Advanced Industrial Synthesis of GCLE: A Strategic Route for High-Purity Cephalosporin Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for key beta-lactam intermediates that balance high purity with economic viability. Patent CN101260116A introduces a refined methodology for synthesizing 7-phenylacetamide-3-chloromethyl-4-cephalosporanic acid p-methoxybenzyl ester, commonly known as GCLE, which serves as a critical building block for third and fourth-generation cephalosporins. This technology addresses the longstanding challenges associated with traditional GCLE production by utilizing Penicillin G potassium salts as the foundational starting material, a commodity chemical with established global supply chains. The disclosed process integrates a sequence of esterification, oxidation, ring expansion, and chlorination steps that collectively enhance the overall yield and simplify the operational complexity. By shifting the synthetic strategy to prioritize the stability of the penicillin ester prior to oxidation, the inventors have created a route that minimizes degradation and maximizes the efficiency of the subsequent ring-expansion reactions. This innovation represents a significant leap forward for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of GCLE has been plagued by reliance on expensive halogenating agents and complex reaction sequences that hinder cost reduction in pharmaceutical intermediate manufacturing. Traditional routes often employ N-halogenated succinimides such as N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS) for the critical ring-expansion step, which not only increases raw material costs but also introduces difficult-to-remove byproducts that complicate purification. Furthermore, conventional methods frequently struggle with the stability of the penicillin nucleus during oxidation, leading to lower yields and inconsistent impurity profiles that require extensive downstream processing. The use of less stable acid forms of penicillin in early stages often results in premature degradation, forcing manufacturers to operate with narrow safety margins and increased waste generation. These inefficiencies create substantial bottlenecks in the supply chain, making it challenging to meet the rigorous demand for high-purity cephalosporin intermediates required by regulatory agencies.
The Novel Approach
In stark contrast, the methodology outlined in CN101260116A revolutionizes the production landscape by implementing a strategic esterification-first protocol that stabilizes the beta-lactam core before subjecting it to oxidative stress. This novel approach utilizes p-methoxybenzyl chloride to convert Penicillin G potassium salts directly into the corresponding ester, which exhibits superior stability compared to the free acid form. Following esterification, the process employs peracetic acid for oxidation, a reagent that offers excellent atom economy and ease of handling compared to heavier metal oxidants. The subsequent ring expansion leverages a unique combination of 2-mercaptobenzothiazole and benzenesulfinic acid, effectively bypassing the need for costly N-halogenated reagents while ensuring a clean conversion to the open-loop cephalosporin structure. Finally, the direct feeding of chlorine gas for chlorination provides precise control over the reaction stoichiometry, eliminating the variability associated with solid chlorinating agents. 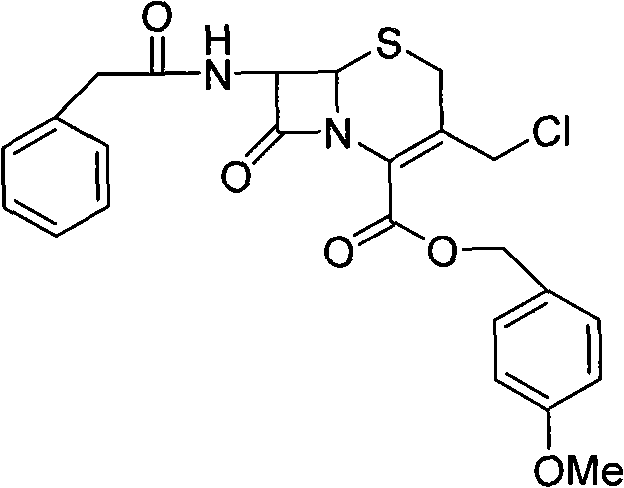
Mechanistic Insights into Ring Expansion and Chlorination
The core chemical innovation of this patent lies in the mechanistic elegance of the ring expansion step, where the penicillin sulfoxide ester is transformed into the cephalosporin framework. The reaction initiates with the nucleophilic attack of 2-mercaptobenzothiazole on the sulfoxide species, facilitating the opening of the five-membered thiazolidine ring to form a reactive intermediate. This intermediate is then treated with benzenesulfinic acid, which acts as a mild yet effective promoter for the rearrangement that constructs the six-membered dihydrothiazine ring characteristic of cephalosporins. This specific reagent combination avoids the formation of harsh acidic byproducts that could otherwise hydrolyze the sensitive beta-lactam bond, thereby preserving the integrity of the molecular scaffold. The careful control of solvent systems, utilizing mixtures of toluene, acetone, and alcohols, ensures that the intermediates remain in solution long enough to react completely without precipitating prematurely or decomposing.
Following the ring expansion, the chlorination mechanism is executed with high precision through the direct introduction of elemental chlorine gas into the reaction mixture. This step targets the methyl group at the 3-position of the cephalosporin nucleus, converting it into a chloromethyl group which is essential for subsequent side-chain modifications in drug synthesis. The use of gaseous chlorine allows for real-time monitoring of the reaction progress, enabling operators to halt the feed exactly when the desired conversion is reached, thus preventing over-chlorination or degradation of the sensitive ester moiety. The final cyclization with ammonia gas closes the loop, restoring the double bond in the dihydrothiazine ring and yielding the final GCLE product with exceptional structural fidelity. This meticulous control over each mechanistic step results in a final product with HPLC purity exceeding 96%, demonstrating the robustness of the chemical design against impurity formation.
How to Synthesize GCLE Efficiently
Implementing this synthesis route requires strict adherence to the specified temperature gradients and solvent ratios to ensure optimal yield and safety. The process begins with the esterification of Penicillin G potassium salts in a mixed solvent system of DMF and water, followed by extraction into toluene to isolate the stable ester intermediate. Subsequent oxidation with peracetic acid must be maintained at low temperatures between 10°C and 15°C to prevent thermal runaway, followed by a quenching step using sodium sulfite to neutralize excess oxidant. The ring expansion phase involves refluxing with 2-mercaptobenzothiazole in toluene, followed by treatment with benzenesulfinic acid in acetone, requiring careful solvent swaps to maintain reaction homogeneity. For the detailed standardized synthesis steps including exact reagent quantities and workup procedures, please refer to the guide below.
- Esterify Penicillin G potassium salts with p-methoxybenzyl chloride in a mixed solvent system to form the stable penicillin ester.
- Oxidize the resulting ester using peracetic acid at controlled low temperatures to generate the penicillin sulfoxide ester intermediate.
- Perform ring expansion by reacting the sulfoxide with 2-mercaptobenzothiazole followed by benzenesulfinic acid to obtain the open-loop product.
- Chlorinate the open-loop product with chlorine gas and subsequently cyclize with ammonia gas to finalize the GCLE structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers transformative benefits for procurement managers and supply chain directors seeking to optimize their sourcing strategies for beta-lactam intermediates. By anchoring the synthesis on Penicillin G potassium salts, a globally traded commodity, the method decouples production from the volatility of specialized niche reagents, ensuring a steady and predictable flow of raw materials. The elimination of expensive N-halogenated succinimides in favor of direct chlorine gas and benzenesulfinic acid drastically reduces the variable cost per kilogram of the final product, allowing for more competitive pricing structures in the marketplace. Additionally, the extensive use of recoverable solvents such as toluene, methanol, and DMF means that waste disposal costs are significantly minimized, contributing to a more sustainable and economically efficient operation. These factors combine to create a supply model that is resilient against market fluctuations and capable of supporting long-term contractual agreements with major pharmaceutical buyers.
- Cost Reduction in Manufacturing: The substitution of high-cost halogenating agents with elemental chlorine gas and the use of bulk industrial Penicillin G salts fundamentally alters the cost structure of GCLE production. This shift removes the premium associated with specialized reagents and simplifies the inventory management required for hazardous materials, leading to substantial operational savings. Furthermore, the high yields reported in the patent examples indicate that less raw material is wasted per unit of output, directly improving the gross margin potential for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: Utilizing starting materials that are produced on a massive industrial scale ensures that supply disruptions are rare and easily mitigated through multiple sourcing channels. The robustness of the ester intermediate allows for potential storage or transport between stages if necessary, adding flexibility to the production schedule. This reliability is crucial for maintaining continuous manufacturing lines for downstream cephalosporin antibiotics, preventing costly downtime due to intermediate shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like extraction, distillation, and crystallization that are familiar to chemical engineers. The ability to recycle solvents through rectification not only lowers costs but also aligns with increasingly stringent environmental regulations regarding volatile organic compound emissions. The absence of heavy metal catalysts or persistent organic pollutants in the waste stream simplifies effluent treatment, making it easier to obtain and maintain the necessary environmental permits for large-scale production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this GCLE synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of using Penicillin G potassium salts as the starting material for GCLE?
A: Using Penicillin G potassium salts allows manufacturers to leverage widely available, cost-effective bulk raw materials rather than relying on more expensive or specialized precursors. This route significantly simplifies the supply chain logistics and reduces the overall raw material cost basis for the final cephalosporin intermediate.
Q: How does the novel ring expansion method improve impurity profiles compared to traditional N-halogenated reagents?
A: The patented method utilizes 2-mercaptobenzothiazole and benzenesulfinic acid for ring expansion, avoiding the use of N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS). This substitution eliminates bromine or chlorine residues associated with succinimide byproducts, leading to a cleaner reaction profile and facilitating easier downstream purification to achieve high HPLC purity.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for industrial scalability, utilizing common organic solvents like toluene, DMF, and methanol that can be recovered and recycled via rectification. The operational conditions, such as temperature controls between -10°C and 100°C, are manageable in standard chemical reactors, ensuring consistent quality and yield at multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GCLE Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving antibiotics, and we are uniquely positioned to support your production needs with our advanced capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of GCLE meets the highest international standards. Our commitment to excellence extends beyond mere compliance, as we actively collaborate with clients to refine processes for maximum efficiency and sustainability.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis routes can reduce your overall expenditure without compromising on quality. We encourage potential partners to contact us directly to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics and reliable supply assurances.
