Scalable Synthesis of Cefepime Hydrochloride: A Technical Breakthrough for Global Supply Chains
Scalable Synthesis of Cefepime Hydrochloride: A Technical Breakthrough for Global Supply Chains
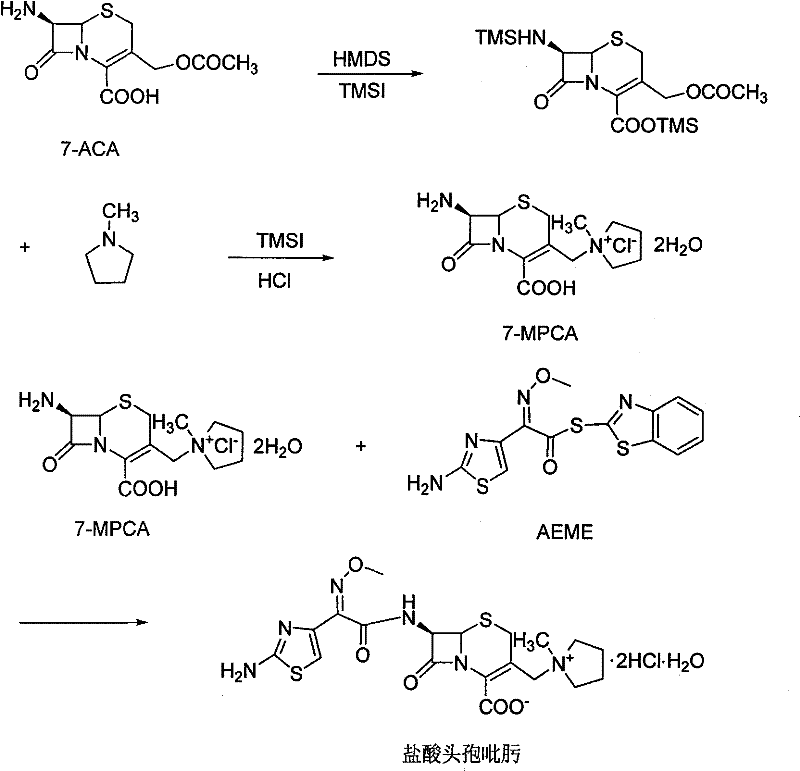
The pharmaceutical landscape for fourth-generation cephalosporins is constantly evolving, driven by the need for more efficient, cost-effective, and high-purity manufacturing processes. Patent CN101337971B introduces a robust synthetic method for Cefepime Hydrochloride, a critical broad-spectrum antibiotic, addressing longstanding challenges in stability and scalability. This technology leverages 7-aminocephalosporanic acid (7-ACA) as the foundational starting material, utilizing a sophisticated sequence of silylation protection and quaternization to achieve superior yields. For R&D directors and supply chain leaders, this patented route represents a significant departure from traditional methods that often rely on expensive or difficult-to-source precursors like GCLE. By optimizing reaction conditions and introducing novel purification techniques, this method ensures a stable supply of high-quality API intermediates essential for meeting global healthcare demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefepime has been plagued by significant technical and economic hurdles that hinder efficient commercialization. Traditional routes often depend on starting materials such as 7-phenylacetamide-3-chloromethyl-4-cephalosporanic acid methoxybenzyl ester (GCLE), which are not only costly but also present substantial procurement challenges due to limited supplier availability. Furthermore, existing methodologies frequently necessitate severe reaction conditions, including extreme temperatures and prolonged reaction times, which complicate process control and increase energy consumption. These factors collectively contribute to higher production costs and inconsistent product quality, particularly regarding the stability and color of the final hydrochloride salt. The difficulty in achieving continuous, large-scale production with these legacy methods creates bottlenecks for pharmaceutical manufacturers aiming to secure reliable antibiotic supplies.
The Novel Approach
In stark contrast, the methodology outlined in CN101337971B offers a streamlined, one-pot strategy that significantly mitigates these operational complexities. By initiating the synthesis with the widely available 7-ACA, the process bypasses the need for exotic precursors, immediately lowering the barrier to entry for production. The core innovation lies in the sequential protection of amino and carboxyl groups followed by a direct quaternization to form the key intermediate, 7-MPCA. This approach not only simplifies the operational workflow but also enhances the overall yield and stability of the product. The integration of mild reaction conditions and efficient solvent systems allows for a more manageable industrial process, ensuring that the final Cefepime Hydrochloride meets stringent purity specifications required for injectable formulations.
Mechanistic Insights into Silylation Protection and Quaternization
The chemical elegance of this synthesis is rooted in the precise manipulation of the cephalosporin nucleus through silylation and subsequent nucleophilic substitution. Initially, 7-ACA undergoes protection using hexamethyldisilazane (HMDS) and iodotrimethylsilane (TMSI), which effectively masks the reactive amino and carboxyl functionalities. This protection is crucial as it prevents unwanted side reactions during the subsequent quaternization step at the C-3 position. The introduction of the N-methylpyrrolidine moiety is achieved through a reaction with TMSI, generating the quaternary ammonium salt 7-MPCA in situ. This one-pot transformation is kinetically favorable and minimizes the formation of by-products, thereby preserving the integrity of the beta-lactam ring which is sensitive to harsh conditions. The mechanistic pathway ensures high regioselectivity, a critical factor for maintaining the biological activity of the final antibiotic.
Furthermore, the purification mechanism employed in this patent addresses the persistent issue of product discoloration, a common defect in cephalosporin synthesis. Following the acylation reaction with the AE active ester, the crude mixture undergoes a dual-stage purification process involving activated carbon decolorization followed by gamma-alumina column chromatography. The gamma-alumina acts as a selective adsorbent, effectively trapping trace impurities and colored by-products such as mercaptobenzothiazole that are not removed by standard extraction methods. This step is vital for achieving the high visual and chemical purity required for parenteral drugs. Additionally, the process allows for the recycling of solvents like acetone and dichloromethane through rectification, aligning the chemical mechanism with principles of green chemistry and waste reduction.
How to Synthesize Cefepime Hydrochloride Efficiently
The practical implementation of this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the dissolution of 7-ACA in a mixed organic solvent system, typically comprising acetonitrile and tetrahydrofuran, followed by the addition of silylating agents under reflux conditions. Once the protected intermediate is formed, the temperature is carefully lowered to facilitate the addition of iodotrimethylsilane and N-methylpyrrolidine, driving the formation of 7-MPCA. The subsequent acylation with AE active ester must be conducted under controlled pH and temperature conditions to prevent hydrolysis of the sensitive beta-lactam ring. For a comprehensive understanding of the specific reagent ratios, temperature profiles, and work-up procedures, please refer to the detailed technical guide below.
- Protect 7-ACA amino and carboxyl groups using HMDS and TMSI in organic solvent.
- React protected 7-ACA with TMSI and N-methylpyrrolidine to synthesize 7-MPCA via one-pot quaternization.
- Perform acylation with AE active ester followed by salt formation and gamma-alumina purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthetic route offers compelling advantages that directly impact the bottom line and supply chain resilience. By shifting the starting material base to 7-ACA, manufacturers can leverage a well-established global supply chain for bulk antibiotics, reducing dependency on niche intermediates like GCLE that are prone to price volatility and supply disruptions. The simplification of the reaction sequence into fewer steps inherently reduces the consumption of utilities and labor hours, leading to a more cost-efficient manufacturing footprint. Moreover, the ability to recycle solvents and regenerate purification media such as gamma-alumina further drives down the variable costs associated with production, making the final API more competitive in the global market without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive and hard-to-obtain starting materials like GCLE significantly lowers the raw material input costs. Additionally, the one-pot synthesis of the 7-MPCA intermediate reduces the number of isolation and purification steps required, which translates to lower solvent usage and reduced waste disposal costs. The process design inherently supports high throughput, allowing for better asset utilization and economies of scale that are critical for maintaining margin stability in the generic antibiotic sector.
- Enhanced Supply Chain Reliability: Utilizing 7-ACA as the primary feedstock ensures a more robust supply chain, as this material is produced in massive quantities globally for various cephalosporin derivatives. This abundance mitigates the risk of raw material shortages that can plague specialized intermediate markets. Furthermore, the simplified process conditions reduce the likelihood of batch failures due to operational complexity, ensuring a consistent and reliable output of finished goods that can meet tight delivery schedules for downstream formulation partners.
- Scalability and Environmental Compliance: The method is explicitly designed for large-scale industrial production, featuring straightforward unit operations that are easily transferable from pilot plant to commercial manufacturing scales. The inclusion of solvent recovery systems and the use of regenerable purification columns align with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste generation. This compliance readiness future-proofs the manufacturing site against regulatory changes and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology, we have compiled answers to common inquiries regarding the process specifics and quality outcomes. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the method's capabilities. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: What are the key advantages of the 7-ACA route over GCLE for Cefepime?
A: The 7-ACA route utilizes more accessible starting materials compared to GCLE, avoids severe reaction conditions, and simplifies the purification process through novel alumina column chromatography, ensuring better color and stability.
Q: How is product color and purity controlled in this synthesis?
A: The process employs a specific refinement step where the cefepime solution passes through a gamma-alumina column chromatography after activated carbon decolorization, effectively removing impurities and solving color issues common in traditional methods.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights simple processing conditions, convenient operation, and high product yield, making it highly suitable for continuity and large-scale industrialized production of antibiotic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to implement complex chemistries, such as the silylation and quaternization steps described in this patent, positions us as a strategic partner for your long-term API sourcing needs.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic impacts of switching to this more efficient route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your supply chain is built on a foundation of quality, reliability, and scientific excellence.
