Advanced Chromone Synthesis for Oncology: Scalable Routes and Commercial Viability
Advanced Chromone Synthesis for Oncology: Scalable Routes and Commercial Viability
The pharmaceutical landscape is constantly evolving, with a significant pivot towards targeted oncology therapies that demand highly specialized chemical scaffolds. Patent CN1155590C introduces a pivotal advancement in the synthesis of novel chromone compounds exhibiting potent antineoplastic activity, marking a departure from their historical classification merely as antimicrobial agents. This technology provides a robust framework for generating diverse heterocyclic libraries, specifically focusing on chromone derivatives fused with benzimidazole, benzoxazole, or benzothiazole moieties. For R&D directors and procurement strategists, understanding the nuances of this patent is critical, as it outlines a pathway to high-value intermediates that address unmet medical needs in cancer treatment. The innovation lies not just in the biological activity against cell lines like KB and A2780, but in the chemical flexibility that allows for the tuning of physicochemical properties through systematic structural modifications. By leveraging this intellectual property, stakeholders can secure a competitive edge in the development of next-generation antitumor drugs, ensuring a pipeline rich with novel chemical entities that possess optimized pharmacokinetic profiles and enhanced therapeutic indices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chromone-based heterocycles relied heavily on direct condensation methods that often lacked precision and scalability. Prior art, such as the methods disclosed by K.R.S. Reddy, typically involved reacting 7-methoxychromone-2-methyl formate directly with o-phenylenediamine in polyphosphoric acid at extreme temperatures around 170°C. While effective for small-scale laboratory synthesis, these conventional routes suffer from significant drawbacks when viewed through the lens of industrial manufacturing. The harsh thermal conditions often lead to the degradation of sensitive functional groups, limiting the scope of substituents that can be introduced onto the chromone core. Furthermore, the lack of an isolated intermediate step makes process control difficult, resulting in inconsistent batch-to-batch quality and challenging purification protocols. The inability to selectively steer the reaction towards specific ring-opened or ring-closed products without altering the fundamental starting materials restricts the chemical diversity available for medicinal chemistry optimization, thereby slowing down the drug discovery timeline.
The Novel Approach
The methodology presented in CN1155590C overcomes these historical bottlenecks by introducing a modular three-step synthetic sequence that prioritizes control and versatility. Instead of a direct one-pot condensation, the process begins with the activation of chromone carboxylic acid into an acyl chloride using phosphorus pentachloride in toluene. This activation step is crucial as it enhances the electrophilicity of the carbonyl carbon, facilitating a milder and more efficient amidation reaction with various ortho-substituted anilines. 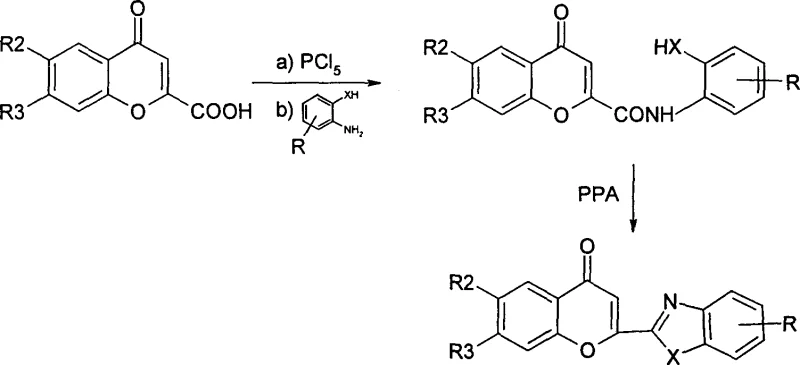 This strategic insertion of an amide intermediate allows chemists to purify the precursor before the final cyclization, significantly improving the overall purity of the final API intermediate. Moreover, the use of polyphosphoric acid as a cyclization agent at tunable temperatures enables the selective formation of either the closed-ring chromone derivatives or their open-ring counterparts. This level of control is unprecedented in prior art, offering a reliable chromone intermediate supplier the ability to produce distinct chemical entities from a common set of precursors, thereby maximizing asset utilization and reducing waste generation in the manufacturing process.
This strategic insertion of an amide intermediate allows chemists to purify the precursor before the final cyclization, significantly improving the overall purity of the final API intermediate. Moreover, the use of polyphosphoric acid as a cyclization agent at tunable temperatures enables the selective formation of either the closed-ring chromone derivatives or their open-ring counterparts. This level of control is unprecedented in prior art, offering a reliable chromone intermediate supplier the ability to produce distinct chemical entities from a common set of precursors, thereby maximizing asset utilization and reducing waste generation in the manufacturing process.
Mechanistic Insights into Polyphosphoric Acid Catalyzed Cyclization
The core of this technological breakthrough resides in the mechanistic behavior of the amide intermediate under the influence of polyphosphoric acid (PPA). PPA acts not merely as a solvent but as a powerful dehydrating agent and acid catalyst that promotes intramolecular nucleophilic attack. In the formation of Formula I compounds, the nitrogen or oxygen atom of the ortho-substituted aniline moiety attacks the activated carbonyl group, leading to ring closure and the elimination of water. 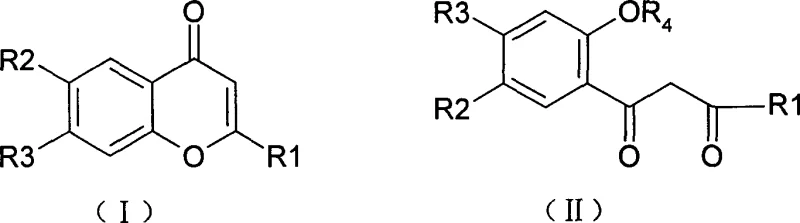 The elegance of this mechanism is revealed in the temperature-dependent divergence observed in the patent data. At lower temperature ranges, specifically between 70°C and 160°C, the kinetic pathway favors the formation of the stable, closed-ring heterocyclic system found in Formula I. However, as the thermal energy is increased to the 140°C to 190°C range, the system accesses a different thermodynamic minimum or undergoes a subsequent hydrolysis-like ring-opening event, yielding the Formula II open-chain diketone structures. This mechanistic understanding is vital for process chemists, as it implies that impurity profiles can be managed by strict temperature control, ensuring that the desired isomer is produced with high selectivity. Such precision reduces the burden on downstream purification units, directly translating to cost reduction in antitumor agent manufacturing by minimizing solvent usage and chromatography steps.
The elegance of this mechanism is revealed in the temperature-dependent divergence observed in the patent data. At lower temperature ranges, specifically between 70°C and 160°C, the kinetic pathway favors the formation of the stable, closed-ring heterocyclic system found in Formula I. However, as the thermal energy is increased to the 140°C to 190°C range, the system accesses a different thermodynamic minimum or undergoes a subsequent hydrolysis-like ring-opening event, yielding the Formula II open-chain diketone structures. This mechanistic understanding is vital for process chemists, as it implies that impurity profiles can be managed by strict temperature control, ensuring that the desired isomer is produced with high selectivity. Such precision reduces the burden on downstream purification units, directly translating to cost reduction in antitumor agent manufacturing by minimizing solvent usage and chromatography steps.
Furthermore, the substituent effects on the phenyl ring play a critical role in the electronic distribution and subsequent reactivity of the intermediate. The patent specifies that electron-withdrawing or donating groups such as halogens, nitro, or alkoxy groups at the R2 and R3 positions do not inhibit the cyclization but rather allow for the fine-tuning of the electronic properties of the final molecule. This is particularly important for optimizing the binding affinity of the drug candidate to its biological target. The ability to incorporate diverse substituents without compromising the integrity of the cyclization step demonstrates the robustness of the reaction mechanism. For supply chain heads, this means that a single production line can be adapted to manufacture a wide array of analogues simply by swapping the aniline starting material, enhancing the agility of the supply chain to respond to changing clinical trial requirements. The rigorous control over the reaction environment ensures that even complex substituted chromones can be produced with stringent purity specifications, meeting the demanding standards of global regulatory bodies.
How to Synthesize Antitumor Chromone Derivatives Efficiently
Implementing this synthesis route requires a disciplined approach to unit operations, beginning with the careful handling of moisture-sensitive reagents. The initial conversion of chromone carboxylic acid to its acyl chloride derivative must be conducted under strictly anhydrous conditions to prevent hydrolysis, typically utilizing toluene as an azeotropic solvent to remove generated HCl gas. Following isolation, the amide coupling step employs mild bases like triethylamine to scavenge acid, ensuring high conversion rates without epimerization or degradation of the sensitive chromone core.
- Convert substituted chromone carboxylic acid to acyl chloride using phosphorus pentachloride in toluene under anhydrous conditions.
- React the resulting acyl chloride with ortho-substituted anilines such as o-phenylenediamine to form the critical amide intermediate.
- Execute cyclization in polyphosphoric acid at controlled temperatures between 70°C and 190°C to selectively produce closed or open-ring structures.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial strategic benefits for procurement managers and supply chain leaders looking to optimize their vendor portfolios. The primary advantage lies in the utilization of commodity chemicals such as phosphorus pentachloride, toluene, and polyphosphoric acid, which are readily available in the global market at stable price points. This reliance on established industrial reagents mitigates the risk of supply disruptions often associated with exotic catalysts or proprietary enzymes, ensuring a continuous and reliable flow of materials for production campaigns. Additionally, the modularity of the synthesis allows for the consolidation of multiple product SKUs onto a single manufacturing platform, drastically simplifying inventory management and reducing the capital expenditure required for dedicated equipment. By streamlining the production process, manufacturers can achieve significant cost savings in fine chemical intermediates manufacturing, passing these efficiencies on to partners in the form of more competitive pricing models for clinical and commercial supply agreements.
- Cost Reduction in Manufacturing: The elimination of complex multi-step protection and deprotection sequences traditionally required for heterocyclic synthesis leads to a drastic simplification of the overall process flow. By utilizing a direct acyl chloride activation strategy, the number of unit operations is minimized, which directly correlates to reduced labor costs, lower energy consumption, and decreased solvent waste disposal fees. The high selectivity of the PPA-mediated cyclization further enhances yield efficiency, ensuring that a greater proportion of raw materials are converted into valuable saleable product rather than lost as byproducts. This operational efficiency creates a lean manufacturing environment that supports substantial cost savings without compromising on the quality or purity of the final antitumor intermediates supplied to pharmaceutical clients.
- Enhanced Supply Chain Reliability: The robustness of the chemical pathway ensures that production timelines are predictable and less susceptible to the variability often seen in sensitive biocatalytic processes. Since the reaction conditions are well-defined and rely on stable inorganic acids and organic solvents, scale-up from laboratory to pilot plant and finally to commercial tonnage can be executed with high confidence. This predictability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing drug developers to adhere to strict clinical trial schedules without the fear of material shortages. Furthermore, the ability to store stable amide intermediates provides a buffer stock capability, adding an extra layer of security to the supply chain against unforeseen demand spikes or logistical delays in raw material delivery.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex heterocyclic compounds, utilizing reactors and separation techniques that are standard in the fine chemical industry. The use of polyphosphoric acid, while requiring careful handling, allows for easier workup procedures compared to strong liquid acids, potentially reducing the volume of aqueous waste generated. Moreover, the high atom economy of the cyclization step aligns with modern green chemistry principles, minimizing the environmental footprint of the manufacturing process. This commitment to sustainable production practices not only meets regulatory compliance standards but also appeals to environmentally conscious stakeholders, reinforcing the long-term viability of the supply partnership.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and sourcing of these chromone derivatives. Understanding these details is essential for establishing a transparent and productive collaboration between chemical suppliers and pharmaceutical innovators.
Q: What distinguishes this chromone synthesis from conventional antimicrobial routes?
A: Unlike traditional methods that focus on antimicrobial properties using direct high-temperature condensation, this patented approach utilizes a controlled acyl chloride intermediate strategy. This allows for precise temperature modulation during the polyphosphoric acid cyclization step, enabling the selective production of either closed-ring chromones or open-ring derivatives with specific antitumor profiles.
Q: How does the temperature variation affect the product profile in this process?
A: The process demonstrates remarkable tunability where reaction temperature dictates the structural outcome. Lower temperature ranges around 70°C to 160°C favor the formation of the closed-ring Formula I compounds, while elevating the temperature to 140°C to 190°C promotes the formation of open-ring Formula II products, providing a versatile platform for structure-activity relationship studies without changing starting materials.
Q: Are the reagents used in this synthesis suitable for large-scale manufacturing?
A: Yes, the synthesis relies on industrially established reagents such as phosphorus pentachloride, toluene, and polyphosphoric acid. These materials are globally sourced and cost-effective, eliminating the dependency on exotic or prohibitively expensive catalysts, which significantly de-risks the supply chain for commercial kilogram-to-ton production campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chromone Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a partner who understands both the scientific intricacies and the commercial imperatives of drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from preclinical research to market launch is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of chromone intermediates meets the exacting standards required for oncology drug formulation. Our commitment to technical excellence means we can navigate the complexities of the PPA cyclization process with precision, delivering materials that accelerate your research timelines.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis routes can improve your project economics. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us be the foundation of your supply chain success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
