Scalable Production of High-Purity Aminomethylbenzoic Acid via Optimized Chlorination Pathway
Introduction to Advanced Aminomethylbenzoic Acid Synthesis
The pharmaceutical industry continuously demands higher purity intermediates with robust supply chains, particularly for critical hemostatic agents. Patent CN112047849A introduces a groundbreaking preparation method for aminomethylbenzoic acid, a vital precursor for tranexamic acid and a standalone procoagulant medicine. This technology addresses the longstanding challenges of safety and purity in chemical synthesis by utilizing a streamlined two-step route starting from p-toluic acid. Unlike conventional methods that rely on hazardous nitro reductions or expensive cyanide coupling, this novel approach employs a controlled radical chlorination followed by a catalytic ammonolysis. The result is a product with exceptional purity exceeding 99.9% and single impurity content below 0.1%, achieved with an overall yield surpassing 63%. For R&D directors and procurement specialists, this represents a significant leap forward in process reliability and cost-efficiency.
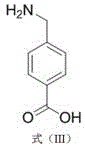
The strategic value of this patent lies in its ability to bypass toxic reagents and noble metal catalysts, which are often bottlenecks in supply chain continuity. By focusing on readily available starting materials and optimizing reaction parameters such as temperature and catalyst loading, the process ensures minimal environmental impact while maximizing output. This aligns perfectly with modern green chemistry principles and the rigorous quality standards required for active pharmaceutical ingredients (APIs) and their intermediates. As we delve deeper into the technical specifics, it becomes clear why this method is poised to become the industry standard for reliable aminomethylbenzoic acid supplier operations globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aminomethylbenzoic acid has been plagued by significant technical and safety drawbacks that hinder efficient commercial scale-up of complex pharmaceutical intermediates. One prevalent method involves the reduction of p-nitrobenzoic acid using iron powder and hydrochloric acid, followed by diazotization and cyanide coupling. This multi-step sequence is not only lengthy but also introduces severe safety hazards due to the handling of explosive diazonium salts and highly toxic cyanides. Another common approach utilizes direct chlorination of p-toluic acid with chlorine gas; however, this often suffers from low efficiency and the uncontrollable generation of dichloro-byproducts, making the reaction system difficult to treat and purify. Furthermore, alternative routes involving p-cyanobenzyl chloride or bromination with hydrogen bromide face issues with raw material availability and the generation of large amounts of solid salt waste, respectively. These legacy processes impose heavy burdens on waste treatment facilities and inflate production costs through low yields and complex purification requirements.
The Novel Approach
In stark contrast, the patented method offers a streamlined and economically superior pathway by leveraging the stability and abundance of p-toluic acid. 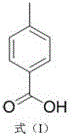 The process initiates with a highly selective chlorination reaction where p-toluic acid reacts with a chlorinating agent, preferably sulfuryl chloride, under the influence of a radical initiator. This step converts the methyl group into a chloromethyl group with high precision, avoiding the over-chlorination issues seen in gas-phase chlorination. The resulting intermediate, p-chloromethylbenzoic acid, is then subjected to a mild ammonolysis reaction using ammonia water and a specific catalyst. This two-step sequence eliminates the need for dangerous nitro reductions or expensive noble metals, drastically simplifying the workflow. The ability to carry the intermediate directly into the second step without intermediate purification further reduces solvent consumption and processing time, offering substantial cost savings in pharmaceutical intermediates manufacturing while maintaining a short synthetic route.
The process initiates with a highly selective chlorination reaction where p-toluic acid reacts with a chlorinating agent, preferably sulfuryl chloride, under the influence of a radical initiator. This step converts the methyl group into a chloromethyl group with high precision, avoiding the over-chlorination issues seen in gas-phase chlorination. The resulting intermediate, p-chloromethylbenzoic acid, is then subjected to a mild ammonolysis reaction using ammonia water and a specific catalyst. This two-step sequence eliminates the need for dangerous nitro reductions or expensive noble metals, drastically simplifying the workflow. The ability to carry the intermediate directly into the second step without intermediate purification further reduces solvent consumption and processing time, offering substantial cost savings in pharmaceutical intermediates manufacturing while maintaining a short synthetic route.
Mechanistic Insights into Radical Chlorination and Catalytic Ammonolysis
The core of this technological breakthrough lies in the precise control of radical chemistry during the first step. The reaction utilizes a free radical initiator, such as azobisisobutyronitrile (AIBN), to generate radicals that abstract hydrogen atoms from the methyl group of p-toluic acid. This creates a benzylic radical which subsequently reacts with the chlorinating agent to form the carbon-chlorine bond. Maintaining the reaction temperature strictly between 50-60°C is critical; this thermal window ensures sufficient energy for radical propagation while preventing thermal decomposition of the initiator or uncontrolled side reactions that could lead to polychlorinated byproducts. The choice of solvent, typically dichloroethane, plays a pivotal role in stabilizing the radical species and facilitating heat transfer. By optimizing the mass ratio of p-toluic acid to chlorinating agent to approximately 1:2, the process maximizes the conversion rate while minimizing the formation of dichloro-impurities, which are notoriously difficult to separate in downstream processing.
Following the chlorination, the mechanism shifts to nucleophilic substitution during the ammonolysis phase. The intermediate p-chloromethylbenzoic acid 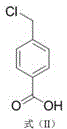 reacts with ammonia water in the presence of a catalyst like urotropine. Urotropine acts as a phase transfer catalyst or a nucleophilic promoter, enhancing the reactivity of the ammonia towards the benzylic chloride. The reaction is conducted in a mixed solvent system of methanol and water, which ensures good solubility for both the organic intermediate and the aqueous ammonia. The temperature is again carefully controlled at 50-60°C to drive the substitution to completion without degrading the sensitive amino group. This mechanistic precision is what allows the final product to achieve a purity of greater than 99.9% with single impurities below 0.1%, as the specific catalyst system suppresses competing hydrolysis or elimination reactions that typically plague ammonolysis processes.
reacts with ammonia water in the presence of a catalyst like urotropine. Urotropine acts as a phase transfer catalyst or a nucleophilic promoter, enhancing the reactivity of the ammonia towards the benzylic chloride. The reaction is conducted in a mixed solvent system of methanol and water, which ensures good solubility for both the organic intermediate and the aqueous ammonia. The temperature is again carefully controlled at 50-60°C to drive the substitution to completion without degrading the sensitive amino group. This mechanistic precision is what allows the final product to achieve a purity of greater than 99.9% with single impurities below 0.1%, as the specific catalyst system suppresses competing hydrolysis or elimination reactions that typically plague ammonolysis processes.
How to Synthesize Aminomethylbenzoic Acid Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high yield. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions. The key to success lies in the sequential addition of reagents and the maintenance of specific thermal profiles throughout the reaction timeline. Operators must ensure that the chlorination step is allowed to proceed until the residual raw material content drops below 5% before proceeding to solvent recovery. The subsequent ammonolysis step requires extended stirring times of 15 to 20 hours to ensure complete conversion of the intermediate. Detailed standardized operating procedures regarding reagent grades, addition rates, and cooling protocols are essential for transferring this laboratory success to pilot and commercial plants.
- Dissolve p-toluic acid and a radical initiator (e.g., AIBN) in dichloroethane, heat to 50-60°C, and add sulfuryl chloride dropwise to form p-chloromethylbenzoic acid.
- Concentrate the reaction mixture under reduced pressure to recover solvent and obtain the solid intermediate without further purification.
- Dissolve the intermediate in methanol/water, add urotropine and ammonia water, heat to 50-60°C for ammonolysis, then cool and filter to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic advantages beyond mere chemical yield. The elimination of noble metal catalysts and highly toxic reagents like cyanides removes significant regulatory hurdles and disposal costs associated with hazardous waste management. This shift not only lowers the direct cost of goods sold but also mitigates the risk of production stoppages due to environmental compliance audits. Furthermore, the reliance on commodity chemicals such as p-toluic acid and sulfuryl chloride ensures a stable and resilient supply chain, reducing vulnerability to market fluctuations that often affect specialty reagents. The high recovery rate of solvents, exceeding 70% through vacuum concentration, further contributes to a leaner manufacturing model with reduced raw material consumption.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the synthetic route and the avoidance of expensive purification steps. By eliminating the need for noble metal catalysts, the process removes the costly requirement for metal scavenging and residue testing, which are mandatory for pharmaceutical grade products. Additionally, the ability to use the crude intermediate directly in the second step without isolation significantly reduces solvent usage, energy consumption for drying, and labor hours. The high overall yield of over 63% means that less starting material is wasted, directly improving the margin per kilogram of finished product and allowing for more competitive pricing strategies in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available, bulk-grade raw materials. Unlike methods dependent on p-cyanobenzyl chloride, which can be subject to supply shortages and price volatility, p-toluic acid is a commodity chemical produced at massive scales globally. This abundance ensures that production schedules can be maintained without interruption, even during periods of market tightness. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain inconsistencies and ensuring consistent delivery timelines to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for large-scale industrial production, as evidenced by successful examples at the 100 kg scale. The reaction conditions operate at atmospheric pressure and moderate temperatures, removing the need for complex high-pressure reactors that limit batch sizes. From an environmental perspective, the absence of heavy metal waste and the high efficiency of solvent recovery significantly reduce the facility's environmental footprint. This alignment with green chemistry principles facilitates easier permitting and long-term operational sustainability, making it an ideal candidate for expanding production capacity to meet growing global demand for hemostatic agents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, purity, and scalability. Understanding these aspects is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios or for procurement teams assessing supplier capabilities.
Q: Why is the chlorination-ammonolysis route safer than traditional nitro reduction methods?
A: Traditional methods often involve reducing nitro groups with iron powder or handling unstable diazonium salts, which pose significant explosion risks and generate heavy metal waste. The patented chlorination-ammonolysis route avoids nitro compounds entirely, utilizing stable p-toluic acid and controlled radical chemistry, thereby eliminating high-energy intermediates and reducing potential safety hazards in large-scale manufacturing.
Q: How does this process achieve >99.9% purity without complex purification?
A: The process achieves exceptional purity through precise control of reaction conditions, specifically maintaining temperatures between 50-60°C and using optimized catalyst ratios. This minimizes the formation of dichloro-byproducts and other side reactions. Furthermore, the intermediate p-chloromethylbenzoic acid is carried forward directly without purification, yet the final crystallization step effectively separates the target aminomethylbenzoic acid, ensuring single impurity levels remain below 0.1%.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the method is explicitly designed for industrial scalability. It utilizes common, inexpensive raw materials like p-toluic acid and sulfuryl chloride, and the solvents used (dichloroethane, methanol) have high recovery rates exceeding 70%. The patent demonstrates successful operation at the 100 kg scale with consistent yields over 63%, proving its viability for commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminomethylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our capability to implement the advanced chlorination-ammonolysis route described in CN112047849A allows us to offer aminomethylbenzoic acid with unmatched consistency and purity, catering to the exacting needs of the pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and cost-effective chemical solutions available in the market.
