Revolutionizing Dinotefuran Production: A Novel One-Pot Catalytic Route for Global Agrochemical Supply Chains
The global demand for high-efficiency, low-toxicity neonicotinoid insecticides continues to surge, placing immense pressure on supply chains to deliver high-purity dinotefuran with consistent quality and competitive pricing. A significant technological breakthrough in this sector is documented in patent CN113214193B, which discloses a novel preparation method that fundamentally reimagines the synthetic architecture of this critical agrochemical active ingredient. Unlike legacy processes that rely on hazardous triflates or multi-step isourea condensations, this invention introduces a streamlined one-pot strategy utilizing 1,3-dicarbonyl compounds and tetrahydrofuran-3-methylamine. For R&D Directors and Procurement Managers alike, this represents a pivotal shift towards greener, more atom-economical manufacturing that drastically reduces waste generation while unlocking new avenues for cost optimization through valuable byproduct recovery.
This report provides a deep technical analysis of the patented methodology, evaluating its feasibility for commercial scale-up of complex agrochemical intermediates. By leveraging a dual catalytic system comprising acidic promoters and water-scavenging agents, the process achieves reaction efficiencies that far exceed the industry standard of less than 60% yield seen in older triflate-based routes. The ability to synthesize the target molecule directly from accessible precursors like acetylacetone or malondialdehyde not only simplifies the supply chain but also mitigates the risks associated with handling unstable intermediates. As we delve into the mechanistic nuances and operational parameters, it becomes clear that this technology offers a robust solution for reducing lead time for high-purity insecticides in a volatile global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of dinotefuran has been plagued by significant inefficiencies and environmental burdens inherent to traditional synthetic pathways. The most common prior art methods include the 3-tetrahydrofuryl methyl triflate route, the S-methyl-N-nitro-N'-phthaloyl isourea method, and the O-methyl-N-nitroisourea method. Among these, the triflate and phthaloyl isourea routes are particularly problematic for large-scale manufacturing due to their inherently low yields, which typically remain stubbornly below 60%. Such poor atom economy translates directly into inflated raw material costs and massive volumes of chemical waste that require expensive disposal protocols. Furthermore, the O-methyl-N-nitroisourea method, while more widely adopted, suffers from an excessively long synthetic sequence that necessitates the use of large quantities of nitrogen-containing reagents. This not only increases the complexity of the process control but also results in severe pollution issues, making it increasingly difficult to comply with tightening environmental regulations in major chemical manufacturing hubs. The reliance on harsh reagents and multi-step isolation procedures in these conventional methods creates bottlenecks that hinder the ability to respond rapidly to market demand fluctuations.
The Novel Approach
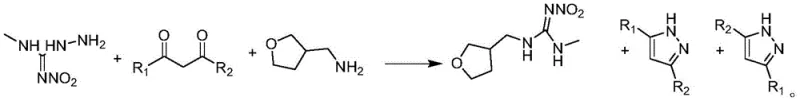
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in patent CN113214193B establishes a brand-new synthesis route that dramatically shortens the production timeline and enhances overall efficiency. This method employs a convergent one-pot strategy where Compound I (a nitroguanidine derivative), Compound II (a 1,3-dicarbonyl compound), and tetrahydrofuran-3-methylamine are reacted together under controlled thermal conditions. By eliminating the need for pre-activation steps like triflation or complex protecting group manipulations, the process reduces the number of unit operations and minimizes solvent consumption. The core innovation lies in the direct condensation of these three components in the presence of a specialized catalyst system, which facilitates the rapid formation of the dinotefuran skeleton. This streamlined workflow not only improves the throughput of the manufacturing facility but also significantly lowers the energy footprint per kilogram of product. Moreover, the reaction is designed such that the byproducts formed are not toxic wastes but rather valuable pyrazole derivatives, transforming a cost center into a potential profit center and offering a compelling value proposition for cost reduction in agrochemical manufacturing.
Mechanistic Insights into Dual-Catalytic One-Pot Condensation
The success of this novel synthesis hinges on a sophisticated understanding of reaction kinetics and equilibrium dynamics, specifically managed through a dual-catalyst system. The reaction mechanism involves the initial activation of the carbonyl groups in Compound II by an acid catalyst, such as formic acid, acetic acid, or p-toluenesulfonic acid. This protonation increases the electrophilicity of the carbonyl carbon, making it more susceptible to nucleophilic attack by the amino groups of Compound I. However, condensation reactions of this nature produce water as a byproduct, which can drive the equilibrium backward and hydrolyze sensitive intermediates. To counteract this, the process incorporates a water-absorbing catalyst, such as molecular sieves, silica gel, or anhydrous sodium sulfate. This desiccant effectively sequesters the generated water in situ, shifting the equilibrium position strongly towards the product side according to Le Chatelier's principle. The synergy between the acid promoter and the dehydrating agent is critical; experimental data within the patent indicates that using either catalyst alone results in suboptimal yields, whereas their combination pushes conversion rates to near-quantitative levels. This mechanistic elegance ensures that the reaction proceeds smoothly even with less reactive substrates, providing a robust platform for synthesizing various analogues by simply varying the R groups on the 1,3-dicarbonyl starting material.
Furthermore, the process exhibits exceptional selectivity, which is paramount for maintaining a clean impurity profile in the final agrochemical intermediate. The reaction conditions are tuned to favor the formation of the desired guanidine scaffold while simultaneously cyclizing the excess 1,3-dicarbonyl compound into pyrazole derivatives. This side reaction, rather than being a nuisance, serves as a self-purification mechanism. The pyrazole byproducts possess different solubility characteristics compared to dinotefuran, allowing for easy separation during the workup phase. Specifically, upon cooling the reaction mixture to 0-20°C, the target dinotefuran crystallizes out as a solid, while the pyrazole derivatives remain largely in the filtrate or can be washed away. This intrinsic difference in physical properties simplifies the downstream purification process, reducing the need for complex chromatography or recrystallization steps that often erode yield. The ability to isolate high-purity product (>98%) directly from the reaction mixture underscores the precision of this catalytic design, offering R&D teams a reliable pathway to generate reference standards and commercial batches with minimal impurity burden.
How to Synthesize Dinotefuran Efficiently
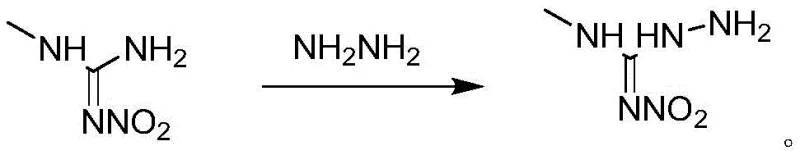
The implementation of this synthesis route requires precise control over stoichiometry, temperature, and catalyst loading to maximize the economic and technical benefits described above. The process begins with the preparation of the key intermediate, Compound I, via the reaction of 1-methyl-3-nitroguanidine with hydrazine hydrate, followed by a carefully managed acid quench to ensure high purity before entering the main reactor. Once Compound I is secured, the one-pot condensation is initiated by combining it with the chosen 1,3-dicarbonyl compound and the dual catalyst system in a non-alcoholic solvent like toluene. The mixture is heated to promote the initial condensation, after which tetrahydrofuran-3-methylamine is introduced to complete the molecular assembly. The subsequent cooling and crystallization steps are critical for defining the particle size and purity of the final product. For a comprehensive guide on the exact operational parameters, including specific molar ratios and cooling rates, please refer to the standardized protocol below.
- Prepare Compound I by reacting 1-methyl-3-nitroguanidine with hydrazine hydrate in aqueous solution, followed by acid quenching and crystallization.
- Conduct a one-pot reaction by heating Compound I, a 1,3-dicarbonyl compound (Compound II), and an acid/water-absorption catalyst system in a non-alcoholic solvent.
- Add tetrahydrofuran-3-methylamine to the mixture, maintain reflux, then cool the solution to precipitate dinotefuran while separating valuable pyrazole byproducts from the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For Procurement Managers and Supply Chain Heads, the adoption of this novel dinotefuran synthesis method offers transformative advantages that extend far beyond simple yield improvements. The primary value driver is the drastic simplification of the supply chain architecture. By replacing exotic, hazard-prone reagents like triflates with commodity chemicals such as acetylacetone and hydrazine, the process significantly de-risks the sourcing strategy. These raw materials are produced on a massive global scale, ensuring stable availability and shielding the manufacturing operation from the volatility often associated with specialty reagent markets. This shift to ubiquitous feedstocks enhances supply continuity, allowing production planners to forecast with greater confidence and reduce the safety stock levels required for critical inputs. Additionally, the elimination of multiple isolation and purification stages reduces the total cycle time from raw material intake to finished goods, thereby improving inventory turnover rates and freeing up working capital.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven principally by the enhancement in atom economy and the valorization of byproducts. Traditional methods discard significant portions of the starting mass as waste, whereas this one-pot approach incorporates the majority of the reactant atoms into either the target product or a saleable co-product. The generation of pyrazole derivatives, which possess their own market value in pharmaceutical and agrochemical applications, creates an ancillary revenue stream that effectively subsidizes the cost of dinotefuran production. Furthermore, the reduction in solvent usage and the avoidance of expensive heavy metal catalysts or complex protecting groups lower the variable cost per kilogram substantially. The simplified workup procedure also translates to reduced labor hours and lower utility consumption for distillation and drying, contributing to a leaner, more competitive cost structure without compromising on quality.
- Enhanced Supply Chain Reliability: Resilience is a key metric for modern supply chains, and this synthesis route bolsters reliability by minimizing dependency on single-source or geographically constrained reagents. The robustness of the dual-catalyst system means that the process is less sensitive to minor fluctuations in raw material quality, reducing the frequency of batch failures and reworks. The ability to operate in common solvents like toluene, which are easily recovered and recycled, further insulates the operation from solvent supply disruptions. Moreover, the shorter synthetic route reduces the number of handovers between different processing units or external contractors, thereby minimizing the logistical friction and potential for delays. This streamlined flow ensures that orders can be fulfilled with greater predictability, strengthening relationships with downstream formulators who depend on just-in-time delivery schedules.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this method represents a significant leap forward in sustainable manufacturing. The avoidance of chlorinated reagents and the reduction in nitrogenous waste streams align perfectly with increasingly stringent global environmental regulations. The process generates less hazardous waste, lowering disposal costs and reducing the regulatory burden on the manufacturing site. Scalability is inherently supported by the one-pot nature of the reaction, which reduces the equipment footprint required for production. Fewer reactors and separation units are needed to achieve the same output volume, allowing for capacity expansion without proportional increases in capital expenditure. This scalability ensures that the technology can meet surging market demand for neonicotinoids while maintaining a low environmental impact, a crucial factor for maintaining social license to operate in the chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented dinotefuran synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or licensing.
Q: How does the new dinotefuran synthesis method improve upon traditional triflate or isourea routes?
A: Traditional methods often suffer from yields below 60% or require long, polluting routes with excessive nitrogen reagents. This novel one-pot approach utilizes readily available 1,3-dicarbonyl compounds to achieve significantly higher yields (exceeding 90% in optimized examples) while simplifying the workflow.
Q: What is the economic benefit of the pyrazole byproduct generated in this process?
A: Unlike previous methods where byproducts were waste, this reaction generates pyrazole or its derivatives as a co-product. These compounds have independent economic value and can be easily separated from the filtrate, creating an additional revenue stream that offsets production costs.
Q: Why is a dual catalyst system of acid and water absorbent critical for this reaction?
A: The reaction equilibrium is driven forward by the removal of water. Using a combination of an acid catalyst (like formic acid) and a water-absorbing agent (like molecular sieves) synergistically accelerates the condensation rate and pushes the conversion to completion, resulting in superior purity and yield compared to using either catalyst alone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dinotefuran Supplier
As the agrochemical landscape evolves, the need for innovative, efficient, and sustainable manufacturing processes has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in CN113214193B to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in a GMP-compliant industrial setting. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of dinotefuran meets the exacting standards required for modern crop protection formulations. Our infrastructure is designed to handle complex chemistries with precision, offering a seamless bridge between laboratory innovation and commercial reality.
We invite you to explore how this cutting-edge synthesis route can optimize your supply chain and enhance your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how partnering with NINGBO INNO PHARMCHEM can drive efficiency and profitability in your agrochemical operations. Let us collaborate to build a more resilient and sustainable future for global agriculture.
