Optimizing Praziquantel Production: A Technical Analysis of Patent CN101445507B for Commercial Scale-Up
Optimizing Praziquantel Production: A Technical Analysis of Patent CN101445507B for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for essential anthelmintic agents like praziquantel. Patent CN101445507B, filed in late 2010, introduces a refined methodology that addresses critical bottlenecks in traditional manufacturing, specifically focusing on reaction step reduction and energy conservation. This technical insight report analyzes the proprietary three-step synthesis detailed in the patent, evaluating its viability for large-scale commercial production. By leveraging isoquinoline as a primary starting material and optimizing catalytic conditions, the disclosed process offers a compelling alternative to legacy routes that suffer from harsh reaction conditions and prohibitive raw material costs. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable praziquantel intermediate supplier capable of meeting stringent global quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of praziquantel has been plagued by significant logistical and chemical challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional routes often rely on diacetonitrile amine as a main raw material, a substance that is notoriously difficult to source reliably in bulk quantities, leading to supply chain volatility and inflated costs. Alternatively, methods utilizing phenylethylamine, while greener, require expensive reagents such as halo dimethylacetal, which drastically increases the cost of goods sold and renders the process economically unviable for mass market distribution. Furthermore, the most widely adopted existing method using isoquinoline typically involves an excessively long production process with complicated operational steps at each stage. These legacy processes demand high energy consumption and intricate control measures, resulting in thinner profit margins and a higher risk of batch-to-batch variability due to the complexity of the unit operations involved.
The Novel Approach
In stark contrast, the methodology outlined in CN101445507B streamlines the entire synthetic sequence into just three distinct reaction steps, fundamentally altering the economic landscape of praziquantel manufacturing. This novel approach capitalizes on the high availability and low cost of isoquinoline, eliminating the dependency on scarce or prohibitively expensive precursors. By optimizing the reaction conditions to be significantly milder, the process reduces the thermal and pressure loads on equipment, thereby lowering energy consumption and extending the lifespan of reactor infrastructure. The simplicity of the technology allows for routine operational control, meaning that complex automation is not a prerequisite for success, making it highly adaptable for facilities aiming for cost reduction in pharmaceutical intermediates manufacturing. Additionally, the avoidance of benzoyl chloride in favor of more manageable acylating agents further simplifies the waste treatment profile, aligning with modern green chemistry principles.
Mechanistic Insights into the Three-Step Synthetic Route
The core of this innovation lies in a carefully orchestrated sequence of nucleophilic addition, catalytic reduction, and cyclization. The first step involves the reaction of isoquinoline with sodium cyanide or potassium cyanide in the presence of a base such as triethylamine or pyridine. This nucleophilic attack generates a cyanide-substituted intermediate, a critical junction where the nitrogen heterocycle is functionalized for subsequent reduction. The patent specifies precise molar ratios, typically ranging from 1:1.05 to 1.2 for the cyanide source relative to isoquinoline, ensuring complete conversion while minimizing excess reagent waste. The reaction is conducted in a biphasic system involving water and 1,2-dichloroethane, cooled to temperatures between -25°C and -15°C to control exothermicity and prevent side reactions, yielding a yellow solid intermediate with high fidelity.
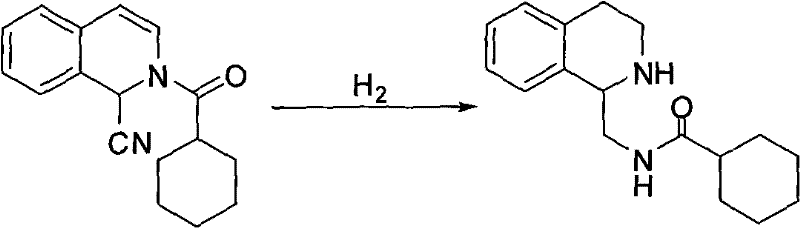
Following the formation of the nitrile intermediate, the process advances to a high-pressure hydrogenation stage, which is pivotal for establishing the saturated heterocyclic core of the praziquantel molecule. As illustrated in the reaction scheme, this step utilizes a Ruthenium on Carbon (Ru/C) catalyst, typically containing 5% ruthenium, to facilitate the reduction under hydrogen pressures of 2 to 4 MPa. The reaction temperature is maintained between 40°C and 60°C in anhydrous methanol, conditions that are sufficiently vigorous to reduce both the nitrile group and the aromatic double bonds without degrading the sensitive molecular framework. This catalytic choice is superior to traditional metal hydride reductions, as it allows for easy catalyst recovery via filtration and reuse, significantly contributing to the overall cost reduction in pharmaceutical intermediates manufacturing. The resulting second-step product is an amine derivative that serves as the direct precursor for the final ring closure.
How to Synthesize Praziquantel Efficiently
Executing this synthesis requires strict adherence to the stoichiometric and thermal parameters defined in the patent to ensure the highest possible yield and purity profile. The process begins with the preparation of the cyanide adduct, followed by the critical hydrogenation step which sets the stereochemical and structural foundation for the final API. The final transformation involves a cyclization reaction where the amine intermediate reacts with chloroacetyl chloride in the presence of anhydrous potassium carbonate. This step effectively closes the piperazine ring, forming the characteristic praziquantel structure. The use of ethyl acetate as a solvent in this final stage provides an optimal medium for the precipitation of the crude product, which is subsequently purified via recrystallization from anhydrous methanol to achieve pharmaceutical grade specifications.
- React isoquinoline with sodium cyanide or potassium cyanide in the presence of triethylamine or pyridine to form a cyanide-substituted intermediate.
- Subject the first intermediate to high-pressure hydrogenation using a Ruthenium on Carbon (Ru/C) catalyst to reduce the nitrile and double bonds.
- Cyclize the resulting amine intermediate with chloroacetyl chloride in ethyl acetate using anhydrous potassium carbonate as a base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage is the drastic simplification of the raw material portfolio; by relying on isoquinoline and common inorganic salts like sodium cyanide, manufacturers can mitigate the risks associated with sourcing exotic or monopolized reagents. This shift ensures a more stable supply chain, reducing lead time for high-purity pharmaceutical intermediates and protecting production schedules from upstream volatility. Furthermore, the elimination of complex purification steps and the ability to use standard mechanical installations mean that capital expenditure for new capacity is minimized, allowing for rapid deployment of manufacturing lines to meet surging global demand for anthelmintic treatments.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the reduction in reaction steps and energy intensity. By condensing the synthesis into three steps and utilizing a recoverable heterogeneous catalyst (Ru/C), the operational expenditure is significantly lowered compared to multi-step legacy routes. The avoidance of expensive reagents like halo dimethylacetal and the optimization of solvent usage directly translate to substantial cost savings per kilogram of finished product. Additionally, the milder reaction conditions reduce the utility load on the facility, further enhancing the margin profile for commercial producers.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on commodity chemicals such as isoquinoline and chloroacetyl chloride ensures that production is not held hostage by single-source suppliers. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in environmental controls, leading to consistent batch quality and reliable delivery schedules. This stability is crucial for maintaining the continuity of supply for essential medicines, particularly in regions where praziquantel is a critical component of public health initiatives against parasitic infections.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing unit operations that are standard in the fine chemical industry, such as high-pressure hydrogenation and crystallization. The simplified workflow reduces the generation of hazardous waste streams, facilitating easier compliance with increasingly stringent environmental regulations. The ability to apply existing mechanical installations without major retrofitting allows for a seamless transition from pilot scale to commercial production, ensuring that capacity can be ramped up quickly to address market needs without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: This method significantly shortens the reaction process compared to prior art, utilizing readily available raw materials like isoquinoline instead of expensive or hard-to-source precursors. It operates under milder conditions, reducing energy consumption and simplifying operational control, which translates to lower production costs and higher overall yields.
Q: How does this process address impurity control and product purity?
A: The process utilizes specific recrystallization steps using anhydrous methanol to refine the crude product. Furthermore, the use of selective catalytic hydrogenation with Ru/C minimizes side reactions common in harsher reduction methods, ensuring a cleaner impurity profile suitable for pharmaceutical applications.
Q: Is this method scalable for industrial manufacturing?
A: Yes, the methodology is designed for scalability. It avoids complex apparatus requirements and relies on routine unit operations such as high-pressure hydrogenation and standard stirring reactions. The elimination of difficult-to-handle reagents facilitates green production and easier adaptation to existing mechanical installations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Praziquantel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101445507B are fully realized in practice. We are committed to delivering high-purity praziquantel intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to process optimization allows us to offer competitive pricing without compromising on the quality or consistency that global regulatory bodies demand.
We invite potential partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your supply chain objectives. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and cost-effective chemical solutions available in the market.
