Advanced Aqueous Hydrogenation Technology for High-Purity Biotin Intermediates
Advanced Aqueous Hydrogenation Technology for High-Purity Biotin Intermediates
The landscape of vitamin synthesis, particularly for high-value nutrients like Biotin (Vitamin B7), is constantly evolving towards greener and more efficient methodologies. A pivotal advancement in this domain is documented in Chinese Patent CN100369872C, which discloses a novel mild catalytic hydrogenation process tailored for thiazole ring systems. This technology addresses a critical bottleneck in the production of biotin intermediates: the selective reduction of carbon-carbon double bonds in the presence of sensitive functional groups. By shifting the reaction medium from traditional organic solvents to a controlled aqueous environment with a pH greater than 7, this process offers a robust pathway for manufacturing high-purity pharmaceutical intermediates. For R&D directors and process chemists, this represents a significant opportunity to optimize impurity profiles and streamline synthetic routes, ensuring that the final active ingredient meets stringent global regulatory standards while minimizing environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biotin involves the hydrogenation of unsaturated intermediates, such as thieno[3,4-d]imidazolyl-4-ylidenevaleric acid derivatives. As referenced in prior art like European Patent EP-B-0 273 270, these reactions were typically conducted in organic solvents such as isopropanol. While effective, these conventional protocols present several logistical and economic challenges for large-scale manufacturing. The reliance on volatile organic compounds (VOCs) necessitates extensive solvent recovery systems to meet environmental regulations, adding capital expenditure and operational complexity. Furthermore, when soluble palladium catalysts are employed to enhance reactivity, the downstream removal of residual metal becomes a arduous task, often requiring additional purification steps like polymer flocculation or specialized filtration to ensure the final product complies with heavy metal limits. These inefficiencies accumulate, leading to longer cycle times and higher production costs.
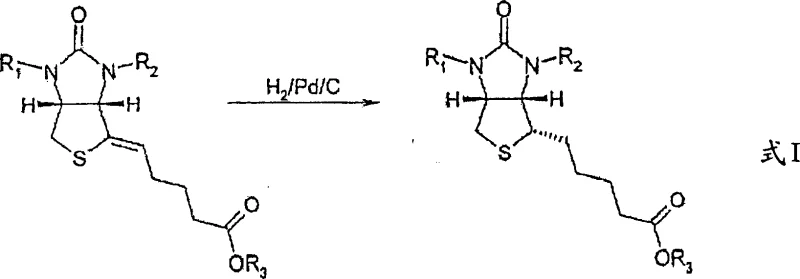
The Novel Approach
The methodology outlined in CN100369872C fundamentally reimagines the reaction environment by leveraging the acid-base properties of the substrate. Instead of forcing solubility through organic media, the inventors discovered that adjusting the pH of a protic solvent (preferably water) to a basic range (pH > 7, optimally 8.5 to 8.9) converts the carboxylic acid moiety of the intermediate into a soluble salt. This simple yet profound modification allows the use of water as the primary reaction medium. The process utilizes heterogeneous catalysts, specifically Palladium on Carbon (Pd/C), which can be easily filtered off post-reaction. This approach not only eliminates the safety hazards associated with large volumes of flammable organic solvents but also simplifies the workup procedure dramatically. The ability to perform the hydrogenation directly in the medium where the precursor was formed, or in simple aqueous alkali, removes the need for intermediate drying and solvent swapping, representing a major leap in process intensification.
Mechanistic Insights into pH-Controlled Aqueous Hydrogenation
The core innovation of this technology lies in the precise control of the reaction medium's acidity to manipulate substrate solubility and catalyst interaction. In the neutral or acidic state, the thienoimidazole valeric acid derivative is poorly soluble in water, which would hinder mass transfer and reaction kinetics in an aqueous phase. However, by introducing a weak base such as sodium carbonate or sodium hydroxide to raise the pH to approximately 8.7, the carboxylic acid group is deprotonated to form a carboxylate anion. This ionic species exhibits high solubility in water, creating a homogeneous reaction phase for the substrate while the solid Pd/C catalyst remains suspended. This pseudo-homogeneous condition ensures excellent contact between the hydrogen gas, the catalyst surface, and the dissolved substrate, facilitating rapid and selective reduction of the exocyclic double bond without affecting other sensitive functionalities like the ureido ring or protecting groups.
Furthermore, this mechanistic pathway offers superior control over stereoselectivity and impurity formation. The mild conditions (temperatures around 100°C and pressures of 3 to 5 bar) prevent thermal degradation of the delicate bicyclic ring system. The use of a heterogeneous catalyst in a basic aqueous medium minimizes side reactions such as hydrolysis of the ester or amide protecting groups, which might occur under harsher acidic or purely organic conditions. Post-reaction, the mechanism allows for a straightforward isolation strategy: acidifying the mixture to pH 6 reprotonates the carboxylate, rendering the product less soluble in water and amenable to extraction into organic solvents like xylene or ethyl acetate. This pH-switchable solubility acts as an intrinsic purification step, effectively separating the product from inorganic salts and water-soluble impurities, thereby enhancing the overall purity profile of the biotin intermediate.
How to Synthesize Hexahydro-thienoimidazole Derivatives Efficiently
Implementing this hydrogenation protocol requires careful attention to pH buffering and catalyst loading to maximize yield and safety. The process begins by dissolving the nitrogen-protected precursor in water, followed by the precise addition of alkali to achieve the target pH window. Once the substrate is fully solubilized, the heterogeneous catalyst is introduced, and the system is pressurized with hydrogen. The detailed standardized synthesis steps, including specific molar ratios, agitation speeds, and safety protocols for handling hydrogen gas at scale, are outlined in the guide below for technical teams evaluating this route for commercial adoption.
- Dissolve the nitrogen-protected thieno[3,4-d]imidazolyl-4-ylidenevaleric acid precursor in water and adjust the pH to between 8.5 and 8.9 using a weak base such as sodium hydroxide.
- Add a heterogeneous metal catalyst, specifically 5% Palladium on Carbon (Pd/C), at a loading of approximately 20-30% by weight relative to the starting material.
- Conduct the hydrogenation reaction at elevated temperatures (around 100°C) and moderate hydrogen pressure (3 to 5 bar) until uptake is complete, followed by product isolation via pH adjustment and solvent extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this aqueous hydrogenation technology translates into tangible operational efficiencies and risk mitigation. The most immediate impact is the drastic reduction in solvent costs. Replacing expensive, flammable organic solvents like isopropanol with water significantly lowers the raw material bill and reduces the energy burden associated with solvent distillation and recovery. Additionally, the simplified workup procedure—filtering the catalyst and extracting the product after acidification—reduces the number of unit operations required. This streamlining leads to shorter batch cycle times, allowing manufacturing facilities to increase throughput without additional capital investment. The robustness of the process also enhances supply chain reliability by reducing the likelihood of batch failures due to solvent quality issues or complex purification bottlenecks.
- Cost Reduction in Manufacturing: The elimination of volatile organic solvents and the use of inexpensive inorganic bases for pH adjustment result in substantial cost savings. By avoiding the need for complex solvent recovery infrastructure and reducing energy consumption for heating and distilling organics, the overall cost of goods sold (COGS) for the biotin intermediate is significantly optimized. Furthermore, the ability to recycle the aqueous phase or treat it with lower environmental impact reduces waste disposal costs, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: Utilizing water as a solvent mitigates the risks associated with the supply and storage of hazardous organic chemicals. This improves facility safety profiles and reduces regulatory compliance burdens related to VOC emissions. The simplicity of the process also means that production can be scaled up more predictably, ensuring consistent delivery schedules for downstream API manufacturers. The robustness of the Pd/C catalyst system ensures high conversion rates, minimizing the risk of supply shortages caused by low-yielding batches.
- Scalability and Environmental Compliance: This green chemistry approach aligns perfectly with modern sustainability goals. The aqueous waste stream is easier to treat compared to mixed organic waste, facilitating compliance with increasingly strict environmental regulations. The process is inherently scalable from pilot plant to multi-ton production, as heat transfer and mixing in aqueous systems are generally more manageable than in viscous organic media. This scalability ensures that suppliers can meet growing global demand for biotin without compromising on environmental standards or safety protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented hydrogenation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: Why is the pH adjusted to greater than 7 during the hydrogenation of biotin intermediates?
A: Adjusting the pH to above 7, specifically around 8.7, converts the carboxylic acid functional group on the intermediate into a water-soluble salt. This solubility allows the reaction to proceed efficiently in a protic solvent like water, eliminating the need for expensive organic solvents and simplifying the workup process.
Q: What catalyst system is preferred for this selective hydrogenation process?
A: The patent specifies the use of heterogeneous catalysts, with Palladium on Carbon (Pd/C) being particularly preferred. A loading of 5% Pd on an activated carbon support is effective, typically used at 20-30% by weight relative to the substrate, ensuring high conversion rates without the complications of removing soluble metal complexes.
Q: How does this aqueous method improve upon conventional organic solvent hydrogenation?
A: Conventional methods often rely on organic solvents like isopropanol and may require complex catalyst removal steps if soluble catalysts are used. This aqueous method allows for simple filtration of the heterogeneous catalyst and easy product isolation through acidification and extraction, significantly reducing solvent costs and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biotin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced process technologies like the one described in CN100369872C for the production of high-quality vitamins and supplements. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of biotin intermediate meets the exacting standards required by the global pharmaceutical and nutraceutical industries. Our commitment to technical excellence allows us to offer clients a secure and reliable source for critical vitamin precursors.
We invite procurement leaders and R&D directors to collaborate with us to leverage these efficiency gains for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your transition to this advanced manufacturing method is seamless, compliant, and economically advantageous.
