Revolutionizing Praziquantel Manufacturing: A Deep Dive into the Novel One-Pot Cyclization Technology
Revolutionizing Praziquantel Manufacturing: A Deep Dive into the Novel One-Pot Cyclization Technology
The pharmaceutical industry is constantly seeking methodologies that balance high purity with operational efficiency, particularly for essential anti-parasitic agents like Praziquantel. Patent CN103570710A introduces a groundbreaking preparation process that fundamentally shifts the paradigm from multi-step, hazardous syntheses to a streamlined, one-pot reaction system. This innovation addresses critical bottlenecks in the existing supply chain by eliminating intermediate isolation steps, thereby drastically reducing raw material ratios and simplifying post-reaction processing. For R&D directors and procurement managers alike, this technology represents a significant leap forward, offering a pathway to achieve yields exceeding 95% while maintaining stringent purity specifications through simple aftertreatment protocols. The ability to synthesize high-purity Praziquantel using inexpensive, readily available starting materials under mild conditions marks a pivotal advancement for reliable praziquantel supplier networks aiming to secure long-term production stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Praziquantel has been plagued by significant safety and efficiency challenges that hinder optimal cost reduction in API manufacturing. The most prevalent industrial route, often referred to as Route 1, relies on isoquinoline as a starting material and necessitates the use of highly toxic potassium cyanide alongside high-pressure hydrogenation operations. This combination creates severe safety hazards for plant personnel and imposes rigorous, costly requirements for three-waste treatment and environmental compliance. Alternative pathways, such as Route 3 and Route 5, attempt to circumvent these toxicity issues but fail on economic grounds due to unacceptably low yields ranging from merely 15% to 32%. Furthermore, solid-phase synthesis methods (Route 4) introduce complexities regarding resin supply chains and recovery, while other solution-phase routes often leave difficult-to-remove residues like methanesulfonic acid in the final product, compromising quality and necessitating expensive purification steps that erode profit margins.
The Novel Approach
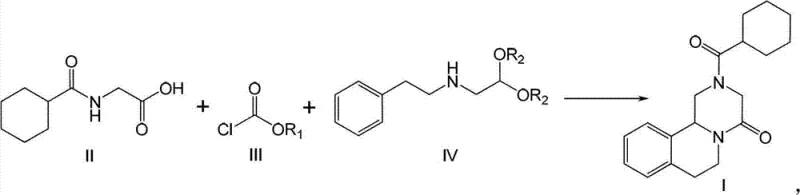
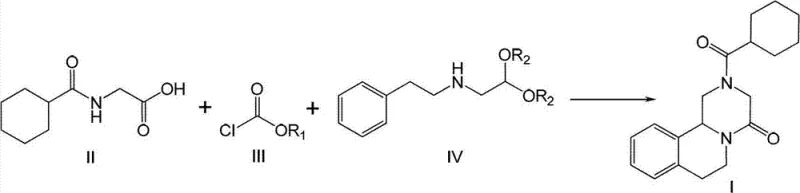
In stark contrast to these fragmented and hazardous legacy methods, the novel approach detailed in the patent consolidates the synthesis into a cohesive, telescoped one-pot reaction that maximizes atom economy and operational safety. By reacting N-cyclohexylcarbonyl glycine with a chloroformate derivative followed by the direct addition of a phenylethylamine acetal derivative, the process constructs the complex piperazine backbone without ever isolating unstable intermediates. This seamless integration allows for the direct addition of a strong acid to induce cyclization within the same vessel, effectively merging condensation, amidation, and ring-closure into a single continuous operation. The result is a drastic simplification of the workflow where the high-purity product can be obtained simply by filtration or basic extraction, bypassing the need for complex chromatography or recrystallization sequences that typically drive up production costs and lead times.
The strategic advantage of this methodology lies not just in the chemical transformation but in the profound impact it has on the commercial viability of the supply chain. By removing the necessity for high-pressure equipment and toxic cyanide reagents, the barrier to entry for safe manufacturing is lowered, allowing for more flexible production scheduling and reduced insurance liabilities. The use of mild reaction temperatures, typically between 5°C and 50°C, further ensures that energy consumption is minimized compared to the high-thermal demands of older routes. For supply chain heads, this translates to a more robust and resilient sourcing strategy where the risk of production stoppages due to safety incidents or regulatory scrutiny is significantly mitigated, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream formulation partners.
Mechanistic Insights into One-Pot Telescoped Cyclization
At the heart of this technological breakthrough is a sophisticated understanding of nucleophilic substitution and acid-catalyzed cyclization mechanisms that allow for such high convergence. The process initiates with the activation of the glycine derivative (Formula II) using a chloroformate (Formula III) in the presence of an acid-binding agent, generating a highly reactive mixed anhydride or activated ester species in situ. This activated intermediate is immediately susceptible to nucleophilic attack by the amine group of the phenylethylamine derivative (Formula IV), forming the linear amide precursor without the need for isolation. The brilliance of the design is evident in the subsequent step, where the introduction of a strong acid, such as sulfuric acid, serves a dual purpose: it catalyzes the deprotection of the acetal group to reveal the reactive aldehyde equivalent and simultaneously promotes the intramolecular electrophilic aromatic substitution or iminium cyclization required to close the tetrahydroisoquinoline ring system.
From an impurity control perspective, this one-pot design is exceptionally elegant because it prevents the accumulation of side products that often arise during the handling and storage of isolated intermediates. In traditional multi-step syntheses, each isolation event introduces opportunities for hydrolysis, oxidation, or thermal degradation, leading to a complex impurity profile that is difficult to purge. By maintaining the reaction mixture in a controlled environment and transitioning directly from amidation to cyclization, the process minimizes the exposure of sensitive functional groups to adverse conditions. Furthermore, the specific selection of solvents like methylene dichloride, ether, or methyl tert-butyl ether allows for precise control over solubility and phase separation during the workup, ensuring that inorganic salts and acidic byproducts are efficiently removed in the aqueous phase, leaving the organic phase enriched with the target molecule ready for final crystallization.
How to Synthesize Praziquantel Efficiently
Implementing this advanced synthesis route requires careful attention to stoichiometry and temperature control to replicate the high yields reported in the patent data. The process begins with the preparation of the key glycine intermediate, which can be synthesized in water using cyclohexanecarbonyl chloride, a green chemistry feature that eliminates organic solvent waste at the very first stage. Following this, the one-pot sequence is executed by sequentially adding reagents to a single reactor, monitoring the completion of each stage via TLC or HPLC before proceeding to the next without workup. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and temperature ramps required to achieve the reported >95% yield, are outlined in the technical guide below for process engineers to review.
- Condensation Reaction: React N-cyclohexylcarbonyl glycine (Formula II) with vinyl chloroformate or methyl chloroformate (Formula III) in an organic solvent at -10 to 30°C using an acid binding agent.
- Amidation Step: Without separation, add N-(2,2-diethoxy)ethyl-N-(2-phenyl)ethylamine (Formula IV) to the mixture and react at 5 to 50°C to form the amide intermediate.
- Cyclization and Isolation: Directly add strong acid (e.g., sulfuric acid) to the reaction mixture at 5 to 50°C to induce ring closure, followed by aqueous workup and filtration to obtain high-purity Praziquantel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this one-pot technology offers a compelling value proposition centered on cost efficiency and operational reliability. The elimination of multiple isolation and purification steps inherently reduces the consumption of solvents, filtering aids, and labor hours, leading to substantial cost savings in the overall manufacturing budget. Unlike traditional routes that require specialized high-pressure reactors or extensive safety infrastructure for toxic reagents, this method utilizes standard glass-lined or stainless steel reactors capable of operating at atmospheric pressure and mild temperatures, thereby lowering capital expenditure requirements for new production lines. The simplified workflow also means that batch cycle times are significantly shortened, allowing facilities to increase throughput and respond more agilely to market demand fluctuations without compromising on the quality of the high-purity praziquantel delivered to clients.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the telescoping of three distinct chemical transformations into a single vessel operation. By avoiding the isolation of intermediates, manufacturers save significantly on solvent volumes required for crystallization and washing, as well as the energy costs associated with drying and concentrating multiple batches. Furthermore, the use of inexpensive and commercially available raw materials like glycine and phenylethylamine derivatives ensures that the bill of materials remains low and stable, shielding the supply chain from volatility associated with exotic or specialized reagents. The high yield of over 95% directly correlates to better raw material utilization, meaning less waste is generated per kilogram of product, which further drives down the effective cost of goods sold.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses where a failure in any single step can halt the entire production line. This robust one-pot method reduces the number of potential failure points by minimizing unit operations and handling steps. The ability to perform the initial glycine acylation in water and the subsequent steps in common organic solvents ensures that raw material sourcing is not dependent on niche suppliers with long lead times. Additionally, the mild reaction conditions reduce the risk of runaway reactions or equipment damage, ensuring that production schedules are met consistently. This reliability is crucial for maintaining the inventory levels required by global pharmaceutical customers who demand just-in-time delivery of critical anti-parasitic medications.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste generation, but this technology is inherently designed to minimize environmental impact. The reduction in solvent usage and the avoidance of toxic cyanide reagents significantly lower the burden on wastewater treatment facilities and hazardous waste disposal contractors. The process generates fewer byproducts and simplifies the three-waste treatment profile, making it easier for manufacturing sites to maintain compliance with increasingly stringent environmental regulations. This eco-friendly profile not only reduces regulatory risk but also aligns with the sustainability goals of major pharmaceutical companies, making the manufacturer a preferred partner for green chemistry initiatives and long-term contracts focused on responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Praziquantel synthesis technology. These insights are derived directly from the comparative data and experimental embodiments provided in the patent documentation, offering clarity on how this method outperforms legacy routes in terms of yield, safety, and purity. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term value of this manufacturing approach.
Q: How does the new one-pot process improve upon traditional Praziquantel synthesis routes?
A: Traditional routes often involve toxic reagents like potassium cyanide (Route 1) or suffer from low yields around 15-32% (Routes 3 and 5). The new one-pot process eliminates intermediate isolation, reduces raw material consumption, and achieves yields exceeding 95% with simpler post-treatment.
Q: What are the environmental benefits of this preparation method?
A: The process significantly reduces three-waste treatment requirements by combining multiple steps into a single reactor. Additionally, the preparation of key intermediates can be performed in water or under solvent-free conditions, minimizing organic solvent usage and enhancing environmental compliance.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the technology is specifically designed for industrial suitability. It uses inexpensive, easily available raw materials, operates under mild reaction conditions (5-50°C), and avoids high-pressure hydrogenation, making it safer and more cost-effective for mass production compared to legacy methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Praziquantel Supplier
As the global demand for effective anti-parasitic treatments continues to rise, the need for a manufacturing partner capable of delivering high-quality Praziquantel at scale has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one-pot cyclization process to ensure consistent supply and superior product quality. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of multinational pharmaceutical corporations without delay. With our stringent purity specifications and rigorous QC labs, we guarantee that every batch of Praziquantel meets the highest international standards, free from the toxic residues and impurities often associated with older synthesis routes.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this superior technology. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how switching to our process can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-purity pharmaceutical intermediates and APIs in the global market.
