Advanced Acid-Catalyzed Cyclization for Scalable Indolizine Pharmaceutical Intermediates
Introduction to Next-Generation Indolizine Synthesis
The pharmaceutical industry constantly seeks more efficient and sustainable pathways for constructing nitrogen-containing heterocycles, which serve as the core scaffolds for numerous bioactive molecules. Patent CN113429407B introduces a groundbreaking advancement in this domain by disclosing a simple yet highly effective synthesis method for 1-alkyl-3-aryl substituted indolizine compounds. These structures are pivotal in medicinal chemistry, frequently appearing in central nervous system depressants, anticancer agents, and antibacterial inhibitors. The disclosed technology leverages a dual-strategy acid catalysis approach, utilizing either inorganic acid aqueous solutions or organic acid catalysts in organic solvents to drive an intramolecular dehydration cyclization. This innovation addresses critical pain points in current manufacturing, such as the reliance on scarce transition metals and complex multi-step sequences, offering a streamlined route that enhances both economic viability and environmental compliance for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of multi-substituted indolizine skeletons has relied heavily on intermolecular chemical reactions mediated by transition metal complexes. Traditional protocols often necessitate the use of expensive and potentially toxic metals such as rhodium, copper, cobalt, or palladium to facilitate cycloaddition or coupling reactions. Furthermore, alternative strategies involving pyridine ylides and alkenes via [3+2] cycloaddition followed by oxidation are not only synthetically cumbersome but also generate significant waste streams. These conventional methods suffer from high operational complexity, stringent requirement for inert atmospheres, and the challenging removal of trace metal residues, which is a critical regulatory hurdle for active pharmaceutical ingredient (API) manufacturing. The cumulative effect of these limitations results in inflated production costs, extended lead times, and a larger environmental footprint, making them less attractive for modern, green chemistry-focused procurement strategies.
The Novel Approach
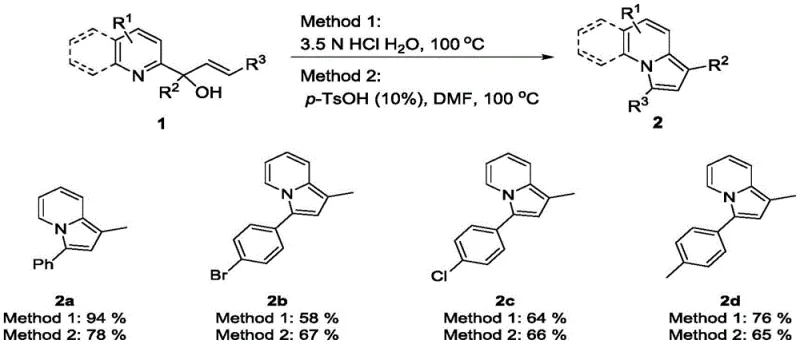
In stark contrast, the novel methodology described in the patent utilizes a direct acid-catalyzed dehydration of (E)-4-aryl-2-pyridyl-3-enyl-2-ol precursors to form the indolizine core in a single pot. This approach bifurcates into two complementary pathways: Method 1 employs inexpensive inorganic acids like hydrochloric acid in water, while Method 2 utilizes organic acids such as p-toluenesulfonic acid in polar aprotic solvents. This flexibility allows chemists to tailor the reaction conditions to the specific solubility and electronic properties of the substrate, ensuring optimal yields across a wide range of derivatives. By eliminating the need for transition metals entirely, this process drastically simplifies the downstream purification workflow and removes the risk of heavy metal contamination. The reaction proceeds efficiently under aerobic conditions at moderate temperatures, demonstrating remarkable robustness and operational simplicity that is highly desirable for industrial scale-up.

Mechanistic Insights into Acid-Catalyzed Dehydration Cyclization
The underlying mechanism of this transformation involves a classic acid-promoted dehydration followed by an intramolecular electrophilic aromatic substitution. Initially, the hydroxyl group of the starting alcohol is protonated by the acid catalyst, converting it into a superior leaving group (water). Upon loss of the water molecule, a stabilized carbocation intermediate is generated at the benzylic position. This electrophilic center is then attacked by the electron-rich nitrogen atom of the pyridine ring, initiating the ring-closing step to form the fused bicyclic indolizine system. Finally, deprotonation restores the aromaticity of the newly formed ring, yielding the stable 1-alkyl-3-aryl indolizine product. This mechanistic pathway is highly efficient because it leverages the inherent nucleophilicity of the pyridine nitrogen and the stability of the intermediate carbocation, avoiding the high energy barriers associated with metal-mediated bond formations.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for producing high-purity pharmaceutical intermediates. In traditional metal-catalyzed routes, side reactions often include homocoupling of the metal catalyst or incomplete conversion leading to metal-complexed byproducts that are difficult to separate. In this acid-catalyzed system, the primary byproducts are simply water and unreacted starting material, which are easily removed during the aqueous workup or chromatographic purification. The patent data indicates that varying the substituents on the aryl ring (R3) or the alkyl group (R2) does not significantly impede the reaction, suggesting a tolerant mechanism that accommodates both electron-donating and electron-withdrawing groups. This broad substrate tolerance ensures that the process can be applied to synthesize diverse libraries of indolizine derivatives without requiring extensive re-optimization for each new analog.
How to Synthesize 1-Alkyl-3-aryl Indolizines Efficiently
To implement this synthesis effectively, operators should select the appropriate acid system based on the lipophilicity of their specific substrate. For standard aryl substrates, the aqueous hydrochloric acid method is preferred due to its cost-effectiveness and ease of workup, whereas more hydrophobic substrates may benefit from the DMF/p-TsOH system to ensure homogeneous reaction conditions. The reaction typically requires heating to 100°C for approximately 12 hours to reach full conversion, after which standard extraction and purification techniques yield the target compound in high purity. Detailed standardized operating procedures regarding stoichiometry, specific temperature ramping rates, and safety protocols for handling concentrated acids are critical for successful replication and are outlined in the technical guide below.
- Prepare the reaction mixture by combining the pyridyl alcohol substrate with either 3.5 M hydrochloric acid aqueous solution or an organic solvent system containing p-TsOH catalyst.
- Heat the reaction mixture to 100°C under an air atmosphere and stir continuously for approximately 12 hours to facilitate the dehydration and cyclization process.
- Upon completion, neutralize the acidic mixture with base if using the aqueous method, extract with ethyl acetate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this acid-catalyzed technology represents a strategic opportunity to optimize the cost structure and reliability of the supply chain for nitrogen heterocycle intermediates. By shifting away from precious metal catalysis, manufacturers can decouple their production costs from the volatile market prices of rhodium, palladium, and copper. Furthermore, the simplified purification process reduces the consumption of specialized scavenging resins and silica gel, leading to substantial cost savings in consumables. The use of commodity chemicals like hydrochloric acid and p-toluenesulfonic acid ensures a stable and secure supply of catalysts, mitigating the risk of production delays caused by shortages of specialized reagents. This resilience is crucial for maintaining continuous manufacturing operations and meeting tight delivery schedules for downstream API clients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts directly translates to a significant reduction in raw material expenditures. Additionally, the removal of metal scavenging steps and the simplification of the purification workflow reduce labor hours and utility consumption. The ability to use water as a solvent in Method 1 further decreases solvent purchase and disposal costs, aligning with green chemistry principles that often correlate with lower operational expenses. These cumulative efficiencies allow for a more competitive pricing structure for the final indolizine intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Relying on widely available inorganic and organic acids rather than specialized metal complexes enhances the robustness of the supply chain. Commodity acids are produced in massive volumes globally, ensuring that supply disruptions are rare compared to the niche market for fine chemical catalysts. This reliability allows for better inventory planning and reduces the need for safety stockpiling of expensive catalysts. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, further stabilizing the production output and ensuring consistent on-time delivery to customers.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-ton commercial production. The aqueous method, in particular, minimizes the generation of hazardous organic waste, simplifying wastewater treatment and reducing the environmental compliance burden. The absence of heavy metals eliminates the need for complex effluent treatment processes required to meet strict regulatory limits on metal discharge. This environmental friendliness not only reduces disposal costs but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major pharmaceutical companies that prioritize green suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on reaction scope, purification, and scalability. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines and for procurement teams assessing the long-term viability of the supply source.
Q: What are the primary advantages of this acid-catalyzed method over traditional metal-catalyzed routes?
A: The primary advantage is the complete elimination of expensive and toxic transition metal catalysts such as rhodium, palladium, or copper. This significantly simplifies the purification process, reduces the risk of heavy metal contamination in the final API, and lowers the overall raw material costs associated with precious metal recovery.
Q: How does the choice between inorganic and organic acid methods affect substrate scope?
A: The patent demonstrates that the two methods are complementary. The inorganic acid aqueous method (Method 1) generally provides higher yields for standard aryl substrates, while the organic acid method (Method 2) in solvents like DMF offers better solubility and reactivity for more lipophilic or sterically hindered substrates, ensuring broad applicability across diverse chemical libraries.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is highly amenable to scale-up. It utilizes inexpensive, commodity-grade acids like hydrochloric acid or p-toluenesulfonic acid and operates under relatively mild thermal conditions (100°C). The absence of sensitive metal catalysts and the use of robust reaction conditions make it ideal for multi-kilogram to ton-scale manufacturing in standard glass-lined or stainless steel reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Alkyl-3-aryl Indolizines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this acid-catalyzed synthesis route for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of indolizine derivative meets the exacting standards required for drug substance manufacturing. We are committed to leveraging this innovative technology to deliver cost-effective and high-quality solutions to our global partners.
We invite you to collaborate with us to explore how this efficient synthesis method can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this metal-free route can impact your bottom line. Please contact us today to request specific COA data for our indolizine portfolio and to discuss detailed route feasibility assessments for your target molecules, ensuring a partnership built on transparency, quality, and mutual success.
