Advanced Manufacturing of High-Purity 4-Hydroxybenzophenone via Optimized Friedel-Crafts Acylation
Introduction to Advanced 4-Hydroxybenzophenone Manufacturing
The global demand for high-purity 4-hydroxybenzophenone continues to surge, driven by its critical applications in pharmaceutical intermediates and advanced photo-curing materials. A pivotal breakthrough in this domain is documented in patent CN101693652B, which outlines a refined synthetic methodology that addresses long-standing inefficiencies in traditional production routes. This innovation centers on the strategic substitution of reaction solvents and the precise optimization of thermal profiles during Friedel-Crafts acylation. By shifting away from volatile chlorinated aliphatic hydrocarbons to chlorobenzene, the process not only enhances the reaction kinetics but also drastically improves the recoverability of solvents, a key factor in sustainable chemical manufacturing. The technical implications of this patent extend beyond mere yield improvements; it represents a fundamental shift towards greener, more cost-effective production paradigms for fine chemical intermediates.
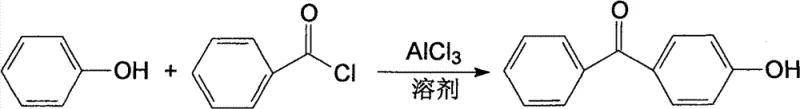
For R&D directors and process engineers, the significance of this patent lies in its ability to deliver product specifications that meet the rigorous standards of the electronics and pharmaceutical industries. The reported purity levels of 99.5% are achieved through a combination of controlled reagent addition and a multi-stage heating protocol that minimizes side reactions. Furthermore, the use of chlorobenzene as a solvent medium mitigates the environmental hazards associated with traditional solvents like methylene dichloride, aligning modern manufacturing practices with increasingly stringent global environmental regulations. This report analyzes the technical nuances of this process to provide actionable insights for procurement and supply chain stakeholders seeking reliable sources of high-performance chemical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-hydroxybenzophenone has relied heavily on traditional Friedel-Crafts acylation protocols utilizing solvents such as methylene dichloride, chloroform, or ethylene dichloride. While these solvents are effective at dissolving reactants, they suffer from significant drawbacks regarding volatility and toxicity. The low boiling points of these aliphatic chlorinated hydrocarbons necessitate energy-intensive distillation processes for recovery, often resulting in substantial solvent loss and increased operational costs. Moreover, the toxicity profile of solvents like chloroform poses serious health and safety risks to plant personnel, requiring expensive containment and ventilation systems. From a yield perspective, conventional methods often struggle to exceed 60% to 68% efficiency, with product purity frequently plateauing around 97% to 98%, necessitating additional purification steps that erode profit margins.
The Novel Approach
The novel approach detailed in the patent data introduces chlorobenzene as the primary reaction medium, fundamentally altering the thermodynamic and kinetic landscape of the synthesis. Chlorobenzene, being an aromatic solvent, exhibits better compatibility with the aromatic reactants, facilitating a more homogeneous reaction environment that promotes higher conversion rates. The process utilizes a specific molar ratio of phenol to benzoyl chloride ranging from 1:1 to 1:1.5, coupled with an aluminum trichloride catalyst loading of 1 to 2 equivalents relative to phenol. This stoichiometric precision, combined with a staged temperature profile starting at 40°C to 45°C and ramping to 60°C to 70°C, ensures complete acylation while suppressing the formation of ortho-substituted byproducts. The result is a robust process capable of delivering yields up to 89.5% with exceptional purity, effectively resolving the bottlenecks of yield and solvent recovery inherent in older technologies.
Mechanistic Insights into AlCl3-Catalyzed Friedel-Crafts Acylation
The core chemical transformation in this process is the electrophilic aromatic substitution of phenol with benzoyl chloride, mediated by the Lewis acid catalyst aluminum trichloride (AlCl3). Mechanistically, the AlCl3 coordinates with the carbonyl oxygen of the benzoyl chloride, generating a highly reactive acylium ion complex. This electrophile then attacks the electron-rich aromatic ring of the phenol. The presence of the hydroxyl group on the phenol ring acts as a strong activating group, directing the incoming acyl group predominantly to the para-position due to steric hindrance at the ortho-positions. The choice of chlorobenzene as a solvent is critical here; unlike aliphatic solvents, chlorobenzene does not compete for the catalyst nor does it participate in side reactions, thereby maintaining the integrity of the catalytic cycle. The initial addition temperature of 10°C to 50°C is carefully controlled to manage the exothermic nature of the complex formation, preventing thermal runaway that could lead to polymerization or tar formation.
Impurity control is achieved through the precise thermal staging described in the patent embodiments. By holding the reaction at 40°C to 45°C for two hours, the system allows for the gradual consumption of the acylium species, minimizing the risk of poly-acylation. The subsequent increase to 60°C to 70°C for one hour serves to drive the reaction to completion, ensuring that residual starting materials are consumed. The workup procedure involves quenching the reaction mixture into cold water, which hydrolyzes the aluminum complexes and precipitates the product. The use of toluene for recrystallization further refines the impurity profile, removing trace isomers and unreacted phenol. This multi-layered approach to purity assurance ensures that the final 4-hydroxybenzophenone meets the stringent requirements for use in sensitive applications such as UV-curable coatings and pharmaceutical syntheses where trace impurities can be detrimental.
How to Synthesize 4-Hydroxybenzophenone Efficiently
The synthesis of 4-hydroxybenzophenone via this optimized route requires strict adherence to the specified operational parameters to maximize yield and safety. The process begins with the preparation of the catalyst slurry in chlorobenzene, followed by the controlled addition of phenol to establish the reaction matrix. Subsequent addition of benzoyl chloride must be managed to maintain the thermal balance within the reactor. The detailed标准化 synthesis steps involve specific temperature ramps and hold times that are critical for reproducibility on a commercial scale. Operators must ensure that the quenching phase is conducted with sufficient agitation to prevent agglomeration of the aluminum salts, which can trap product and reduce recovery. For a comprehensive guide on the exact operational sequence and safety precautions, please refer to the standardized protocol below.
- Prepare the catalyst system by adding aluminum trichloride to chlorobenzene solvent in a reactor, followed by the dropwise addition of a phenol-chlorobenzene mixture at 10°C to 50°C.
- Introduce benzoyl chloride to the reaction mixture and stage the temperature profile: maintain 40°C to 45°C for 2 hours, then increase to 60°C to 70°C for 1 hour to drive completion.
- Quench the reaction in cold water, isolate the crude solid via centrifugation, and perform recrystallization using toluene to obtain the final white crystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chlorobenzene-based synthesis route offers tangible economic and logistical benefits that directly impact the bottom line. The primary advantage lies in the drastic reduction of solvent consumption and the enhanced efficiency of solvent recovery systems. Unlike traditional methods where low-boiling solvents are lost to the atmosphere or require complex cryogenic recovery, chlorobenzene can be easily distilled and recycled multiple times without significant degradation. This closed-loop solvent capability translates into substantial cost savings on raw material procurement and waste disposal fees. Furthermore, the higher reaction yield of nearly 90% means that less feedstock is required to produce the same amount of finished goods, effectively lowering the unit cost of production and improving overall resource utilization efficiency in the manufacturing plant.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-recover aliphatic chlorinated solvents significantly lowers the variable costs associated with production. By utilizing chlorobenzene, which is cheaper and easier to recycle, manufacturers can achieve a more stable cost structure that is less susceptible to fluctuations in solvent markets. Additionally, the improved yield reduces the cost of goods sold by maximizing the output from every kilogram of phenol and benzoyl chloride input. The simplified downstream processing, which avoids complex purification columns in favor of straightforward crystallization, further reduces energy consumption and labor costs, contributing to a leaner and more competitive manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent production schedules and reliable delivery timelines for downstream customers. Because the process is less sensitive to minor variations in temperature and mixing compared to traditional methods, batch-to-batch variability is minimized, reducing the risk of off-spec material that could disrupt supply chains. The use of widely available and stable raw materials like chlorobenzene and aluminum trichloride mitigates the risk of supply shortages that might occur with more specialized or regulated solvents. This stability allows suppliers to maintain higher inventory levels of finished goods with confidence, ensuring that just-in-time delivery commitments to pharmaceutical and electronic material clients are consistently met without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as evidenced by successful demonstrations in 1000L reactors, making it suitable for rapid expansion to meet growing market demand. From an environmental compliance perspective, the reduction in toxic solvent usage simplifies the permitting process and reduces the regulatory burden on manufacturing facilities. The lower toxicity profile of the waste stream facilitates easier treatment and disposal, aligning with corporate sustainability goals and international environmental standards such as REACH. This eco-friendly profile not only future-proofs the supply chain against tightening regulations but also enhances the brand value of the end products by associating them with green chemistry principles, a key differentiator in modern B2B markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 4-hydroxybenzophenone using this advanced methodology. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature. They are intended to clarify the operational advantages and quality benchmarks associated with this specific synthetic route. Understanding these details is crucial for technical buyers evaluating potential suppliers for long-term partnerships.
Q: Why is chlorobenzene preferred over methylene dichloride for this synthesis?
A: Chlorobenzene offers superior solvent recovery rates compared to low-boiling chlorinated aliphatic hydrocarbons like methylene dichloride. It reduces toxicity risks and significantly lowers solvent consumption per batch, enhancing both economic and environmental performance.
Q: What purity levels can be achieved with this patented process?
A: By optimizing the molar ratios of phenol to benzoyl chloride and utilizing a specific temperature gradient, this process consistently achieves product purity of 99.5% (HPLC) with yields reaching 89.5%, surpassing traditional methods that typically cap at 98% purity.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability in 1000L reactors. The simplified workup procedure involving centrifugation and straightforward solvent distillation makes it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxybenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation pharmaceuticals and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of 4-hydroxybenzophenone meets or exceeds the 99.5% purity benchmark established by leading industry patents. Our facility is equipped to handle the specific solvent recovery and safety requirements of this chlorobenzene-based process, guaranteeing a sustainable and reliable supply for your operations.
We invite you to collaborate with us to optimize your supply chain for this essential chemical building block. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our efficient manufacturing processes can reduce your total landed costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. We look forward to supporting your R&D and production goals with our premium grade intermediates and unwavering commitment to quality and service excellence.
