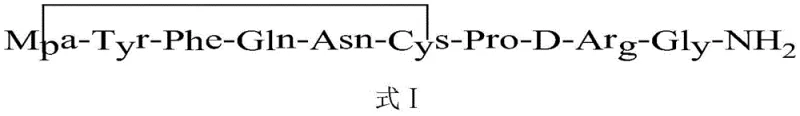
Impurity control is another critical aspect where this mechanism excels. In traditional chemical oxidation, the presence of excess oxidant can lead to the formation of sulfoxides or sulfones, which are structurally similar to the target molecule and difficult to separate. The electrochemical method, however, allows for the reaction to be stopped precisely when the theoretical amount of charge has been passed, preventing over-oxidation. Additionally, the liquid-phase nature of the cyclization step ensures homogeneous reaction conditions, unlike solid-phase oxidation which can suffer from diffusion limitations and incomplete conversion. The patent data indicates that this method achieves a crude peptide purity of 88.9% before the final purification step, which is exceptionally high for peptide synthesis. The final purification via reversed-phase high-performance liquid chromatography further refines the product to a purity of 99.98%, meeting the stringent specifications required for injectable pharmaceutical products. This high level of purity reduces the risk of immunogenic responses in patients and ensures consistent therapeutic efficacy.
How to Synthesize Desmopressin Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-efficiency process in a laboratory or pilot plant setting. The procedure begins with the selection of an appropriate solid-phase carrier, such as Rink Amide resin, which is essential for generating the C-terminal amide required for Desmopressin. The amino acid coupling sequence is strictly defined, proceeding from the C-terminus to the N-terminus using activated esters formed by DIPCDI and HOBt. This ensures minimal racemization and high coupling yields at each step. Following the assembly of the linear chain, the cleavage step utilizes a TFA-based cocktail to simultaneously remove the peptide from the resin and deprotect the side chains. The resulting crude linear peptide is then immediately subjected to the electrochemical oxidation step. Detailed standardized synthesis steps see the guide below.
- Select Rink Amide resin and synthesize the backbone peptide sequence using Fmoc/tBu strategy with DIPCDI/HOBt coupling agents.
- Cleave the peptide from the resin using a TFA-based cocktail to remove protecting groups and obtain the crude linear peptide.
- Perform liquid-phase electrochemical oxidation using platinum electrodes to form the critical disulfide bond without chemical oxidants.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical synthesis route offers substantial strategic advantages beyond mere technical novelty. The primary benefit lies in the significant cost reduction in pharmaceutical intermediates manufacturing driven by improved process efficiency. By eliminating the need for stoichiometric amounts of expensive and hazardous chemical oxidants like iodine, the raw material costs are directly lowered. Furthermore, the reduction in impurity formation translates to higher yields in the final purification step, meaning more saleable product is obtained from the same amount of starting materials. This improvement in atom economy is a powerful lever for reducing the overall cost of goods. Additionally, the simplified waste profile, devoid of heavy metal contaminants, reduces the cost and complexity of environmental compliance and waste disposal. These factors combine to create a more resilient and cost-effective supply chain for this critical API.
- Cost Reduction in Manufacturing: The elimination of chemical oxidants and the associated scavenging agents removes a significant cost center from the production budget. Traditional methods often require multiple washing and treatment steps to remove iodine residues, which consume solvents and time. The electrochemical method streamlines this workflow, allowing for a more direct path from crude peptide to purified product. The higher utilization rate of the amino acid fragments also means that less raw material is wasted, further driving down the variable costs per kilogram. This efficiency gain is crucial for maintaining competitiveness in the global market for peptide therapeutics.
- Enhanced Supply Chain Reliability: Reliance on specialized chemical oxidants can sometimes introduce supply chain bottlenecks, especially if regulatory restrictions on certain reagents tighten. By shifting to an electrochemical process, the manufacturing dependency is moved towards electricity and standard electrode materials, which are generally more stable and universally available. This diversification of input materials enhances the robustness of the supply chain against external shocks. Moreover, the simplified process flow reduces the overall cycle time, allowing for faster turnaround on orders and reducing lead time for high-purity pharmaceutical intermediates. This agility is vital for meeting the dynamic demands of the pharmaceutical market.
- Scalability and Environmental Compliance: The electrochemical oxidation step is inherently scalable, as electrochemical cells can be designed to handle large volumes with consistent current density. This facilitates the commercial scale-up of complex peptide additives without the need for massive reactors or complex mixing systems. From an environmental perspective, the process aligns with increasingly strict global regulations on hazardous waste. The absence of iodine waste simplifies the permitting process for manufacturing facilities and reduces the risk of regulatory fines or shutdowns. This sustainability profile is increasingly becoming a key criterion for procurement decisions by major pharmaceutical companies committed to green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation. They are intended to provide clarity on the feasibility and advantages of adopting this method for large-scale production. Understanding these details is crucial for stakeholders evaluating the potential integration of this technology into their existing manufacturing portfolios.
Q: How does electrochemical oxidation improve Desmopressin purity compared to traditional methods?
A: Traditional methods using iodine or hydrogen peroxide often lead to over-oxidation or mismatched disulfide impurities. The electrochemical method described in patent CN112062813B allows for precise control of the oxidation potential, significantly reducing side reactions and improving the purity of the final cyclic peptide to over 99%.
Q: What are the environmental benefits of this synthesis route?
A: This route eliminates the need for stoichiometric chemical oxidants like iodine, which generate hazardous waste. By using electricity as the reagent for disulfide bond formation, the process aligns with green chemistry principles, reducing the environmental footprint and simplifying waste treatment protocols.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method utilizes standard solid-phase synthesis equipment and scalable electrochemical cells. The high atom economy and improved yield of the backbone fragments make it economically viable for scaling from pilot batches to multi-ton commercial production of peptide APIs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Desmopressin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the electrochemical synthesis route for Desmopressin and are well-positioned to leverage this technology for our partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art electrochemical reactors and solid-phase synthesis units capable of executing this precise protocol. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the highest international standards. Our team of process chemists is dedicated to optimizing this route further to maximize yield and minimize environmental impact, ensuring a sustainable supply of this vital medication.
We invite procurement leaders and R&D directors to collaborate with us to explore how this advanced synthesis method can optimize your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a reliable, high-quality, and cost-effective supply of Desmopressin that meets your commercial and regulatory needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
