Advanced Solid-Phase Cyclization Strategy for High-Purity Carbetocin Manufacturing
Introduction to Advanced Carbetocin Synthesis Technology
The pharmaceutical landscape for uterotonic agents has been significantly advanced by the disclosure in patent CN112409458A, which details a robust preparation method for Carbetocin, a synthetic long-acting oxytocin analogue. This molecule is critical for preventing postpartum hemorrhage, distinguished by its unique thioether bridge which offers superior metabolic stability compared to the disulfide bond in natural oxytocin. The patent introduces a novel solid-phase synthesis strategy that addresses the longstanding challenges of low yield and high impurity profiles associated with traditional liquid-phase cyclization methods. By utilizing a specialized cysteine derivative with a removable side-chain protecting group, the process enables efficient on-resin ring closure. This technological breakthrough not only enhances the chemical integrity of the final product but also streamlines the manufacturing workflow, positioning it as a vital development for any reliable carbetocin supplier aiming to meet global quality standards.
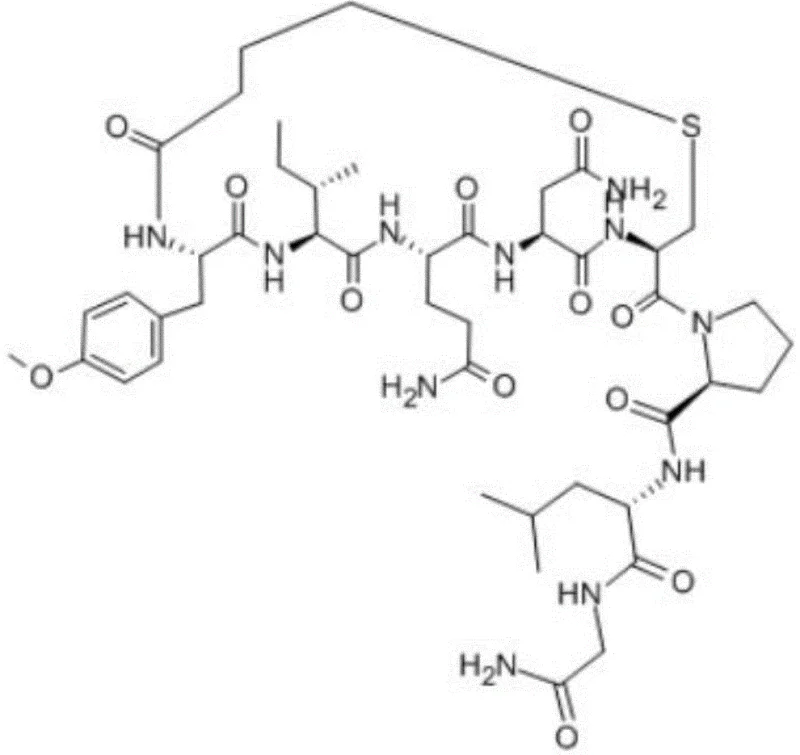
Carbetocin functions as a potent agonist with a prolonged duration of action, making it indispensable in modern obstetric care. The structural modification at the N-terminal, where the cysteine is replaced by a butyric acid moiety forming a thioether bond with the C-terminal cysteine, is the key to its enhanced bioactivity and resistance to enzymatic degradation. However, synthesizing this specific cyclic nonapeptide has historically been fraught with difficulties, particularly in forming the macrocycle without generating polymeric byproducts. The methodology described in CN112409458A overcomes these hurdles by anchoring the peptide chain to a solid support throughout the cyclization event. This approach effectively mimics high-dilution conditions inherently, thereby suppressing intermolecular reactions that typically plague solution-phase synthesis. For procurement managers and supply chain heads, understanding this shift from liquid to solid-phase cyclization is crucial, as it directly correlates to improved batch consistency and reduced production lead times.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Carbetocin has relied heavily on liquid-phase cyclization or hybrid solid-liquid strategies that introduce significant inefficiencies into the manufacturing process. Prior art, such as European patent ES2115543 and Czech patent CS8605461, often necessitates the cleavage of the linear peptide from the resin before attempting ring closure in solution. This liquid-phase oxidation or cyclization step is notoriously problematic; it requires extremely dilute conditions to prevent intermolecular disulfide or thioether bond formation between different peptide chains, leading to the generation of dimers and higher-order polymers. Furthermore, some legacy methods employ expensive reagents like TCEP·HCl to prevent unwanted disulfide formation, or require high-pressure hydrogenation steps to remove benzyl-based protecting groups, which introduces safety risks and complicates scale-up. The comparative examples in the patent data highlight these deficiencies starkly, showing that liquid-phase cyclization can result in crude peptide purities as low as 24% and overall yields dropping to approximately 14-16%, rendering such processes economically unviable for cost reduction in peptide manufacturing.
The Novel Approach
In contrast, the novel approach detailed in CN112409458A leverages a sophisticated Fmoc solid-phase synthesis strategy that keeps the peptide anchored to the resin during the critical cyclization step. The core innovation lies in the use of a specifically protected cysteine derivative, Fmoc-Cys(CH2CH2CH2CO2R1)-OH, where the R1 group is orthogonal to the resin linker. This allows for the selective removal of the side-chain protecting group under mild acidic conditions—such as 1-4% TFA in DCM—without detaching the growing peptide chain from the solid support. Once the side-chain carboxylic acid is exposed, it is activated and condensed with the N-terminal amine of the tyrosine residue while the molecule is still immobilized. This on-resin cyclization effectively eliminates the need for high-dilution techniques and prevents the formation of intermolecular polymers. The result is a dramatic improvement in process efficiency, with crude peptide purities exceeding 85% and total yields surpassing 80%, demonstrating a clear pathway for the commercial scale-up of complex peptide intermediates.
Mechanistic Insights into On-Resin Thioether Bridge Formation
The mechanistic elegance of this synthesis lies in the precise control of orthogonality between the resin linker, the N-terminal Fmoc group, and the side-chain R1 protecting group on the cysteine residue. The process begins with the sequential coupling of amino acids onto an amino-functionalized resin, such as Rink Amide-AM resin, building the linear sequence from the C-terminus to the N-terminus. The critical intermediate is the fully protected peptide resin where the cysteine side chain bears the R1 group (e.g., 2-Chlorotrityl or 2,4-dimethoxybenzyl). Upon completion of the linear sequence, the N-terminal Fmoc group is removed using standard piperidine treatment. Subsequently, a mild acid treatment selectively cleaves the R1 ester linkage on the cysteine side chain, liberating the free carboxylic acid required for cyclization while leaving the acid-labile resin linker intact. This selective deprotection is the linchpin of the entire strategy, ensuring that the reactive functionalities are generated in the correct spatial proximity for intramolecular reaction.
Following the exposure of the side-chain carboxyl group, the cyclization is effected using standard peptide coupling reagents such as HOBt and DIC directly on the solid phase. The immobilization of the peptide chain creates a "pseudo-dilution" effect, where the effective concentration of the reactive ends favors intramolecular ring closure over intermolecular polymerization. This mechanistic advantage is crucial for impurity control, as it virtually eliminates the formation of dimeric or oligomeric byproducts that are common in solution-phase reactions. The resulting cyclic peptide resin is then subjected to global cleavage using a cocktail like TFA/phenol/water/thioanisole/EDT, which simultaneously releases the peptide from the resin and removes remaining side-chain protecting groups. This streamlined mechanism ensures that the final crude product is of exceptionally high quality, significantly reducing the burden on downstream purification processes and enhancing the overall viability of high-purity pharmaceutical intermediate production.
How to Synthesize Carbetocin Efficiently
The synthesis of Carbetocin via this optimized solid-phase route involves a carefully orchestrated sequence of coupling, deprotection, and on-resin cyclization steps designed to maximize yield and purity. The process initiates with the swelling of the amino resin followed by the iterative addition of Fmoc-protected amino acids, utilizing activation agents like HOBt and DIC to ensure high coupling efficiency at each position. Special attention is paid to the incorporation of the modified cysteine residue and the subsequent selective deprotection steps that enable the formation of the thioether bridge. While the general workflow follows standard SPPS protocols, the specific conditions for side-chain deprotection and cyclization are critical parameters that distinguish this method from conventional approaches. For a detailed breakdown of the specific reagent quantities, reaction times, and temperature controls required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Sequentially couple Fmoc-protected amino acids (Gly, Leu, Pro, Cys derivative, Asn, Gln, Ile, Tyr) onto amino resin using HOBt/DIC activation.
- Remove the Fmoc group and selectively deprotect the Cys side-chain carboxyl group (R1) using mild acidic conditions (e.g., 1-4% TFA/DCM) without cleaving the peptide from the resin.
- Perform on-resin cyclization by condensing the exposed side-chain carboxyl group with the N-terminal amine, followed by cleavage, purification, and lyophilization.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from legacy liquid-phase cyclization to this advanced on-resin methodology offers profound commercial advantages that extend far beyond simple yield improvements. For procurement managers focused on cost reduction in peptide manufacturing, the elimination of expensive reagents and the reduction of solvent volumes are key drivers of economic efficiency. Traditional methods often require vast amounts of solvent to maintain the high-dilution conditions necessary for liquid-phase cyclization, creating significant waste disposal costs and environmental burdens. By contrast, the solid-phase approach confines the reaction to the resin bead, drastically reducing solvent consumption and simplifying waste management. Furthermore, the avoidance of high-pressure hydrogenation steps and expensive scavengers like TCEP lowers the raw material cost profile, making the overall process more resilient to supply chain fluctuations and reagent price volatility.
- Cost Reduction in Manufacturing: The significant increase in crude peptide purity observed in the patent data implies a substantial reduction in downstream processing costs. When crude purity rises from approximately 24% to over 85%, the load on preparative HPLC columns is dramatically decreased, extending column life and reducing the volume of mobile phase required for purification. This efficiency gain translates directly into lower operational expenditures per gram of active pharmaceutical ingredient. Additionally, the higher overall yield means that less starting material is wasted, optimizing the utilization of expensive protected amino acids and further driving down the cost of goods sold without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the simplicity and robustness of this solid-phase protocol enhance production reliability and shorten lead times. The removal of complex and hazardous steps, such as high-pressure hydrogenation or sensitive liquid-phase oxidations, reduces the risk of batch failures and equipment downtime. The use of standard Fmoc chemistry and readily available coupling reagents ensures that the supply chain is not dependent on niche or single-source specialty chemicals. This flexibility allows for more agile manufacturing schedules and ensures a consistent supply of high-purity Carbetocin, which is critical for meeting the rigorous demands of global pharmaceutical markets and regulatory bodies.
- Scalability and Environmental Compliance: The inherent scalability of solid-phase peptide synthesis makes this method ideally suited for commercial scale-up of complex peptide intermediates. The process avoids the generation of polymeric impurities that are difficult to separate, thereby simplifying the purification train and reducing the environmental footprint associated with solvent-intensive chromatography. The ability to perform the cyclization on-resin also minimizes the handling of reactive intermediates in solution, improving operator safety and facilitating compliance with increasingly stringent environmental and safety regulations. This combination of scalability and sustainability positions the technology as a future-proof solution for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Carbetocin using this novel solid-phase cyclization technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this synthesis route for large-scale production. The answers below clarify the mechanistic advantages and practical benefits that differentiate this approach from traditional manufacturing methods.
Q: Why is solid-phase cyclization superior to liquid-phase methods for Carbetocin?
A: Liquid-phase cyclization often suffers from intermolecular polymerization and requires extremely dilute conditions, leading to low yields (around 14-16%). Solid-phase on-resin cyclization utilizes the pseudo-dilution effect to favor intramolecular ring closure, achieving crude purities over 85% and total yields exceeding 80%.
Q: What is the key advantage of the Cys(CH2CH2CH2CO2R1) derivative used in this process?
A: The R1 protecting group (such as 2-Cl-Trt or Dmb) can be removed under very mild acidic conditions that do not cleave the peptide from the resin. This allows the side-chain carboxylic acid to be exposed for cyclization while the peptide backbone remains anchored, preventing intermolecular side reactions.
Q: How does this method impact the scalability of Carbetocin production?
A: By eliminating the need for high-pressure hydrogenation steps found in older patents and avoiding complex liquid-phase oxidation with expensive reagents like TCEP, this method simplifies the workflow. The high crude purity reduces the burden on preparative HPLC, making large-scale industrial production more feasible and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbetocin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to deliver high-value pharmaceutical intermediates to the global market. Our technical team has extensively analyzed the advancements presented in CN112409458A and possesses the expertise to implement this efficient on-resin cyclization strategy at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and compliant. Our facilities are equipped with state-of-the-art peptide synthesizers and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Carbetocin meets the highest international standards for safety and efficacy.
We invite pharmaceutical partners to collaborate with us to leverage these process innovations for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can optimize the production of Carbetocin, ensuring a stable, cost-effective, and high-quality supply of this essential uterotonic agent for patients worldwide.
