Advanced Catalytic Synthesis of Pregabalin Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust and scalable pathways for the production of high-value active pharmaceutical ingredients, and the synthesis of pregabalin remains a critical focus area for generic and innovator companies alike. Patent CN114478422A discloses a groundbreaking approach to preparing a key pregabalin intermediate, specifically a nitro-substituted oxazolidinone derivative, which serves as a pivotal chiral building block. This technology represents a significant departure from legacy methods by utilizing a chiral phase-transfer catalysis system or a base-mediated resolution strategy that avoids the severe safety and environmental liabilities associated with traditional routes. By leveraging mild reaction conditions and readily available starting materials, this innovation offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates. The strategic implementation of this synthetic route allows for precise control over stereochemistry, ensuring that the final API meets stringent regulatory standards for optical purity while minimizing the generation of hazardous waste streams.
The development of this intermediate addresses the long-standing challenges in pregabalin manufacturing, where the control of chirality often dictates the overall economic viability of the process. The patent outlines a method where an alpha,beta-unsaturated amide precursor reacts with nitromethane in the presence of a specialized chiral catalyst, facilitating a highly selective Michael addition. This reaction is not merely a chemical transformation but a strategic enabler for cost reduction in pharmaceutical intermediates manufacturing, as it simplifies the purification workflow and reduces the reliance on expensive chiral pool starting materials. For procurement and technical teams, understanding the nuances of this catalytic system is essential for evaluating potential suppliers who can deliver consistent quality at a commercial scale. The ability to produce this intermediate with high diastereomeric excess directly impacts the efficiency of the subsequent reduction and hydrolysis steps, thereby enhancing the overall throughput of the production facility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of pregabalin has been plagued by significant safety and efficiency bottlenecks inherent in conventional chemical resolution and chiral source synthesis methods. Traditional routes often rely on the use of potassium cyanide, a highly toxic reagent that necessitates rigorous safety protocols, specialized containment infrastructure, and complex waste treatment procedures to mitigate environmental hazards and personnel exposure risks. Furthermore, alternative pathways involving azide intermediates present severe explosion hazards, making them unsuitable for large-scale amplification without substantial capital investment in blast-proof facilities. These legacy methods also suffer from low overall yields, often discarding valuable isomers during resolution steps, which leads to substantial raw material waste and inflated production costs. The reliance on expensive chiral auxiliaries or catalysts that are not commercially available further exacerbates the economic burden, limiting the feasibility of these routes for competitive generic manufacturing. Consequently, supply chain managers face persistent challenges in securing reliable sources of pregabalin intermediates that do not carry the baggage of high operational risk and regulatory scrutiny associated with toxic reagents.
The Novel Approach
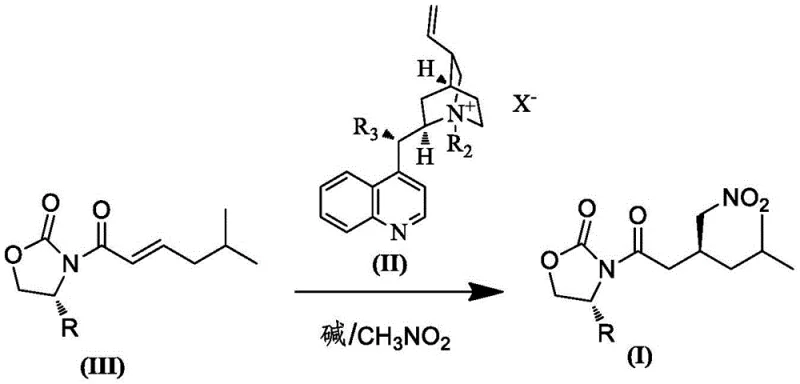
In stark contrast to these hazardous legacy processes, the novel approach disclosed in the patent utilizes a safe and efficient asymmetric Michael addition of nitromethane to an unsaturated amide substrate. This method operates under mild reaction conditions, typically ranging from 10°C to 40°C, which significantly reduces energy consumption and simplifies the thermal management requirements of the reactor system. By employing a chiral cinchona alkaloid derivative or a base-mediated resolution strategy, the process achieves high stereocontrol without the need for toxic cyanide or explosive azide reagents, thereby fundamentally altering the safety profile of the manufacturing operation. The use of common organic solvents such as toluene or dichloromethane, combined with inorganic bases like potassium carbonate or cesium carbonate, ensures that raw materials are easily sourced and cost-effective. This shift towards safer chemistry not only aligns with green chemistry principles but also provides a tangible competitive advantage by reducing the regulatory burden and insurance costs associated with handling hazardous materials. The result is a streamlined process that is inherently more suitable for commercial scale-up of complex pharmaceutical intermediates, offering a clear path to sustainable and economical production.
Mechanistic Insights into Chiral Cinchona Catalyst-Mediated Michael Addition
The core of this technological advancement lies in the precise mechanistic execution of the asymmetric Michael addition, where the chiral catalyst plays a pivotal role in directing the stereochemical outcome of the reaction. The catalyst, typically a quaternary ammonium salt derived from cinchona alkaloids, functions by activating the nitromethane nucleophile and organizing the transition state to favor the formation of the desired enantiomer. This interaction occurs within the organic phase, where the catalyst facilitates the transfer of the nitronate anion to the beta-position of the alpha,beta-unsaturated amide with high facial selectivity. The structural features of the catalyst, including the bulky quinoline and benzyl groups, create a chiral environment that sterically hinders the approach of the nucleophile from the undesired direction, thus ensuring high diastereoselectivity. Understanding this mechanism is crucial for R&D directors who need to validate the robustness of the process, as slight variations in catalyst loading or solvent choice can influence the reaction kinetics and selectivity. The patent data indicates that optimizing the molar ratio of the catalyst to the substrate is essential for balancing reaction rate and cost efficiency, highlighting the importance of fine-tuning process parameters to achieve optimal performance in a manufacturing setting.
Following the initial catalytic step, the control of impurities and the enhancement of optical purity are achieved through a sophisticated recrystallization protocol that leverages the solubility differences between diastereomers. The crude reaction product, which may contain a mixture of isomers, is subjected to sequential crystallization steps using solvents such as isopropanol or mixtures of alcohol and water. This purification strategy is designed to selectively precipitate the desired diastereomer while leaving impurities and the undesired isomer in the mother liquor, effectively upgrading the diastereomeric excess (de) value to over 99%. The temperature control during crystallization, typically cooling from 50-80°C down to 15-30°C, is critical for maximizing recovery and purity, as rapid cooling can lead to the inclusion of impurities within the crystal lattice. This focus on downstream processing ensures that the intermediate meets the stringent purity specifications required for subsequent conversion to the final API, reducing the burden on later purification stages. For quality assurance teams, this mechanism of impurity rejection provides confidence in the consistency of the product batch-to-batch, which is a key requirement for regulatory compliance and long-term supply agreements.
How to Synthesize Pregabalin Intermediate Efficiently
The practical implementation of this synthesis route involves a series of well-defined operational steps that begin with the preparation of the unsaturated amide precursor and culminate in the isolation of the high-purity nitro intermediate. The process is designed to be modular, allowing for the optimization of each stage independently to ensure maximum efficiency and yield. Operators must adhere to strict temperature controls during the addition of reagents to prevent exothermic runaways and maintain the integrity of the chiral catalyst. The reaction mixture is typically monitored using standard analytical techniques such as HPLC or TLC to determine the endpoint, ensuring that the conversion is complete before proceeding to workup. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this process with precision.
- Prepare the reaction system by suspending a base such as potassium carbonate or cesium carbonate in an organic solvent like toluene under controlled temperature conditions.
- Add the unsaturated amide precursor and nitromethane in the presence of a chiral cinchona alkaloid derivative catalyst to initiate the asymmetric Michael addition.
- Purify the resulting intermediate through sequential recrystallization using isopropanol to achieve high diastereomeric excess and optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits that directly address the pain points of procurement managers and supply chain heads regarding cost, reliability, and scalability. The elimination of toxic reagents like potassium cyanide and explosive azides removes the need for specialized hazardous waste disposal services and reduces the regulatory compliance costs associated with handling such materials. This simplification of the safety profile translates into lower operational overheads and reduced insurance premiums, contributing to significant cost savings in the overall manufacturing budget. Furthermore, the use of readily available and inexpensive starting materials ensures that the supply chain is resilient to market fluctuations and raw material shortages, providing a stable foundation for long-term production planning. The mild reaction conditions also mean that existing general-purpose chemical reactors can be utilized without the need for expensive retrofits or specialized equipment, accelerating the time to market for new production lines.
- Cost Reduction in Manufacturing: The process achieves cost optimization by eliminating the need for expensive transition metal catalysts and complex heavy metal removal steps that are often required in alternative synthetic pathways. By utilizing organic base catalysis and simple recrystallization for purification, the consumption of high-cost reagents and chromatography media is drastically reduced, leading to a leaner cost structure. The high selectivity of the reaction minimizes the formation of by-products, which in turn reduces the loss of valuable raw materials and improves the overall mass balance of the process. These factors combine to create a highly economical manufacturing route that allows suppliers to offer competitive pricing without compromising on quality or margin.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as nitromethane, potassium carbonate, and common organic solvents ensures that the supply chain is not dependent on single-source or geographically constrained raw materials. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream customers. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in utility supply or environmental conditions, further enhancing the reliability of the manufacturing operation. For supply chain heads, this translates to a more predictable and secure source of critical intermediates, reducing the need for excessive safety stock and inventory holding costs.
- Scalability and Environmental Compliance: The inherent safety and simplicity of this route make it ideally suited for scaling from pilot plant to full commercial production without the engineering bottlenecks often encountered with hazardous chemistries. The absence of toxic emissions and the generation of less hazardous waste streams simplify the environmental permitting process and reduce the burden on wastewater treatment facilities. This alignment with environmental, social, and governance (ESG) goals is increasingly important for pharmaceutical companies seeking to minimize their carbon footprint and regulatory risk. The process design facilitates easy scale-up of complex pharmaceutical intermediates, allowing manufacturers to respond quickly to market demand increases while maintaining compliance with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pregabalin intermediate synthesis technology. These answers are derived directly from the patent data and are intended to provide clarity on the process capabilities and limitations. Understanding these details is essential for making informed decisions about technology adoption and supplier selection. The responses cover aspects of safety, purity, and scalability to ensure a comprehensive understanding of the value proposition.
Q: How does this new method improve safety compared to traditional pregabalin synthesis?
A: This method eliminates the use of highly toxic potassium cyanide and explosive azide reagents, significantly reducing operational hazards and waste treatment complexity.
Q: What is the expected optical purity of the intermediate using this catalytic route?
A: Through optimized recrystallization processes, the diastereomeric excess (de) value can be elevated to over 99%, ensuring high-quality downstream processing.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction operates under mild temperature conditions (10-40°C) and uses readily available raw materials, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pregabalin Intermediate Supplier
The technical potential of this catalytic asymmetric synthesis route is immense, offering a pathway to high-quality pregabalin intermediates that meet the rigorous demands of the global pharmaceutical market. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of intermediate delivered meets the highest standards of quality and consistency. We understand the critical nature of chiral intermediates in API synthesis and are committed to providing a seamless transition from process development to commercial manufacturing. Our team of chemists and engineers is ready to collaborate with your R&D department to optimize this route for your specific production needs, ensuring that the theoretical benefits of the patent are fully realized in practice.
We invite you to initiate a dialogue with our technical procurement team to discuss how this innovative synthesis route can enhance your supply chain efficiency and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer and more efficient process. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Our goal is to establish a long-term partnership that drives value for your organization through technical excellence and reliable supply. Let us help you navigate the complexities of chemical manufacturing and secure a competitive advantage in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
