Scalable Synthesis of Antineoplastic Chromone Derivatives via Controlled Polyphosphoric Acid Cyclization
Scalable Synthesis of Antineoplastic Chromone Derivatives via Controlled Polyphosphoric Acid Cyclization
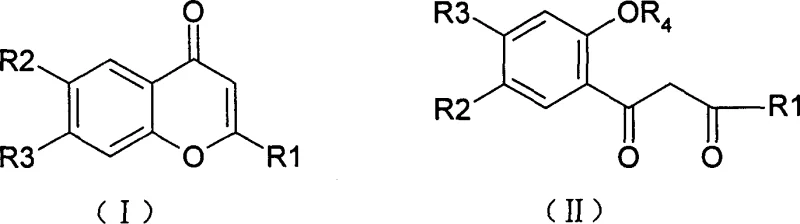
The pharmaceutical industry is constantly seeking novel scaffolds with potent biological activity, particularly in the realm of oncology where resistance to existing therapies remains a critical challenge. Patent CN1382688A introduces a significant advancement in this field by disclosing a series of new chromone compounds of general formula (I) and their corresponding ring-opened products of general formula (II), both exhibiting remarkable antineoplastic activity. This technology represents a pivotal shift from traditional antibacterial chromone syntheses to targeted antitumor applications, leveraging a versatile synthetic pathway that allows for precise structural modulation. By utilizing substituted chromone carboxylic acids as key starting materials, the process enables the efficient construction of complex benzazole-fused heterocycles. For R&D directors and procurement specialists alike, understanding the nuances of this patented methodology is essential for securing a reliable supply of high-purity pharmaceutical intermediates capable of driving next-generation cancer therapeutics forward.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzazole-substituted chromones has relied heavily on methodologies that often suffer from rigid condition requirements and limited substrate scope. As noted in prior art, such as the work by K.R.S. Reddy et al., traditional routes frequently utilize chromone esters like 7-methoxychromone-2-methyl carboxylate as starting materials. These ester-based approaches typically necessitate harsh reaction environments, such as heating with o-phenylenediamine in polyphosphoric acid at temperatures as high as 170°C to achieve direct cyclization. Alternatively, a two-step process involving amide formation at 100°C followed by cyclization at 170°C was required. Such high-temperature protocols not only increase energy consumption but also pose significant risks regarding thermal degradation of sensitive functional groups and the formation of complex impurity profiles. Furthermore, the reliance on ester intermediates adds an unnecessary hydrolysis step if carboxylic acid functionality is desired in the final molecule, thereby elongating the synthetic timeline and reducing overall atom economy.
The Novel Approach
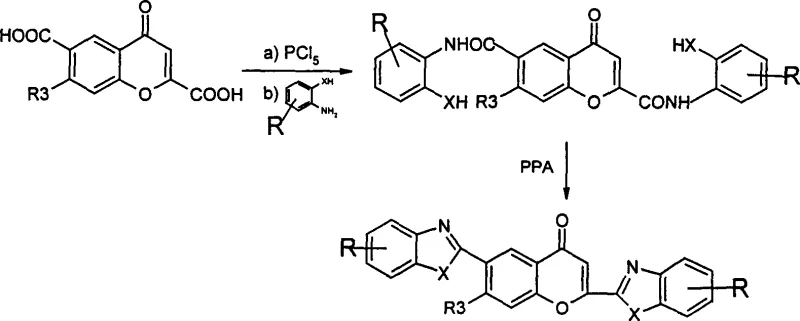
In stark contrast, the methodology described in CN1382688A offers a streamlined and highly controllable alternative that directly utilizes chromone carboxylic acids. This novel approach initiates with the conversion of the carboxylic acid into a reactive acyl chloride using phosphorus pentachloride in toluene, a transformation that proceeds under relatively mild conditions compared to direct ester aminolysis. The subsequent reaction with ortho-substituted anilines, such as o-phenylenediamine or o-aminophenol, generates the crucial amide intermediate with high fidelity. The true innovation lies in the final cyclization step using polyphosphoric acid (PPA), where the reaction outcome is exquisitely sensitive to temperature control. By modulating the temperature between 70°C and 160°C, manufacturers can selectively produce the closed-ring chromone derivatives (Formula I), whereas raising the temperature to the 140°C to 190°C range facilitates ring-opening to yield the distinct diketone structures (Formula II). This level of control provides a strategic advantage in cost reduction in pharmaceutical intermediate manufacturing by allowing a single production line to access two distinct chemical spaces simply by adjusting thermal parameters.
Mechanistic Insights into Polyphosphoric Acid Mediated Cyclization
The core of this synthetic strategy revolves around the electrophilic activation of the amide carbonyl by the strongly acidic medium of polyphosphoric acid. Upon formation of the amide intermediate from the chromone acyl chloride and the diamine or aminophenol partner, the molecule possesses the necessary proximity for intramolecular nucleophilic attack. In the PPA medium, the carbonyl oxygen is protonated, increasing the electrophilicity of the carbonyl carbon. The adjacent nucleophilic nitrogen or oxygen atom from the ortho-substituent then attacks this activated center, leading to the formation of the benzimidazole, benzoxazole, or benzothiazole ring fused to the chromone scaffold. This cyclization is thermodynamically driven and results in the stable aromatic systems observed in Formula (I). The robustness of this mechanism ensures that even with varied substituents on the chromone ring, such as methoxy, nitro, or halogen groups, the cyclization proceeds efficiently, minimizing the risk of side reactions that often plague transition-metal catalyzed couplings.
Furthermore, the mechanism allows for a unique divergence pathway leading to ring-opened products, which is a rare feature in heterocyclic synthesis. When the reaction temperature is elevated significantly, specifically into the 140°C to 190°C range, the stability of the pyrone ring in the chromone core is compromised under the aggressive acidic conditions. This leads to the hydrolytic cleavage of the lactone bond, resulting in the formation of the open-chain 1,3-diketone derivatives represented by Formula (II). The ability to toggle between the closed cyclic form and the open chain form by simply adjusting the thermal profile of the PPA reaction offers profound implications for structure-activity relationship (SAR) studies. It implies that a single batch of amide intermediate can be split and processed under different conditions to generate a diverse library of analogues, thereby accelerating the drug discovery process without the need for synthesizing entirely new starting materials for each variant.
How to Synthesize Antineoplastic Chromone Intermediates Efficiently
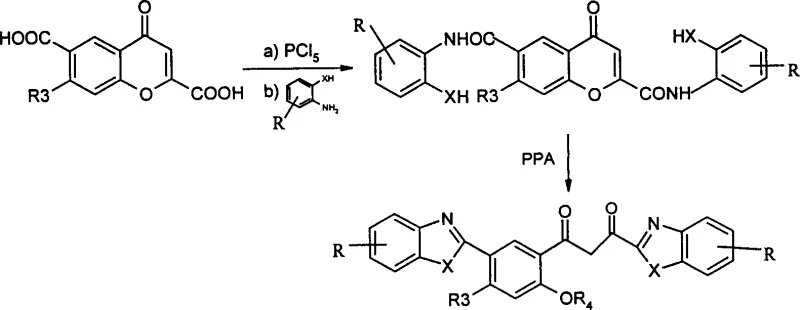
The execution of this synthesis requires careful attention to anhydrous conditions during the acyl chloride formation and precise thermal management during the cyclization phase. The process begins with the activation of the chromone carboxylic acid, followed by coupling with the appropriate amine component to build the molecular framework. The final step in polyphosphoric acid is the critical determinant of product identity, where temperature acts as the switch between cyclic and acyclic architectures. Detailed operational parameters, including specific solvent volumes, stoichiometric ratios, and workup procedures, are essential for reproducing the high yields and purity reported in the patent literature. For process chemists looking to implement this route, adhering to the standardized protocol ensures consistent quality and minimizes the formation of byproducts.
- Convert substituted chromone carboxylic acid to the corresponding acyl chloride using phosphorus pentachloride in toluene under anhydrous conditions.
- React the crude acyl chloride with ortho-substituted anilines such as o-phenylenediamine or o-aminophenol in the presence of a base to form the amide intermediate.
- Cyclize the amide intermediate in polyphosphoric acid at controlled temperatures between 70°C and 190°C to selectively yield either closed-ring chromones or ring-opened products.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this PPA-mediated cyclization technology offers substantial benefits regarding raw material availability and process scalability. Unlike methods relying on precious metal catalysts which are subject to volatile market pricing and supply constraints, this process utilizes commodity chemicals such as phosphorus pentachloride and polyphosphoric acid. These reagents are globally sourced and available in bulk quantities, ensuring that production schedules are not disrupted by raw material shortages. Additionally, the elimination of heavy metal catalysts simplifies the downstream purification process, as there is no need for expensive metal scavenging resins or complex filtration steps to meet stringent residual metal specifications required for pharmaceutical ingredients. This simplification directly translates to reduced processing time and lower operational expenditures.
- Cost Reduction in Manufacturing: The economic efficiency of this route is driven by the high atom economy of the cyclization step and the avoidance of costly catalytic systems. By using polyphosphoric acid as both the solvent and the dehydrating agent, the process eliminates the need for additional coupling reagents that generate stoichiometric waste. Furthermore, the ability to produce two distinct classes of bioactive molecules (Formula I and Formula II) from a common amide intermediate allows for significant inventory optimization. Manufacturers can maintain a stock of the versatile intermediate and convert it to the specific final product based on real-time demand, thereby reducing capital tied up in finished goods and minimizing the risk of obsolescence.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations described ensures high batch-to-b consistency, which is critical for maintaining long-term supply contracts. The reaction conditions are tolerant to a wide range of substituents, meaning that the same equipment train can be used to produce various analogues within the chemical family without extensive requalification. This flexibility enhances the resilience of the supply chain against fluctuations in specific API demand. Moreover, the use of standard solvents like toluene and dichloromethane, which have well-established recovery and recycling protocols, further stabilizes the cost structure and reduces dependency on specialty solvent markets.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of sensitive catalytic species that often behave unpredictably at larger scales. The exothermic nature of the acyl chloride formation can be easily managed with standard cooling jackets, and the high-boiling nature of PPA allows for safe operation at the elevated temperatures required for ring-opening. From an environmental standpoint, while PPA requires careful neutralization during workup, the overall waste profile is cleaner compared to heavy metal catalyzed routes. The aqueous waste streams primarily contain phosphate salts, which are easier to treat and dispose of in compliance with modern environmental regulations compared to toxic heavy metal sludge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chromone derivatives. Understanding these details is vital for partners evaluating the feasibility of integrating these intermediates into their drug development pipelines. The answers provided are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and reliability for decision-making purposes.
Q: How does reaction temperature affect the product distribution in this synthesis?
A: Reaction temperature in polyphosphoric acid is the critical control parameter. Lower temperatures around 70-160°C favor the formation of closed-ring chromone compounds (Formula I), while higher temperatures between 140-190°C promote ring-opening to yield the diketone derivatives (Formula II).
Q: What are the primary starting materials for these antineoplastic intermediates?
A: The synthesis begins with substituted chromone carboxylic acids, such as 7-methoxy-2-chromone carboxylic acid or 2,6-dicarboxylic acid derivatives, which are converted into reactive acyl chlorides before coupling with benzazole precursors.
Q: Can this process be scaled for commercial API production?
A: Yes, the process utilizes standard industrial reagents like phosphorus pentachloride and polyphosphoric acid, avoiding exotic catalysts. This makes the route highly amenable to scale-up from kilogram to multi-ton production levels with consistent quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Antineoplastic Chromone Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the supply of complex oncology intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your clinical and commercial needs are met with unwavering consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chromone intermediate meets the highest standards of quality required for pharmaceutical applications. Our facility is equipped to handle the specific thermal and corrosive challenges associated with polyphosphoric acid chemistry, providing a safe and efficient environment for manufacturing these valuable compounds.
We invite you to collaborate with us to optimize your supply chain for antineoplastic drug development. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your project timelines and budgetary goals effectively.
