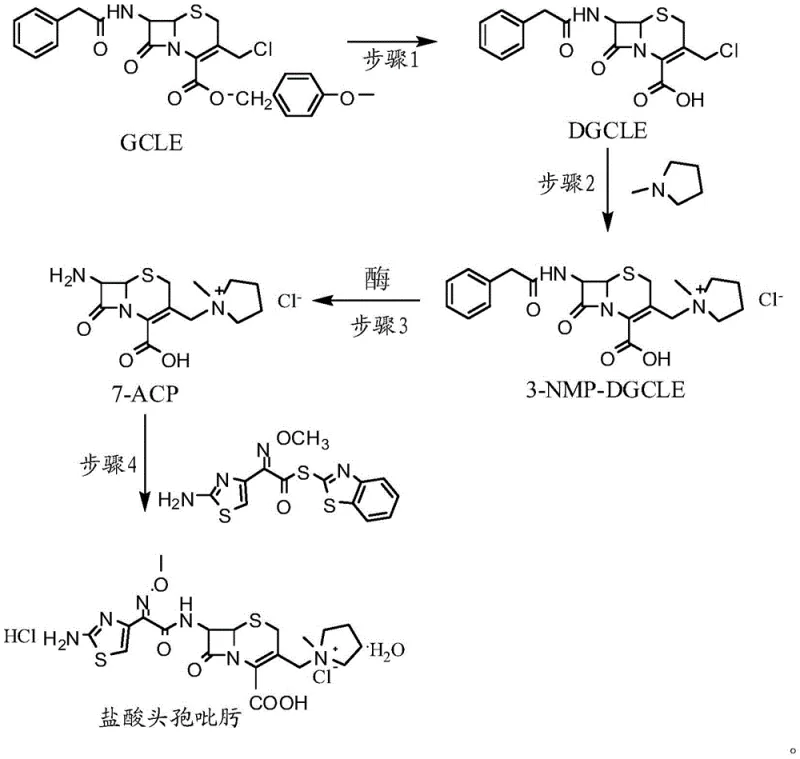
Advanced GCLE-Based Synthesis of Cefepime Hydrochloride for Commercial Scale Production
The pharmaceutical industry continuously seeks robust, scalable, and environmentally compliant pathways for the production of fourth-generation cephalosporins, among which Cefepime Hydrochloride stands out due to its broad antibacterial spectrum and stability against lactamases. A pivotal advancement in this domain is detailed in patent CN107201391B, which discloses a novel synthesis method utilizing GCLE (7-phenylacetamido-3-chloromethyl-3-cephem-4-carboxylic acid p-methoxybenzyl ester) as the primary raw material. This technical breakthrough addresses long-standing challenges associated with isomerism and solvent toxicity that have plagued previous manufacturing protocols. By strategically employing a sequence of acid-mediated deprotection, quaternization, enzymatic hydrolysis, and final acylation, this route achieves a high-purity end product with a molar yield exceeding 60 percent based on GCLE. For R&D directors and procurement specialists, understanding this pathway is critical as it represents a shift towards greener chemistry without compromising the stringent quality specifications required for injectable antibiotics. The method effectively bypasses the need for hazardous polyhalogenated solvents while ensuring the structural integrity of the beta-lactam ring throughout the transformation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefepime key intermediates has been fraught with significant technical and environmental hurdles, particularly concerning the control of 2,3-position isomerism during the introduction of side chains. Prominent prior art, such as the methods reported by Walker et al., relied heavily on 7-ACA as a starting material and utilized Freon TF, a fluorine-containing polychlorinated hydrocarbon, as the reaction solvent. While the low solubility of the product in Freon helped precipitate the compound and reduce isomerization, the reliance on such solvents is now strictly prohibited due to severe environmental regulations regarding ozone depletion and toxicity. Similarly, approaches by Naito et al. employed carbon tetrachloride (CCl4) for quaternization, which poses analogous ecological and safety risks. Furthermore, these conventional routes often necessitated complex activation steps, such as the use of expensive iodine reagents to activate the chloromethyl group, which not only inflated the raw material costs but also introduced additional purification burdens to remove residual heavy metals or halogens from the final API. The cumulative effect of these limitations resulted in lower overall yields, typically ranging between 30 to 40 percent in early steps, and a supply chain vulnerable to regulatory shifts regarding solvent usage.
The Novel Approach
In stark contrast to the legacy processes, the innovative route described in the patent leverages GCLE to streamline the synthesis into a more efficient and sustainable four-step sequence. This methodology ingeniously exploits the physicochemical properties of inner salt compounds to control impurity profiles without resorting to banned solvents. The process initiates with the removal of the 4-position p-methoxybenzyl protecting group under mild acidic conditions, followed by a direct quaternization reaction with N-methylpyrrolidine (NMP) in common organic solvents like dichloromethane or ethyl acetate. Crucially, the intermediate formed exists as an inner salt with extremely low solubility, causing it to precipitate rapidly from the reaction mixture. This spontaneous precipitation acts as a driving force that kinetically traps the desired 3-position isomer, fundamentally suppressing the formation of the unwanted Delta-2 isomer. By eliminating the iodine activation step and replacing toxic halogenated solvents with standard, recoverable organic media, this approach offers a compelling value proposition for cost reduction in pharmaceutical manufacturing while delivering a product with purity levels surpassing 99 percent.
Mechanistic Insights into Enzymatic Deprotection and Quaternization
The core of this synthesis lies in the precise manipulation of the cephem nucleus, particularly during the quaternization and enzymatic deprotection stages which dictate the final stereochemical purity. In the second step, N-methylpyrrolidine acts as a nucleophile attacking the chloromethyl group at the 3-position of the cephem ring. Unlike traditional methods where basic conditions might catalyze the rearrangement of the double bond from the 3-position to the 2-position (forming the Delta-2 isomer), the rapid formation and precipitation of the quaternary ammonium inner salt in this novel route effectively removes the product from the solution phase. This physical separation prevents the base-catalyzed isomerization equilibrium from establishing, thereby locking the molecular structure in the biologically active Delta-3 configuration. Following this, the third step employs immobilized penicillin acylase to cleave the 7-phenylacetamido protecting group. This biocatalytic approach is highly specific, operating efficiently at a controlled pH of 7.9 to 8.1 and moderate temperatures around 28°C. The enzyme's specificity ensures that the sensitive beta-lactam ring remains intact while the amide bond is hydrolyzed, a selectivity that is difficult to achieve with harsh chemical hydrolysis agents which often degrade the core structure or generate complex byproduct mixtures.
Furthermore, the impurity control mechanism is deeply integrated into the workup procedures of each step, ensuring that the final API meets rigorous pharmacopeial standards. The precipitation of the 3-NMP-DGCLE intermediate is not merely a isolation technique but a critical purification event; by adding anti-solvents such as diethyl ether or n-hexane, the process forces the crystallization of the desired inner salt while leaving soluble impurities in the mother liquor. Similarly, in the enzymatic step, the use of activated carbon for decolorization post-filtration removes trace organic impurities and enzyme residues before the final acidification. The final acylation with the AE-active ester (mercaptobenzothiazole active ester of aminothiazoles) is conducted under controlled conditions where the molar ratio is tightly managed to prevent over-acylation or hydrolysis of the active ester. The final product is isolated by adjusting the pH to a highly acidic range (1.15 to 1.35) and inducing crystallization with solvents like acetone or methanol, which ensures the removal of any remaining unreacted 7-ACP or side-chain acids, resulting in a high-purity Cefepime Hydrochloride suitable for parenteral administration.
How to Synthesize Cefepime Hydrochloride Efficiently
The synthesis of Cefepime Hydrochloride via this GCLE-based route requires careful attention to temperature control and stoichiometry to maximize yield and minimize isomer formation. The process is divided into four distinct operational units: deprotection, quaternization, enzymatic hydrolysis, and acylation. Each step builds upon the purity of the previous intermediate, emphasizing the importance of robust isolation techniques such as precipitation and filtration. For process chemists looking to implement this technology, the key lies in maintaining the low temperature (-30 to -20°C) during the initial chemical transformations to suppress side reactions, followed by precise pH control during the biocatalytic step to ensure optimal enzyme activity. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for laboratory validation and subsequent pilot scale-up.
- Deprotect the 4-position p-methoxybenzyl group of GCLE using organic acid at low temperature (-30 to -20°C) to obtain DGCLE.
- React DGCLE with N-methylpyrrolidine (NMP) in organic solvent at low temperature to form the quaternary ammonium salt 3-NMP-DGCLE.
- Perform enzymatic hydrolysis using immobilized penicillin acylase in water at pH 7.9-8.1 to remove the 7-position protecting group, yielding 7-ACP.
- Conduct 7-position acylation of 7-ACP with AE-active ester in organic solvent followed by acid precipitation to isolate Cefepime Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, this synthesis route offers transformative advantages that directly impact the bottom line and operational resilience. The elimination of expensive and specialized reagents, such as iodine activators and fluorinated solvents, drastically simplifies the raw material procurement landscape. Sourcing teams can rely on commodity chemicals like dichloromethane, ethyl acetate, and N-methylpyrrolidine, which are widely available in the global market with stable pricing structures, unlike niche reagents that are subject to volatile supply shocks. Moreover, the avoidance of environmentally prohibited substances like Freon and carbon tetrachloride future-proofs the manufacturing process against tightening environmental regulations, reducing the risk of production shutdowns or costly waste disposal fees associated with hazardous halogenated waste streams. This regulatory compliance translates into a more reliable API intermediate supplier relationship, as the production capacity is not constrained by environmental permits for toxic solvent usage.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the simplification of the synthetic sequence and the substitution of high-cost inputs. By removing the iodine activation step, the process eliminates the expense of purchasing iodine reagents and the downstream costs associated with scavenging residual iodine from the product stream. Additionally, the high molar yield of approximately 60.6 percent from GCLE to the final API implies a more efficient utilization of the starting material, reducing the effective cost per kilogram of the active ingredient. The use of immobilized enzymes further contributes to cost efficiency, as these biocatalysts can often be recovered and reused multiple times, lowering the recurring cost of goods sold compared to stoichiometric chemical reagents that are consumed in a single pass.
- Enhanced Supply Chain Reliability: The reliance on GCLE as a starting material leverages an established supply chain for cephalosporin intermediates, ensuring consistent availability. GCLE is a commercially mature building block produced by multiple manufacturers, mitigating the risk of single-source dependency. The robustness of the precipitation steps in the process also enhances supply continuity; because the intermediates are isolated as solids with low solubility, they are stable and easy to store or transport between different production sites if a multi-site manufacturing strategy is employed. This flexibility allows supply chain heads to buffer inventory at the intermediate stage (such as 7-ACP) without significant degradation concerns, providing a safety net against demand fluctuations or upstream disruptions.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard unit operations such as cooling, stirring, filtration, and drying, which are universally available in multipurpose API plants. The absence of extreme conditions (such as high pressure or cryogenic temperatures below -30°C) reduces the engineering complexity and capital expenditure required for reactor design. From an environmental standpoint, the aqueous nature of the enzymatic step and the ability to recover organic solvents from the precipitation mother liquors align with green chemistry principles. This significantly reduces the volume of hazardous waste generated, simplifying wastewater treatment and lowering the environmental compliance burden, which is increasingly a critical factor for multinational pharmaceutical companies auditing their contract manufacturing organizations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this GCLE-based synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and quality outcomes of the process. Understanding these details is essential for technical teams evaluating the transfer of this technology to their own manufacturing facilities or for procurement officers assessing the quality assurance protocols of their suppliers.
Q: How does this GCLE route control Delta-2 isomer impurities compared to traditional methods?
A: Traditional methods using Freon or CCl4 rely on low solubility to precipitate products and reduce isomerization. This novel route utilizes the formation of an inner salt type compound (3-NMP-DGCLE) which has extremely low solubility in various solvents. This property allows the product to precipitate rapidly from the reaction solution, fundamentally solving the generation of Delta-2 isomers without requiring environmentally prohibited polyhalogenated hydrocarbon solvents.
Q: What are the cost advantages of using GCLE over 7-ACA or ACLH starting materials?
A: The GCLE route eliminates the need for expensive iodine activation reagents required in ACLH-based processes. Furthermore, it avoids the use of costly and environmentally restricted solvents like Freon TF. By utilizing cheap and easily obtained acids for deprotection and standard organic solvents for precipitation, the overall manufacturing cost is significantly reduced while maintaining a high molar yield of approximately 60.6% based on the starting material.
Q: Is the enzymatic step in Step 3 suitable for large-scale industrial production?
A: Yes, the process utilizes immobilized penicillin acylase, which is robust and suitable for industrial applications. The reaction occurs under mild conditions (27-29°C, pH 7.9-8.1) in an aqueous medium. The enzyme can be filtered off and potentially reused, and the subsequent decolorization with activated carbon ensures high purity. The precipitation of 7-ACP is achieved by simple pH adjustment and solvent addition, making the workup straightforward and scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the GCLE-based method to maintain competitiveness in the global antibiotic market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity Cefepime Hydrochloride that meets stringent purity specifications, utilizing our rigorous QC labs to monitor every critical parameter, from isomer ratios to residual solvent levels. Our facility is equipped to handle the specific requirements of this process, including low-temperature reactors for the quaternization step and dedicated zones for enzymatic reactions, guaranteeing consistent batch-to-batch quality.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this optimized synthesis route, we can help you achieve substantial cost savings while securing a stable supply of this essential fourth-generation cephalosporin. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your long-term supply chain goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
