Revolutionizing Eltrombopag Production: A Scalable, Protection-Free Synthetic Strategy
Revolutionizing Eltrombopag Production: A Scalable, Protection-Free Synthetic Strategy
The pharmaceutical industry is constantly seeking more efficient pathways to synthesize complex active pharmaceutical ingredients (APIs) and their precursors. A groundbreaking development in this domain is detailed in patent CN112321454B, which discloses a novel intermediate for Eltrombopag, a crucial medication for treating thrombocytopenia. This patent introduces a streamlined synthetic methodology that fundamentally alters the production landscape by eliminating cumbersome protection and deprotection steps traditionally associated with phenolic hydroxyl groups. By leveraging a direct diazotization and coupling strategy, this innovation not only simplifies the molecular architecture but also drastically enhances the feasibility of industrial amplification. For stakeholders in the fine chemical sector, this represents a pivotal shift towards greener, more cost-effective manufacturing protocols that align with modern sustainability goals while maintaining rigorous purity standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Eltrombopag and its key intermediates has been plagued by inefficient multi-step sequences that rely heavily on protecting group chemistry. As illustrated in prior art such as WO2013072921, traditional routes often necessitate the protection of hydroxyl functionalities using groups like benzyl or methyl ethers to prevent unwanted side reactions during subsequent transformations. 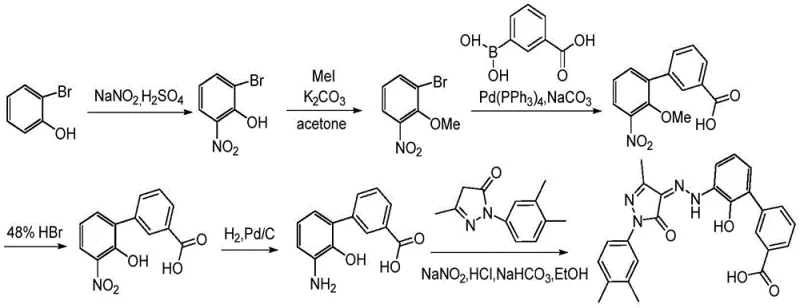 This reliance on protection strategies inherently inflates the step count, requiring additional reagents for both the installation and removal of these protecting groups. Furthermore, methods disclosed in patents like EP2799425 often involve the reduction of nitro groups using palladium on carbon (Pd/C) alongside deprotection steps, which generates substantial volumes of hazardous waste liquid. These legacy processes impose a heavy environmental burden and escalate production costs due to the consumption of expensive catalysts and the complexity of downstream purification, rendering them less competitive in a market that demands high efficiency and low ecological impact.
This reliance on protection strategies inherently inflates the step count, requiring additional reagents for both the installation and removal of these protecting groups. Furthermore, methods disclosed in patents like EP2799425 often involve the reduction of nitro groups using palladium on carbon (Pd/C) alongside deprotection steps, which generates substantial volumes of hazardous waste liquid. These legacy processes impose a heavy environmental burden and escalate production costs due to the consumption of expensive catalysts and the complexity of downstream purification, rendering them less competitive in a market that demands high efficiency and low ecological impact.
The Novel Approach
In stark contrast, the methodology protected under CN112321454B offers a radically simplified pathway that bypasses the need for hydroxyl protection entirely. The core innovation lies in the direct utilization of 2-amino-6-bromophenol as a starting material, which undergoes diazotization and coupling to form the critical hydrazone intermediate without masking the phenolic hydroxyl group. 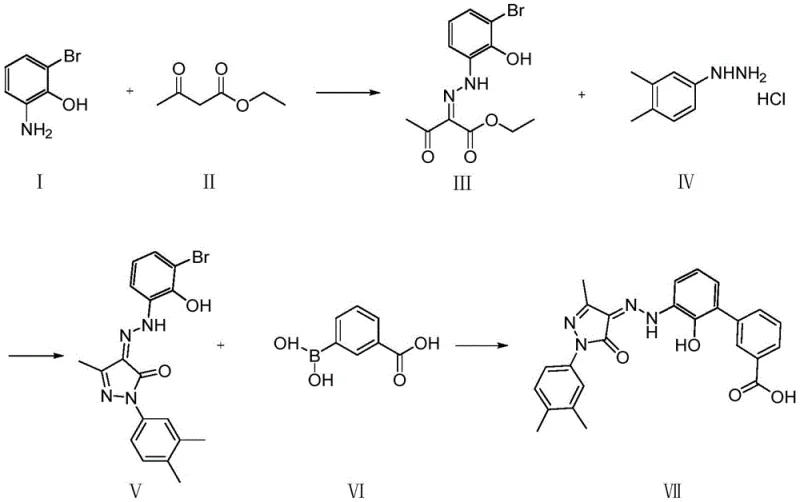 This approach collapses the synthetic tree, reducing the total number of unit operations and significantly improving the overall atom economy. The reaction system is designed to be robust and tolerant, allowing the intermediate to proceed directly to the next cyclization step without intensive post-treatment. By removing the bottlenecks associated with protection chemistry, this novel route facilitates a continuous flow of material through the pipeline, thereby enhancing throughput and reducing the residence time of intermediates in the reactor, which is a critical factor for preventing degradation and ensuring consistent quality in large-scale batches.
This approach collapses the synthetic tree, reducing the total number of unit operations and significantly improving the overall atom economy. The reaction system is designed to be robust and tolerant, allowing the intermediate to proceed directly to the next cyclization step without intensive post-treatment. By removing the bottlenecks associated with protection chemistry, this novel route facilitates a continuous flow of material through the pipeline, thereby enhancing throughput and reducing the residence time of intermediates in the reactor, which is a critical factor for preventing degradation and ensuring consistent quality in large-scale batches.
Mechanistic Insights into Diazotization and Suzuki Coupling
The chemical elegance of this new process is rooted in the precise control of reaction conditions during the initial diazotization and coupling phases. The synthesis begins with the treatment of 2-amino-6-bromophenol with sodium nitrite and a mineral acid, typically hydrochloric acid, at controlled low temperatures ranging from -10°C to 10°C. 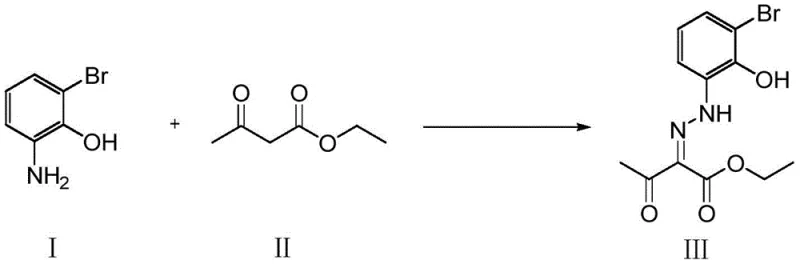 This low-temperature regime is essential for stabilizing the diazonium salt intermediate, preventing its premature decomposition which could lead to phenol formation or other deleterious byproducts. Once the diazonium species is generated in situ, it is immediately coupled with ethyl acetoacetate in a buffered aqueous-organic solvent system. The presence of the bromine atom on the aromatic ring is strategically retained throughout this sequence, serving as a vital handle for the final carbon-carbon bond-forming event. This mechanistic pathway ensures high regioselectivity and minimizes the formation of isomeric impurities, which is paramount for meeting the stringent impurity profiles demanded by pharmacopeial standards for thrombopoietin receptor agonists.
This low-temperature regime is essential for stabilizing the diazonium salt intermediate, preventing its premature decomposition which could lead to phenol formation or other deleterious byproducts. Once the diazonium species is generated in situ, it is immediately coupled with ethyl acetoacetate in a buffered aqueous-organic solvent system. The presence of the bromine atom on the aromatic ring is strategically retained throughout this sequence, serving as a vital handle for the final carbon-carbon bond-forming event. This mechanistic pathway ensures high regioselectivity and minimizes the formation of isomeric impurities, which is paramount for meeting the stringent impurity profiles demanded by pharmacopeial standards for thrombopoietin receptor agonists.
Following the formation of the hydrazone intermediate, the process advances through a condensation ring-closing reaction to construct the pyrazolone core, followed by a palladium-catalyzed Suzuki-Miyaura cross-coupling. The final step involves the coupling of the bromo-substituted pyrazolone with 3-carboxyphenylboronic acid. This transition metal-catalyzed transformation is highly efficient, utilizing ligands such as dppf to facilitate the oxidative addition and reductive elimination cycles necessary for biaryl bond formation. The choice of base and solvent system, often a mixture of ethanol and water with potassium carbonate or sodium carbonate, is optimized to solubilize the boronic acid while maintaining the stability of the sensitive pyrazolone ring. This mechanistic understanding allows for fine-tuning of the catalyst loading and reaction temperature (70-100°C) to maximize conversion while minimizing the formation of homocoupling byproducts, ensuring a clean reaction profile that simplifies the final isolation of the API.
How to Synthesize Eltrombopag Intermediate Efficiently
The practical execution of this synthesis requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent literature. The process is divided into three distinct operational stages: the formation of the hydrazone ester, the cyclization to the pyrazolone, and the final biaryl coupling. Each stage has been optimized to use commercially available reagents and standard laboratory equipment, making the technology transfer to pilot and production scales straightforward. The following guide outlines the critical operational parameters derived from the patent examples, serving as a foundational reference for process chemists aiming to implement this superior route. For the complete, standardized operating procedures and safety data sheets, please refer to the detailed technical documentation provided below.
- Perform diazotization of 2-amino-6-bromophenol followed by coupling with ethyl acetoacetate to form Intermediate III.
- Conduct condensation ring-closing reaction of Intermediate III with 3,4-dimethylphenylhydrazine to generate Intermediate V.
- Execute Suzuki coupling of Intermediate V with 3-carboxyphenylboronic acid using a palladium catalyst to yield Final Product VII.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthetic route offers profound advantages for procurement managers and supply chain directors tasked with optimizing the cost of goods sold (COGS) and ensuring supply continuity. The elimination of protection and deprotection steps translates directly into a reduction in raw material consumption, as fewer reagents are required per kilogram of final product. This simplification also reduces the demand for specialized solvents and extensive purification media, leading to a leaner inventory requirement and lower warehousing costs. Furthermore, the shortened reaction sequence decreases the overall cycle time, allowing manufacturing facilities to increase their batch turnover rate and respond more agilely to fluctuations in market demand without the need for significant capital expenditure on new reactor capacity.
- Cost Reduction in Manufacturing: The most significant economic driver of this new method is the drastic simplification of the synthetic pathway. By removing the need for hydroxyl protection, the process avoids the purchase of expensive protecting group reagents and the associated bases or acids required for their removal. Additionally, the reduction in step count minimizes the cumulative yield loss that typically occurs in multi-step syntheses, effectively increasing the mass output from a fixed amount of starting material. The use of common solvents like ethanol and water, rather than exotic or highly regulated chlorinated solvents, further drives down operational expenses related to solvent recovery and waste disposal, resulting in substantial cost savings throughout the production lifecycle.
- Enhanced Supply Chain Reliability: The starting materials for this route, such as 2-amino-6-bromophenol and ethyl acetoacetate, are commodity chemicals with robust and established global supply chains. This contrasts sharply with older methods that may rely on custom-synthesized protected intermediates, which can be subject to long lead times and supply bottlenecks. By anchoring the synthesis in readily available feedstocks, manufacturers can mitigate the risk of production delays caused by raw material shortages. Moreover, the mild reaction conditions reduce the stress on equipment, lowering maintenance frequency and the likelihood of unplanned downtime, thereby ensuring a more reliable and consistent delivery schedule to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions that are easily manageable in large stainless steel reactors. The absence of hazardous reagents like strong reducing agents for nitro groups (in the context of the new route's specific steps) and the minimization of waste streams align perfectly with increasingly stringent environmental regulations. The simplified workup procedures, often involving simple filtration and crystallization, reduce the load on wastewater treatment facilities and lower the carbon footprint of the manufacturing site. This environmental compatibility not only future-proofs the supply chain against regulatory changes but also enhances the corporate social responsibility profile of the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Eltrombopag intermediate synthesis. These insights are derived directly from the experimental data and claims within patent CN112321454B, providing a transparent view of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What are the key advantages of the new synthetic route for Eltrombopag intermediates?
A: The new route eliminates the need for hydroxyl protection and deprotection steps, significantly reducing reaction steps, waste generation, and overall production costs compared to prior art methods.
Q: What yields can be expected from this patented process?
A: The patent demonstrates high yields across all steps, with Intermediate III achieving up to 95% yield, Intermediate V up to 94%, and the final product VII reaching over 85% purity and yield under optimized conditions.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes mild reaction conditions, common solvents like ethanol and water, and simple post-treatment methods such as filtration, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eltrombopag Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN112321454B for the global supply of thrombocytopenia treatments. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into a robust, commercial-scale manufacturing process. Our facilities are equipped to handle complex organic syntheses, including sensitive diazotization reactions and palladium-catalyzed couplings, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering stringent purity specifications through our rigorous QC labs, ensuring that every batch of Eltrombopag intermediate meets the exacting standards required for pharmaceutical registration and patient safety.
We invite pharmaceutical companies and generic drug manufacturers to collaborate with us to leverage this cost-effective and environmentally friendly synthesis. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the marketplace with a reliable, high-quality supply of Eltrombopag intermediates.
