Advanced Chiral Synthesis of 6-Nitro-S-(-)-indoline-2-carboxylic acid for Commercial Scale-up
Advanced Chiral Synthesis of 6-Nitro-S-(-)-indoline-2-carboxylic acid for Commercial Scale-up
In the rapidly evolving landscape of chiral pharmaceutical intermediates, the efficient production of optically pure building blocks remains a critical bottleneck for drug development pipelines. Patent CN101696185A introduces a transformative synthetic methodology for 6-nitro-S-(-)-indoline-2-carboxylic acid, a pivotal precursor in the manufacture of triptan-class migraine medications and other bioactive compounds. Unlike traditional approaches that struggle with isomeric purity and expensive starting materials, this innovation leverages the abundant chiral pool of L-phenylalanine to achieve superior stereochemical control. By systematically addressing the limitations of prior art through a robust three-step sequence involving nitration, bromination, and catalytic cyclization, this technology offers a viable pathway for commercial scale-up of complex pharmaceutical intermediates. As a leading entity in fine chemical manufacturing, we recognize the immense value this process brings to reducing lead time for high-purity intermediates while maintaining rigorous quality standards essential for regulatory compliance.
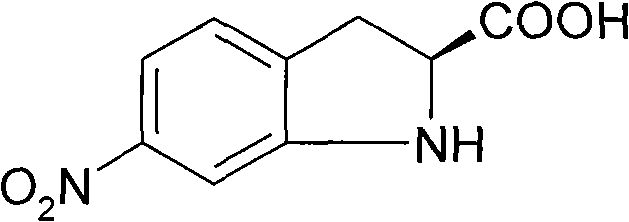
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-nitroindoline-2-carboxylic acid has been plagued by significant chemical and economic inefficiencies that hinder large-scale production capabilities. Conventional literature methods, such as those described in Synthesis (2002), typically rely on the direct nitration of indoline-2-carboxylic acid using mixed acids. This approach suffers from a fundamental lack of regioselectivity, invariably producing a complex mixture of 5-nitro and 6-nitro isomers that are notoriously difficult to separate due to their similar physicochemical properties. Furthermore, these traditional routes often yield racemic products, necessitating additional, costly, and yield-depleting resolution steps to isolate the biologically active S-enantiomer. The reliance on expensive indoline-2-carboxylic acid as a starting material further exacerbates the cost structure, making the process economically unviable for high-volume pharmaceutical intermediates manufacturing. These cumulative drawbacks result in low overall yields, excessive waste generation, and extended production timelines that fail to meet the demands of modern supply chains.
The Novel Approach
The methodology disclosed in CN101696185A represents a paradigm shift by utilizing optically pure L-phenylalanine as a readily available and cost-effective chiral source. This strategic choice eliminates the need for post-synthesis chiral resolution, as the stereochemistry is preserved throughout the synthetic sequence from the abundant amino acid feedstock. The process employs a highly controlled nitration followed by a specific bromination to install the necessary functional groups with precise regiocontrol, effectively bypassing the isomer mixture problem inherent in direct nitration strategies. The final cyclization step, facilitated by transition metal catalysts, efficiently closes the indoline ring to deliver the target molecule with exceptional enantiomeric excess. This streamlined approach not only simplifies the operational workflow but also drastically reduces the consumption of raw materials and solvents, aligning perfectly with the industry's drive towards greener and more sustainable chemical processes. By adopting this route, manufacturers can achieve cost reduction in pharmaceutical intermediates manufacturing while ensuring a consistent supply of high-quality chiral building blocks.
Mechanistic Insights into Cu/Pd-Catalyzed Cyclization
The core of this synthetic innovation lies in the intricate interplay of electrophilic aromatic substitution and transition metal-catalyzed ring closure. The initial nitration of L-phenylalanine proceeds via the generation of the nitronium ion in concentrated sulfuric acid, which selectively attacks the para-position relative to the alanine side chain due to steric and electronic directing effects. Subsequent bromination, utilizing reagents like N-bromosuccinimide (NBS) in an acidic medium, installs a bromine atom ortho to the amino group, setting the stage for the crucial intramolecular coupling. The final cyclization is a sophisticated nucleophilic substitution where the amine nitrogen attacks the benzylic carbon bearing the bromine leaving group. This step is significantly accelerated by the presence of copper or palladium salts, which likely function by coordinating with the halogen and the amine, lowering the activation energy for ring closure and preventing side reactions such as elimination or polymerization. Understanding these mechanistic nuances is vital for optimizing reaction parameters such as temperature, solvent polarity, and catalyst loading to maximize throughput.
Impurity control is rigorously managed through the specific selection of reaction conditions and purification techniques outlined in the patent. The use of low temperatures during the nitration and bromination steps minimizes oxidative degradation of the sensitive amino acid backbone and prevents poly-nitration or poly-bromination side products. During the workup phases, precise pH adjustments allow for the selective precipitation of intermediates, effectively washing away inorganic salts and acidic byproducts before they can contaminate subsequent steps. The final recrystallization from ethyl acetate and petroleum ether serves as a powerful polishing step, removing trace metal catalysts and any remaining organic impurities to ensure the final product meets stringent purity specifications. This multi-layered approach to impurity management ensures that the resulting 6-nitro-S-(-)-indoline-2-carboxylic acid possesses the high optical purity required for downstream API synthesis, thereby reducing the risk of batch failures and regulatory delays.
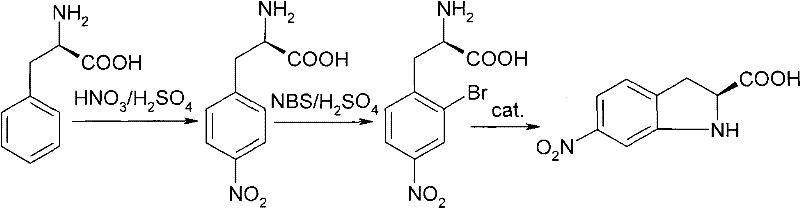
How to Synthesize 6-Nitro-S-(-)-indoline-2-carboxylic acid Efficiently
Executing this synthesis requires strict adherence to the optimized parameters regarding temperature control and reagent stoichiometry to ensure safety and reproducibility. The process begins with the careful addition of L-phenylalanine to pre-cooled mixed acid, followed by a controlled bromination phase where exothermicity must be managed to prevent thermal runaway. The final cyclization demands precise catalyst selection and solvent choice to drive the equilibrium towards the desired cyclic product. For detailed operational protocols, safety data, and specific equipment requirements, please refer to the standardized guide below which outlines the critical process parameters derived from the patent examples.
- Nitration of L-Phenylalanine using mixed acid (H2SO4/HNO3) at controlled low temperatures (-20 to 50°C) to form 4-nitro-L-phenylalanine.
- Bromination of the nitro-intermediate using NBS or similar reagents in concentrated sulfuric acid to yield 2-bromo-4-nitro-L-phenylalanine.
- Intramolecular cyclization using a base and copper or palladium catalyst in solvents like water or DMF to close the indoline ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthetic route offers substantial advantages by decoupling production costs from volatile specialty chemical markets. By anchoring the synthesis on L-phenylalanine, a commodity fermentation product with stable global availability, manufacturers can mitigate supply chain risks associated with scarce or proprietary starting materials. The elimination of chiral resolution steps not only simplifies the process flow but also significantly enhances the overall mass efficiency, meaning less raw material is required to produce the same amount of active intermediate. This efficiency translates directly into improved margin structures and a more competitive pricing model for downstream partners. Furthermore, the robustness of the reaction conditions allows for flexible manufacturing schedules, enabling suppliers to respond more agilely to fluctuating market demands without compromising on product quality or delivery reliability.
- Cost Reduction in Manufacturing: The substitution of expensive indoline precursors with bulk-grade L-phenylalanine fundamentally alters the cost basis of production. By removing the need for complex chromatographic separations or enzymatic resolutions typically required to purify racemic mixtures, the process significantly lowers operational expenditures related to labor, solvents, and waste disposal. The high atom economy of the cyclization step further contributes to cost optimization by maximizing the conversion of input materials into the final valuable product. These cumulative efficiencies allow for a leaner manufacturing model that can sustain lower price points while maintaining healthy profitability, offering a distinct competitive edge in the global marketplace.
- Enhanced Supply Chain Reliability: Utilizing widely available amino acid feedstocks ensures a resilient supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. The simplified three-step sequence reduces the number of unit operations and intermediate handling stages, thereby decreasing the probability of production delays caused by equipment failure or logistical complexities. This streamlined workflow facilitates faster turnaround times from order placement to shipment, providing customers with greater predictability in their own production planning. Additionally, the scalability of the process from laboratory to industrial reactors ensures that supply volumes can be ramped up seamlessly to support clinical trial phases through to commercial launch without the need for extensive process re-engineering.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing the use of hazardous reagents and reducing the generation of toxic byproducts. The ability to perform key steps in aqueous or common organic solvents simplifies waste treatment protocols and lowers the environmental footprint of the manufacturing facility. High yields and selectivity mean that less waste is generated per kilogram of product, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the supply chain partners, appealing to end-users who prioritize eco-friendly sourcing strategies in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and sourcing of this specific chiral intermediate. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific drug development or manufacturing pipeline.
Q: What is the primary advantage of using L-Phenylalanine as a starting material?
A: Using optically pure L-Phenylalanine ensures high enantiomeric excess (ee > 99.8%) in the final product without the need for difficult chiral resolution steps required by racemic synthesis methods.
Q: How does this method improve regioselectivity compared to direct nitration?
A: Direct nitration of indoline-2-carboxylic acid produces a difficult-to-separate mixture of 5-nitro and 6-nitro isomers. This patented route utilizes specific bromination positioning to ensure exclusive formation of the 6-nitro derivative.
Q: What catalysts are suitable for the cyclization step?
A: The patent specifies that copper salts (such as cuprous bromide or copper chloride) and palladium salts (such as palladium chloride) are effective catalysts for promoting the ring-closure reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Nitro-S-(-)-indoline-2-carboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we combine deep technical expertise with robust manufacturing capabilities to deliver high-value chiral intermediates to the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in early-stage R&D or full-scale commercialization. We operate under stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 6-nitro-S-(-)-indoline-2-carboxylic acid meets the highest standards of optical purity and chemical integrity. Our commitment to quality assurance means that you can rely on us as a consistent partner who understands the critical nature of chiral fidelity in drug synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this L-phenylalanine-based route impacts your bottom line compared to traditional methods. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique project needs, allowing us to demonstrate our capability as a trusted partner in your success.
