Optimizing Pinoxaden Production: A 4-Step Catalytic Route for Commercial Scale-Up
Introduction to Advanced Pinoxaden Manufacturing
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate synthesis, particularly for phenyl pyrazole compounds like Pinoxaden. A pivotal advancement in this domain is detailed in patent CN108864144B, which discloses a highly efficient synthetic method that fundamentally alters the production landscape for this critical active ingredient. This technology addresses the longstanding industry challenge of lengthy synthesis routes by introducing a streamlined 4-step process that begins with the readily available starting material, 2,6-diethyl-4-methylaniline. By integrating thermal decomposition via diazotization and advanced palladium-catalyzed coupling strategies, this method achieves a level of operational simplicity and economic viability that was previously difficult to attain. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a competitive edge in the herbicide market. The following analysis dissects the technical merits and commercial implications of adopting this novel manufacturing protocol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Pinoxaden has been plagued by inefficiencies inherent in multi-step sequences that rely on the formation of 2-(2,6-diethyl-4-methylphenyl) malonic acid derivatives. Traditional pathways often necessitate between 5 to 7 distinct chemical steps to reach the final active pharmaceutical ingredient, creating a bottleneck in production throughput. These extended sequences not only accumulate yield losses at every stage but also introduce significant complexity in impurity management and waste treatment. The reliance on synthesizing malonamide or malonic diester intermediates through convoluted routes increases the consumption of solvents and reagents, thereby inflating the overall cost of goods sold (COGS). Furthermore, the operational burden of managing multiple isolation and purification stages across such a long sequence poses substantial risks to supply chain continuity and batch-to-batch consistency. As regulatory pressures mount regarding environmental impact, these older, more wasteful processes are becoming increasingly untenable for modern, sustainability-focused manufacturing facilities.
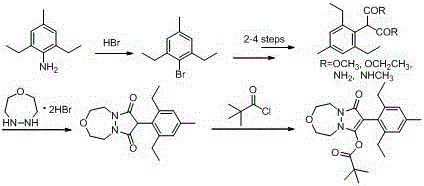
The Novel Approach
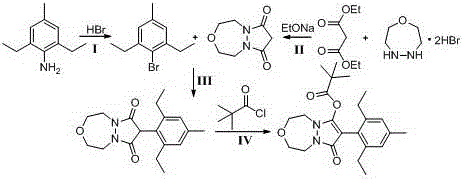
In stark contrast to the cumbersome legacy methods, the innovative route disclosed in the patent offers a radical simplification by condensing the entire synthesis into just four strategic steps. This approach leverages a direct diazotization and Sandmeyer reaction to rapidly generate the key aryl bromide building block, 2,6-diethyl-4-methyl bromobenzene, from the aniline precursor. Subsequently, the construction of the complex heterocyclic core is achieved through a robust condensation reaction involving diethyl malonate and [1,4,5]-oxadiazepine dihydrobromide, catalyzed efficiently by sodium ethoxide. The convergence of these fragments is realized through a high-yielding palladium-catalyzed coupling, which directly installs the aromatic moiety onto the pyrazolo-oxadiazepine scaffold. Finally, a straightforward esterification with pivaloyl chloride delivers the target molecule, Pinoxaden. This telescoped logic not only minimizes material handling but also drastically reduces the time required to bring a batch from raw materials to finished product, representing a paradigm shift in agrochemical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Coupling and Cyclization
The heart of this synthetic breakthrough lies in the precise orchestration of the coupling reaction (Step III) and the preceding cyclization (Step II). In Step II, the formation of dihydro-1H-pyrazolo[1,2-d][1,4,5]oxadiazepine-7,9(2H,8H)-dione is driven by the nucleophilic attack of the enolate derived from diethyl malonate upon the oxadiazepine ring system. The use of sodium ethoxide as a base catalyst is critical here, as it facilitates the deprotonation of the active methylene group under nitrogen atmosphere at elevated temperatures ranging from 160°C to 180°C. This thermal energy is necessary to overcome the activation barrier for the ring closure while simultaneously distilling off the ethanol byproduct, driving the equilibrium towards the desired dione product. The careful control of stoichiometry, specifically maintaining a molar ratio of diethyl malonate to the oxadiazepine salt between 1:0.8 and 1:1, ensures minimal formation of polymeric side products.
Following the core construction, Step III employs a sophisticated transition metal catalysis strategy to unite the heterocyclic core with the lipophilic aryl bromide. The reaction utilizes bis(triphenylphosphine)palladium(II) chloride, Pd(PPh3)2Cl2, which serves as a robust precatalyst for the cross-coupling event. Conducted in dimethylformamide (DMF) at temperatures between 115°C and 135°C, this step likely proceeds through an oxidative addition of the aryl bromide to the Pd(0) species, followed by transmetallation or direct insertion mechanisms depending on the specific nature of the nucleophilic partner generated in situ. The presence of the bulky phosphine ligands stabilizes the palladium center against aggregation, ensuring high turnover numbers and consistent yields exceeding 80% in optimized examples. This mechanistic precision is vital for R&D teams aiming to replicate the process, as it dictates the strict requirement for anhydrous conditions and precise temperature control to prevent catalyst deactivation and ensure the high purity of the resulting 8-(2,6-diethyl-4-methylphenyl) tetrahydropyrazolo derivative.
How to Synthesize Pinoxaden Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters outlined in the patent examples to achieve the reported yields and purity profiles. The process begins with the careful preparation of the aryl bromide via diazotization in hydrobromic acid, followed by the high-temperature cyclization to build the central scaffold. The subsequent coupling and final esterification steps demand precise stoichiometric control of the palladium catalyst and acylating agents, respectively. For process chemists looking to translate this from bench to pilot plant, the detailed standardized synthesis steps provided below offer a comprehensive roadmap for execution, ensuring that critical quality attributes are met at every stage of the workflow.
- Perform diazotization of 2,6-diethyl-4-methylaniline followed by Sandmeyer reaction with cuprous bromide to generate 2,6-diethyl-4-methyl bromobenzene.
- Condense diethyl malonate with [1,4,5]-oxadiazepine dihydrobromide using sodium ethoxide catalyst at elevated temperatures to form the pyrazolo-oxadiazepine dione core.
- Execute a palladium-catalyzed coupling between the pyrazolo-oxadiazepine dione and the aryl bromide intermediate, followed by esterification with pivaloyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this 4-step synthesis route translates into tangible strategic benefits that extend far beyond simple chemical yield. By fundamentally restructuring the manufacturing workflow, this technology addresses the core pain points of cost volatility and lead time uncertainty that plague the agrochemical sector. The reduction in unit operations directly correlates to lower capital expenditure on equipment utilization and reduced labor hours per kilogram of output. Furthermore, the reliance on commodity chemicals such as 2,6-diethyl-4-methylaniline and diethyl malonate mitigates the risk of supply disruptions associated with exotic or proprietary starting materials. This robustness allows for more accurate forecasting and inventory planning, ensuring that production schedules can be maintained even in fluctuating market conditions. The simplified purification protocols, which utilize standard solvent systems like ethyl acetate and petroleum ether, further enhance the economic profile by reducing solvent recovery costs and waste disposal fees.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in synthetic steps from the conventional 5-7 down to just 4. Each eliminated step removes an entire cycle of reagent consumption, solvent usage, energy input for heating and cooling, and labor for monitoring and workup. Qualitatively, this leads to a significant compression of the variable cost structure, allowing manufacturers to operate with healthier margins or offer more competitive pricing to downstream formulators. Additionally, the use of sodium ethoxide as a simple, inexpensive base catalyst in Step II, rather than complex organometallic reagents, further drives down the raw material bill. The high atom economy of the coupling and esterification steps ensures that a greater proportion of the input mass is converted into valuable product rather than waste, maximizing the return on investment for every ton of raw material purchased.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, commodity-grade starting materials that are produced by multiple global suppliers. 2,6-diethyl-4-methylaniline and diethyl malonate are established bulk chemicals, meaning their availability is not subject to the bottlenecks often seen with specialized fine chemical intermediates. This diversification of the supply base reduces the risk of single-source dependency and price gouging. Moreover, the shortened reaction sequence inherently reduces the total processing time, effectively shrinking the manufacturing lead time. Faster turnaround times mean that inventory levels can be kept leaner without risking stockouts, improving cash flow and working capital efficiency. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require extreme cryogenic temperatures or ultra-high pressures, further ensures that production can be sustained reliably across different manufacturing sites.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for commercial scale-up of complex agrochemical intermediates. The process avoids the use of heavy metal catalysts that are difficult to remove and pose significant environmental hazards, relying instead on palladium which can be recovered and recycled, and copper which is used in manageable quantities in the initial step. The workup procedures involve standard liquid-liquid extractions and distillations, which are unit operations that scale linearly and predictably from laboratory glassware to multi-ton reactors. The reduction in the number of steps also implies a proportional reduction in the volume of wastewater and organic waste generated, simplifying compliance with increasingly stringent environmental regulations. This 'green' profile not only lowers waste treatment costs but also aligns with the sustainability goals of major multinational agrochemical companies, making the manufacturer a preferred partner for long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Pinoxaden synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction specifics and scalability potential. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing method.
Q: How does this new synthesis route compare to conventional methods in terms of step count?
A: Conventional methods typically require 5 to 7 steps to synthesize the key malonic acid derivatives and final product. This patented route reduces the process to only 4 distinct chemical transformations, significantly simplifying the operational workflow.
Q: What are the critical reaction conditions for the coupling step?
A: The coupling reaction (Step III) utilizes a palladium catalyst, specifically Pd(PPh3)2Cl2, in dimethylformamide solvent. The reaction is conducted at temperatures between 115°C and 135°C for 2 to 6 hours to ensure high conversion rates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial viability. It utilizes commercially available raw materials like 2,6-diethyl-4-methylaniline and avoids exotic reagents. The purification steps involve standard techniques like distillation, extraction, and column chromatography, which are easily scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pinoxaden Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this 4-step synthesis are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Pinoxaden intermediate meets the highest global standards. Our commitment to quality assurance means that we can consistently deliver high-purity agrochemical intermediates that facilitate smooth downstream formulation processes for our clients.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us be your partner in driving efficiency and reliability in your herbicide production network.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
