Advanced Synthesis of 6-Nitro-S-(-)-Indoline-2-Carboxylic Acid for Commercial Scale-Up
Introduction to High-Purity Chiral Intermediate Manufacturing
The pharmaceutical industry continuously demands robust synthetic routes for optically active intermediates that serve as the backbone for next-generation therapeutics. Patent CN101696185A introduces a groundbreaking methodology for the synthesis of 6-nitro-S-(-)-indoline-2-carboxylic acid, a critical building block in the development of chiral drugs. This innovative approach leverages the chiral pool strategy, utilizing optically pure L-phenylalanine as the foundational starting material to ensure exceptional stereochemical control throughout the synthesis. Unlike traditional methods that often struggle with racemization or complex separation processes, this protocol achieves high enantiomeric excess (ee > 99.8%) while maintaining operational simplicity. The structural integrity of the target molecule, as depicted below, is crucial for its biological activity in downstream applications.
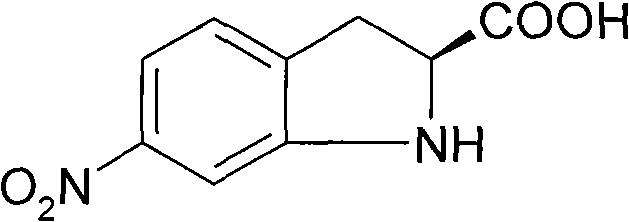
For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this patent is essential. The process not only addresses the purity challenges associated with earlier synthetic routes but also significantly optimizes the cost structure by employing commodity-grade amino acids. By integrating this technology, manufacturers can secure a stable supply of high-purity 6-nitro-S-(-)-indoline-2-carboxylic acid, thereby reducing lead time for high-purity pharmaceutical intermediates and enhancing the overall efficiency of drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-nitro-indoline derivatives has been plagued by significant technical hurdles that impede large-scale production. Prior art, such as the method described in Synthesis (2002), relies on the direct nitration of indoline-2-carboxylic acid using mixed acids. This conventional approach suffers from poor regioselectivity, invariably producing a difficult-to-separate mixture of 5-nitro and 6-nitro isomers. Furthermore, the starting material, indoline-2-carboxylic acid, is considerably more expensive and less readily available than simple amino acids. Perhaps most critically, these older methods typically yield racemic products, necessitating additional, costly, and yield-reducing resolution steps to isolate the desired S-enantiomer. These factors collectively result in a process that is economically unviable for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN101696185A revolutionizes the production landscape by adopting a linear, three-step sequence starting from L-phenylalanine. As illustrated in the reaction scheme below, this route strategically functionalizes the aromatic ring prior to ring closure, thereby exerting precise control over the substitution pattern. The initial nitration occurs para to the alanine side chain, followed by ortho-bromination, setting the stage for an intramolecular cyclization that forms the indoline core. This logical progression eliminates the formation of regioisomers entirely. Moreover, because the chiral center originates from natural L-phenylalanine, the optical purity is inherently locked in, bypassing the need for chiral resolution. This represents a paradigm shift in cost reduction in chiral API manufacturing, offering a streamlined path from bulk chemicals to high-value intermediates.
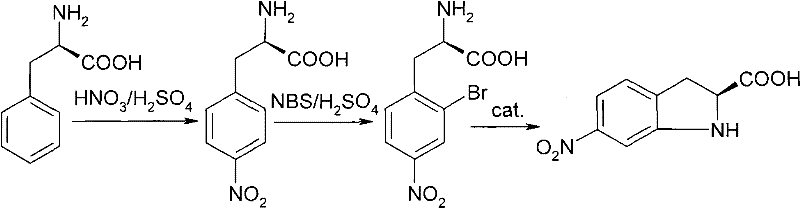
Mechanistic Insights into Fe/Cu-Catalyzed Cyclization and Chiral Retention
The success of this synthesis hinges on the meticulous preservation of stereochemistry during electrophilic aromatic substitutions. In the first step, nitration of L-phenylalanine is conducted in a mixed acid system (H2SO4/HNO3) at controlled low temperatures ranging from -20°C to 5°C. Under these conditions, the protonated amino group directs the nitronium ion primarily to the para-position, yielding 4-nitro-L-phenylalanine. The low temperature is critical; it suppresses the rate of racemization at the alpha-carbon, which could otherwise occur via enolization in strong acid. The subsequent bromination step utilizes reagents like N-bromosuccinimide (NBS) or potassium bromate in concentrated sulfuric acid. Here, the amino group, now fully protonated, acts as a meta-director, but the steric and electronic environment favors bromination ortho to the alkyl side chain due to the specific activation of the ring, resulting in 2-bromo-4-nitro-L-phenylalanine with high fidelity.
The final cyclization step is a masterpiece of catalytic efficiency, transforming the linear precursor into the bicyclic indoline system. This transformation involves an intramolecular nucleophilic substitution where the amine nitrogen attacks the carbon bearing the bromine atom. To facilitate this energetically demanding ring closure, the patent employs transition metal catalysts such as copper salts (e.g., CuBr, CuCl) or palladium salts in the presence of a base like potassium carbonate or sodium hydroxide. The base deprotonates the ammonium species, generating the free amine nucleophile required for the attack. The metal catalyst likely coordinates with the halogen, weakening the C-Br bond and lowering the activation energy for the cyclization. This mechanism ensures that the ring closes rapidly and cleanly without disturbing the adjacent chiral center, delivering the final 6-nitro-S-(-)-indoline-2-carboxylic acid with an ee value exceeding 99.8%, demonstrating exceptional impurity control mechanisms.
How to Synthesize 6-Nitro-S-(-)-Indoline-2-Carboxylic Acid Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters defined in the patent to maximize yield and optical purity. The process begins with the careful addition of L-phenylalanine to pre-cooled mixed acid, followed by a controlled workup involving neutralization and recrystallization to isolate the nitro-intermediate. The bromination step similarly demands temperature control to manage exotherms and ensure selective mono-bromination. Finally, the cyclization is performed in polar solvents such as water, DMF, or DMSO, depending on the specific catalyst system chosen. While the general workflow is straightforward, precise control of pH during workup and solvent selection during extraction are vital for obtaining the product in high purity. For detailed operational protocols, please refer to the standardized guide below.
- Nitration of L-phenylalanine using mixed acid (HNO3/H2SO4) at low temperatures to form 4-nitro-L-phenylalanine.
- Bromination of the nitro-intermediate using NBS or bromates in concentrated sulfuric acid to yield 2-bromo-4-nitro-L-phenylalanine.
- Catalytic cyclization using copper or palladium salts with a base to close the indoline ring and obtain the final chiral acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that directly impact the bottom line and supply chain resilience. By shifting the starting material from specialized indoline derivatives to L-phenylalanine, a globally traded commodity amino acid, manufacturers can drastically reduce raw material costs and mitigate supply risks associated with niche precursors. The elimination of chiral resolution steps not only simplifies the process flow but also removes the substantial yield losses typically associated with separating enantiomers. This efficiency translates into a more predictable and robust supply chain, ensuring consistent availability of this critical intermediate for downstream drug synthesis. Furthermore, the use of common inorganic reagents and solvents facilitates easier regulatory compliance and waste management.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the substitution of expensive starting materials with low-cost bulk chemicals. L-phenylalanine is produced on a massive scale for the food and feed industries, ensuring a stable and competitive price point compared to synthetic indoline derivatives. Additionally, the high overall yield reported in the patent examples (up to 65% over three steps) means less raw material is wasted per kilogram of final product. The avoidance of chromatographic purification, relying instead on crystallization and extraction, further lowers the operational expenditure (OPEX) by reducing solvent consumption and processing time, leading to substantial cost savings in the final API production.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for complex intermediates. This synthesis democratizes production by utilizing widely available reagents such as sulfuric acid, nitric acid, and NBS, which are accessible from multiple global vendors. The robustness of the three-step sequence allows for flexible manufacturing scheduling and rapid scale-up capabilities. By reducing the complexity of the synthesis, the risk of batch failures due to hard-to-control variables is minimized. This reliability ensures that procurement managers can secure long-term contracts with confidence, knowing that the production of high-purity pharmaceutical intermediates will not be disrupted by raw material shortages or technical bottlenecks.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden inefficiencies, but this route is designed for scalability. The reactions utilize standard unit operations such as stirred tank reactors for nitration and bromination, and heated vessels for cyclization, which are commonplace in fine chemical facilities. The workup procedures involve simple phase separations and crystallizations, avoiding the need for specialized equipment like preparative HPLC. From an environmental standpoint, the process generates less hazardous waste compared to resolution-heavy routes. The ability to recycle solvents like ethyl acetate and petroleum ether, combined with the high atom economy of the cyclization step, aligns with modern green chemistry principles, facilitating smoother environmental impact assessments and regulatory approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN101696185A, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs.
Q: What is the primary advantage of using L-phenylalanine over indoline-2-carboxylic acid?
A: L-phenylalanine is a readily available, inexpensive chiral pool starting material that preserves optical purity (ee > 99.8%), whereas direct nitration of indoline-2-carboxylic acid yields racemic mixtures requiring difficult resolution.
Q: How is the chiral integrity maintained during the harsh nitration and bromination steps?
A: The reaction conditions are carefully controlled (e.g., low temperature nitration at -20°C to 5°C) to prevent racemization at the alpha-carbon, ensuring the final product retains high enantiomeric excess.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes common reagents like sulfuric acid and NBS and avoids complex chromatographic separations, making it highly suitable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Nitro-S-(-)-Indoline-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest standards. Our expertise in chiral synthesis allows us to navigate the complexities of this specific patent, guaranteeing the optical purity and chemical integrity required for your downstream applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us be your partner in bringing innovative therapies to market faster and more efficiently.
