Advanced Chiral Pool Synthesis of 6-Nitro-S-(-)-Indoline-2-Carboxylic Acid for Scalable Pharma Production
The pharmaceutical industry continuously seeks robust pathways for constructing complex chiral scaffolds, particularly for the synthesis of bioactive indoline derivatives. Patent CN101696185B discloses a highly efficient synthesizing method for 6-nitro-S-(-)-indoline-2-carboxylic acid, a critical chiral building block used in the development of various optically active drugs. Unlike traditional approaches that struggle with regioselectivity and racemization, this invention leverages a chiral pool strategy starting from optically pure L-phenylalanine. The process involves a sequential three-step transformation comprising nitration, bromination, and cyclization, ultimately delivering the target molecule with exceptional enantiomeric excess exceeding 99.8%. 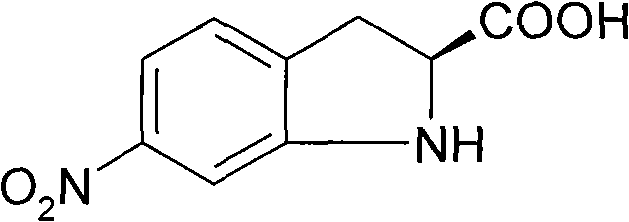 For procurement specialists and R&D directors evaluating reliable pharmaceutical intermediate supplier options, understanding the mechanistic superiority of this route is essential for securing long-term supply chain stability and cost efficiency.
For procurement specialists and R&D directors evaluating reliable pharmaceutical intermediate supplier options, understanding the mechanistic superiority of this route is essential for securing long-term supply chain stability and cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitro-substituted indoline carboxylic acids has been plagued by significant chemical and economic inefficiencies. Prior art, such as the method described in Synthesis (2002), typically involves the direct nitration of indoline-2-carboxylic acid using mixed acids. This conventional pathway suffers from poor regioselectivity, invariably producing a complex mixture of 5-nitro and 6-nitro isomers that are notoriously difficult to separate via standard crystallization or chromatography techniques. Furthermore, because the starting indoline scaffold is often racemic or requires expensive chiral resolution post-synthesis, the overall process yield is drastically compromised. The necessity for multiple purification steps to isolate the correct isomer and enantiomer not only inflates manufacturing costs but also generates substantial chemical waste, creating environmental compliance burdens for large-scale facilities.
The Novel Approach
In stark contrast, the methodology outlined in CN101696185B revolutionizes the production landscape by utilizing L-phenylalanine as a readily available, optically pure starting material. This chiral pool approach inherently transfers the stereochemistry from the amino acid to the final indoline product, effectively bypassing the need for difficult chiral resolution steps entirely. By strategically introducing the nitro group prior to ring closure, the synthesis avoids the formation of regio-isomeric mixtures common in direct indoline nitration. The subsequent bromination and metal-catalyzed cyclization steps are optimized to proceed under mild to moderate conditions, utilizing cost-effective reagents like N-bromosuccinimide (NBS) and copper salts. This streamlined sequence not only simplifies the operational workflow but also significantly enhances the overall atom economy, making it an ideal candidate for cost reduction in chiral intermediate manufacturing.
Mechanistic Insights into Copper-Catalyzed Cyclization
The cornerstone of this synthetic route lies in the final intramolecular cyclization step, which constructs the five-membered nitrogen-containing ring of the indoline core. Mechanistically, this transformation proceeds via a nucleophilic aromatic substitution or a metal-catalyzed coupling pathway, depending on the specific catalyst system employed. When copper salts such as cuprous bromide or cupric chloride are utilized, the metal center likely coordinates with the amine nitrogen and the ortho-bromine atom, facilitating the displacement of the halide through an oxidative addition-reductive elimination cycle. This catalytic activation lowers the energy barrier for ring closure, allowing the reaction to proceed efficiently even in aqueous or polar aprotic solvents like DMF and DMSO. The presence of a base, such as potassium carbonate or sodium hydroxide, is critical for deprotonating the amine, thereby increasing its nucleophilicity and driving the equilibrium towards the cyclized product.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed thermal cyclizations. The use of transition metal catalysts allows for precise control over reaction kinetics, minimizing side reactions such as debromination or polymerization that often occur under harsh thermal conditions. Furthermore, because the chirality is locked in the alpha-carbon of the phenylalanine backbone throughout the nitration and bromination steps, the cyclization proceeds with retention of configuration. This ensures that the final 6-nitro-S-(-)-indoline-2-carboxylic acid maintains an enantiomeric purity greater than 99%, a specification that is paramount for regulatory approval in pharmaceutical applications. The ability to achieve such high optical purity without recrystallization of the final salt form represents a significant technical breakthrough for high-purity pharmaceutical intermediate production.
How to Synthesize 6-Nitro-S-(-)-Indoline-2-Carboxylic Acid Efficiently
The practical execution of this synthesis requires careful attention to temperature control and reagent stoichiometry, particularly during the exothermic nitration and bromination phases. The patent details a robust protocol where L-phenylalanine is first nitrated in a mixed acid system at sub-zero temperatures to prevent oxidation, followed by selective bromination at the ortho-position relative to the amine. The final cyclization is the most versatile step, accommodating a range of solvents and bases to suit specific plant capabilities. While the general workflow is straightforward, adherence to the specific molar ratios and pH adjustments described in the intellectual property is crucial for maximizing yield. For detailed operational parameters, safety data, and exact stoichiometric calculations required for GMP manufacturing, please refer to the standardized synthesis guide below.
- Nitration of L-Phenylalanine using mixed acid (sulfuric and nitric acid) at controlled low temperatures to yield 4-nitro-L-phenylalanine.
- Bromination of the nitro-intermediate in concentrated sulfuric acid using reagents like NBS or sodium bromate to form 2-bromo-4-nitro-L-phenylalanine.
- Intramolecular cyclization of the bromo-intermediate using a base and copper or palladium catalyst in solvents like water or DMF to close the indoline ring.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this patented synthesis route translates directly into tangible strategic benefits regarding cost structure and supply reliability. By shifting the starting material from expensive, specialized indoline derivatives to commodity-grade L-phenylalanine, manufacturers can decouple their production costs from the volatile pricing of niche heterocyclic building blocks. The elimination of chiral resolution steps, which typically consume more than half of the processing time and solvent volume in traditional routes, results in a drastically simplified workflow. This reduction in unit operations not only lowers utility consumption and labor costs but also shortens the overall production cycle time, enabling faster response to market demand fluctuations without compromising quality standards.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the utilization of bulk chemicals. L-phenylalanine is produced on a multi-ton scale globally for the food and feed industries, ensuring a stable and low-cost supply compared to custom-synthesized indoline precursors. Furthermore, the avoidance of chiral resolution eliminates the loss of 50% of the material (the unwanted enantiomer) that occurs in racemic syntheses, effectively doubling the theoretical yield from the chiral source. The use of recoverable solvents like ethyl acetate and petroleum ether in the final workup further contributes to substantial cost savings by minimizing waste disposal fees and solvent purchase expenses.
- Enhanced Supply Chain Reliability: Relying on a chiral pool synthesis mitigates the risk of supply disruptions associated with single-source suppliers of complex intermediates. Since the raw materials (sulfuric acid, nitric acid, L-phenylalanine) are widely available from multiple global vendors, the manufacturing process is resilient to regional shortages. Additionally, the robustness of the reaction conditions, which tolerate a wide range of temperatures and concentrations, ensures consistent batch-to-batch quality. This reliability is critical for maintaining continuous production schedules and meeting the stringent delivery timelines required by downstream pharmaceutical clients who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing standard reactor equipment capable of handling acidic and exothermic reactions. The final cyclization step can be performed in water or green solvents, significantly reducing the environmental footprint compared to processes relying heavily on chlorinated hydrocarbons. By minimizing the generation of hazardous waste and avoiding the use of heavy metals in stoichiometric quantities (using them instead as trace catalysts), the facility can maintain compliance with increasingly strict environmental regulations while achieving high throughput volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this chiral intermediate. These insights are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the technology's capabilities. Understanding these nuances helps stakeholders make informed decisions about integrating this material into their drug development pipelines.
Q: Why is the chiral pool method superior to direct nitration for this intermediate?
A: Direct nitration of indoline-2-carboxylic acid produces a difficult-to-separate mixture of 5-nitro and 6-nitro isomers and yields a racemic product requiring complex resolution. The chiral pool method using L-phenylalanine ensures high optical purity (>99% ee) from the start and avoids regio-isomer separation issues.
Q: What are the key cost drivers in this synthesis route?
A: The primary cost advantage comes from using L-phenylalanine, a commercially abundant and inexpensive amino acid, rather than expensive pre-formed indoline scaffolds. Additionally, the elimination of chiral resolution steps significantly reduces downstream processing costs.
Q: Can this process be scaled for industrial production?
A: Yes, the patent describes robust conditions using common reagents like mixed acid and NBS, with reaction temperatures ranging from cryogenic to reflux. The use of water or polar aprotic solvents in the final cyclization step facilitates large-scale handling and waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Nitro-S-(-)-Indoline-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutics depends on the availability of high-quality, cost-effective building blocks. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering 6-nitro-S-(-)-indoline-2-carboxylic acid with stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and impurity profiles against the highest international standards. Our state-of-the-art facilities are equipped to handle the specific corrosive and exothermic nature of the nitration and bromination steps safely and efficiently.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from recent batches and comprehensive route feasibility assessments to demonstrate how our implementation of this patented technology can drive value for your organization. Let us be your partner in accelerating your drug development timeline with reliable, high-performance chemical solutions.
