Advanced One-Pot Catalytic Strategy for Commercial Pinoxaden Manufacturing
The global agrochemical sector is constantly evolving, driven by the need for more efficient and sustainable manufacturing processes for key active ingredients. A pivotal development in this landscape is documented in Chinese patent CN112028906B, which introduces a revolutionary "one-pot" methodology for the synthesis of pinoxaden, a potent ACCase inhibitor herbicide widely used in cereal crop protection. This technical breakthrough addresses critical bottlenecks in traditional manufacturing, offering a pathway that significantly streamlines production while enhancing overall yield and purity. For R&D directors and supply chain leaders, understanding the nuances of this catalytic system is essential for optimizing procurement strategies and ensuring long-term supply continuity. The patent details a robust protocol that utilizes readily available nano-oxide catalysts to facilitate a tandem coupling-acylation sequence, effectively collapsing what was previously a multi-step, resource-intensive operation into a singular, highly efficient unit operation. This shift not only promises substantial operational savings but also aligns with increasingly stringent environmental regulations regarding waste discharge and solvent usage in fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
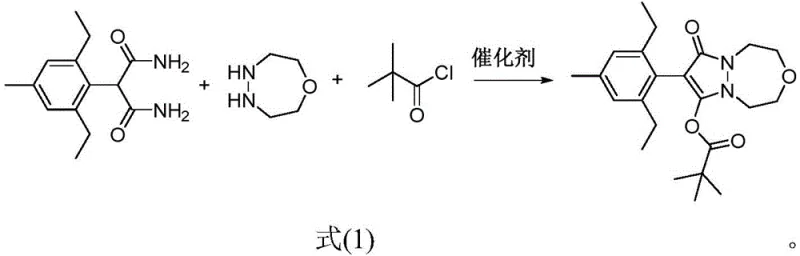
Historically, the industrial synthesis of pinoxaden has relied on a segmented approach where the formation of the core heterocyclic structure and the subsequent acylation were performed as distinct, isolated steps. In these traditional routes, the coupling reaction between the phenylmalonic acid derivative and the diazepane ring often fails to reach full conversion before the introduction of the acylating agent. This incomplete conversion is a major source of inefficiency, as any unreacted 1-oxo-4,5-diazepane remaining in the mixture inevitably reacts with the pivaloyl chloride in the second step. This side reaction generates significant quantities of N-acylated byproducts that are structurally similar to the target molecule, making them extremely difficult and costly to remove during purification. Furthermore, the necessity of isolating intermediates between steps increases the total batch cycle time, consumes excessive amounts of solvents for washing and crystallization, and creates a larger footprint of hazardous waste that requires treatment. These factors collectively drive up the cost of goods sold (COGS) and introduce variability that can compromise the consistency of the final active pharmaceutical ingredient (API) or technical grade material.
The Novel Approach
In stark contrast, the methodology outlined in patent CN112028906B leverages a sophisticated "one-pot" strategy that fundamentally reengineers the reaction workflow. By employing specific nano-structured metal oxide catalysts, the process ensures that the initial condensation between 2-(2,6-diethyl-4-methylphenyl)malonamide and 1-oxo-4,5-diazepane proceeds to near-total completion within the same reactor vessel. Only after this critical coupling phase is finalized is the acylating agent, pivaloyl chloride, introduced along with a base such as triethylamine. This sequential addition within a single pot prevents the premature acylation of the free amine, thereby virtually eliminating the formation of the problematic N-acyl impurities associated with legacy methods. The result is a process that is not only chemically superior in terms of selectivity but also operationally streamlined, removing the need for intermediate filtration, drying, and re-dissolution. This consolidation of steps represents a paradigm shift for manufacturers seeking a reliable agrochemical intermediate supplier capable of delivering high-quality materials with a reduced environmental footprint.
Mechanistic Insights into Nano-Oxide Catalyzed Cyclization
The success of this one-pot transformation hinges on the unique properties of the selected catalysts: nano magnesium oxide, nano titanium oxide, or nano zirconium oxide. Unlike traditional homogeneous bases or acidic catalysts, these nano-oxides provide a heterogeneous surface that appears to specifically promote the aminolysis of the ester/amide functionality without facilitating unwanted side reactions. The mechanism likely involves the adsorption of the malonamide substrate onto the basic sites of the nano-oxide surface, which activates the carbonyl group towards nucleophilic attack by the diazepane nitrogen. This surface-mediated activation lowers the energy barrier for the cyclization step, driving the equilibrium strongly towards the formation of the pyrazolo-diazepine core. Crucially, the catalyst remains stable and active throughout the initial heating phase (100-120°C), ensuring that the concentration of free diazepane is minimized before the acylation reagent is added. This precise control over reaction kinetics is what allows the process to achieve high conversion rates, often exceeding 70% in optimized examples, without the need for exotic or expensive transition metal complexes.
From an impurity control perspective, this mechanistic pathway offers distinct advantages for quality assurance teams. In conventional syntheses, the presence of unreacted starting materials often leads to a complex impurity profile that requires rigorous chromatographic purification to meet regulatory standards. However, by forcing the coupling reaction to completion prior to acylation, the novel method effectively "cleans" the reaction mixture in situ. The primary byproduct generated is simply the salt of the base used (triethylamine hydrochloride), which is easily removed via aqueous workup. This inherent selectivity means that the crude product obtained after solvent removal is of significantly higher purity compared to traditional batches. For a reliable agrochemical intermediate supplier, this translates to a more robust quality control process, reduced risk of batch rejection, and the ability to consistently meet the stringent purity specifications required by global regulatory bodies for herbicide registration.
How to Synthesize Pinoxaden Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it highly attractive for commercial manufacturing environments. The process begins by charging the reactor with the malonamide precursor, the diazepane heterocycle, and a catalytic amount of the nano-oxide in a suitable solvent such as toluene or dioxane. The mixture is then heated to reflux temperatures, typically between 100°C and 120°C, and maintained under agitation for a period of 8 to 12 hours to ensure full conversion of the coupling step. Following this, the reaction mass is cooled to ambient temperature, and the acylation reagents are introduced directly into the same vessel. This seamless transition between reaction phases eliminates the logistical challenges of handling sensitive intermediates and significantly reduces the overall processing time. Detailed standardized operating procedures for this synthesis can be found in the technical documentation below.
- Combine 2-(2,6-diethyl-4-methylphenyl)malonamide, 1-oxo-4,5-diazepane, and a nano-oxide catalyst (MgO, TiO2, or ZrO2) in a solvent like toluene.
- Heat the mixture to 100-120°C and stir for 8-12 hours to complete the coupling reaction.
- Cool to room temperature, add triethylamine and pivaloyl chloride, and stir for 5-6 hours to finalize acylation and isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot catalytic technology offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The consolidation of two distinct chemical transformations into a single reactor run drastically reduces the utilization of utilities such as steam for heating and chilled water for cooling, as well as minimizing the labor hours required for reactor turnover and cleaning. Furthermore, the elimination of intermediate isolation steps means that less solvent is consumed overall, which directly correlates to lower raw material costs and reduced expenses associated with solvent recovery or disposal. This efficiency gain is critical for maintaining competitiveness in the global agrochemical market, where margin pressures are constant. By partnering with a manufacturer utilizing this advanced route, buyers can secure a more stable supply of high-purity pinoxaden while mitigating the risks associated with complex, multi-step supply chains.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the unit operations. By removing the need to isolate, dry, and re-dissolve the intermediate, the process saves significant amounts of time and energy. Additionally, the use of heterogeneous nano-oxide catalysts avoids the need for expensive transition metals that often require complex removal steps to meet heavy metal limits in the final product. This simplification of the downstream processing train leads to substantial cost savings in both capital expenditure (smaller equipment footprint) and operational expenditure (lower utility and waste treatment costs), facilitating cost reduction in agrochemical manufacturing.
- Enhanced Supply Chain Reliability: From a logistics standpoint, shorter batch cycles translate directly to increased production capacity and flexibility. A process that combines two steps into one effectively doubles the throughput of existing reactor assets without the need for new infrastructure investment. This increased agility allows suppliers to respond more rapidly to fluctuations in market demand, ensuring that orders for commercial scale-up of complex herbicides can be fulfilled with shorter lead times. Moreover, the robustness of the reaction against impurity formation reduces the likelihood of batch failures, providing a more predictable and reliable supply stream for downstream formulators.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly improved, aligning with modern green chemistry principles. The reduction in solvent usage and the minimization of hazardous waste streams simplify the permitting process for manufacturing facilities and reduce the liability associated with waste disposal. The heterogeneous nature of the catalyst also facilitates easy separation via simple filtration, avoiding the contamination of the product stream with catalyst residues. These factors make the process highly scalable from pilot plant to multi-ton production, supporting the commercial scale-up of complex herbicides while adhering to strict environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel pinoxaden synthesis route. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's potential impact on your supply chain. Understanding these details is crucial for technical teams assessing the feasibility of integrating this material into their existing portfolios or for procurement specialists negotiating long-term supply agreements.
Q: What is the primary advantage of the one-pot method for pinoxaden synthesis?
A: The primary advantage is the significant reduction in process complexity and waste generation. By combining the coupling and acylation steps into a single reactor vessel, the method eliminates the need for intermediate isolation, thereby reducing solvent consumption and processing time while maintaining high conversion rates through the use of specialized nano-oxide catalysts.
Q: Which catalysts are most effective for this specific transformation?
A: The patent identifies nano-scale metal oxides as the critical catalytic agents. Specifically, nano magnesium oxide, nano titanium oxide, and nano zirconium oxide have demonstrated superior performance in promoting the aminolysis reaction, ensuring high conversion of the malonamide intermediate before the subsequent acylation step occurs.
Q: How does this method impact impurity profiles compared to traditional routes?
A: Traditional methods often suffer from incomplete coupling, leading to unreacted diazepane reacting with pivaloyl chloride to form difficult-to-remove byproducts. This novel catalytic approach drives the initial coupling to near-completion, effectively suppressing these side reactions and resulting in a cleaner crude product profile that simplifies downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pinoxaden Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is vital for the long-term sustainability of the agrochemical industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one-pot pinoxaden synthesis can be seamlessly translated from the laboratory to the manufacturing floor. We are committed to delivering high-purity pinoxaden that meets the rigorous demands of global regulatory agencies, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Our facility is equipped to handle the specific requirements of nano-catalyzed reactions, ensuring consistent quality and batch-to-batch reproducibility.
We invite you to engage with our technical sales team to discuss how this advanced manufacturing technology can benefit your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic advantages of switching to this streamlined supply source. We encourage you to contact our technical procurement team today to obtain specific COA data and detailed route feasibility assessments tailored to your volume requirements, ensuring a partnership built on transparency, quality, and mutual growth.
