Revolutionizing Steroid Esterification: A Scalable Route for High-Purity 17α-Esters
The pharmaceutical industry constantly seeks robust methodologies for synthesizing complex steroidal intermediates with high regioselectivity. Patent CN101891797A introduces a groundbreaking preparation method for steroidal compound 17α-esters, specifically addressing the longstanding challenge of differentiating between the 17α and 21 hydroxyl groups during esterification. This technology utilizes a sophisticated cyclic ester intermediate strategy followed by a highly selective hydrolysis step, offering a reliable steroidal intermediate supplier pathway for producing critical anti-inflammatory agents like Hydrocortisone Butyrate. By leveraging specific directional reagents and optimized solvent systems, this process effectively mitigates the formation of undesirable isomers, ensuring that the final API intermediate meets stringent purity specifications required for topical dermatological formulations.
The significance of this patent lies in its ability to transform raw 17α,21-dihydroxy steroids into valuable 17α-esters with exceptional efficiency. Traditional methods often struggle with the similar reactivity of the hydroxyl groups at positions 17 and 21, leading to complex mixtures that are costly and difficult to separate. The disclosed invention overcomes these limitations by first protecting the diol system as a cyclic orthoester, followed by a controlled cleavage that favors the 17-position. This approach not only simplifies the synthetic route but also aligns with modern green chemistry principles by utilizing recoverable solvents and minimizing hazardous waste generation, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
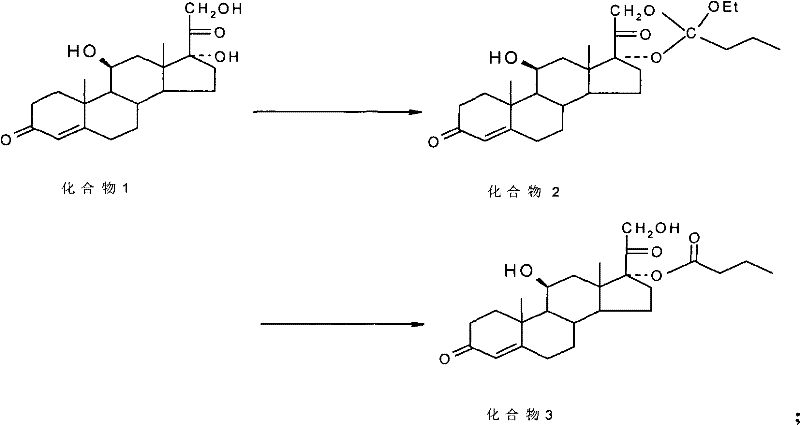
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 17α-esters such as Hydrocortisone Butyrate has been plagued by poor regioselectivity during the hydrolysis of cyclic precursors. Conventional techniques often rely on strong mineral acids like sulfuric acid or solid-state catalysts such as silica gel and Zeo-karb, which fail to adequately distinguish between the steric environments of the 17α and 21 hydroxyl groups. Consequently, these methods frequently result in hydrolysates containing significant amounts of the 21-ester isomer, sometimes reaching ratios as unfavorable as 7:3 or generating up to 10% of the unwanted by-product. This lack of selectivity necessitates extensive and expensive purification steps, such as repeated recrystallization or chromatography, which severely impact the overall yield and economic viability of the production process. Furthermore, the harsh conditions associated with traditional acid hydrolysis can lead to degradation of the sensitive steroid backbone, further compromising the quality of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the method described in CN101891797A employs a mild and highly controlled two-stage process that ensures superior product quality. The innovation begins with the formation of a cyclic ester using orthoesters in solvents like dioxane, creating a stable intermediate that protects both hydroxyl groups simultaneously. The true breakthrough occurs in the second stage, where the introduction of a directional reagent, specifically saturated aqueous ammonium chloride, fundamentally alters the hydrolysis kinetics. This additive works in concert with hydrolyzing agents like aluminum chloride to selectively cleave the ester bond at the 21-position while preserving the ester at the 17α-position, or vice versa depending on the specific target, effectively driving the equilibrium towards the desired 17α-ester. This strategic manipulation of reaction conditions eliminates the need for aggressive reagents and results in a dramatic improvement in conversion rates, allowing manufacturers to achieve high-purity steroidal compounds with minimal impurity profiles.
Mechanistic Insights into Selective Hydrolysis and Cyclization
The core of this technology rests on the precise manipulation of steric and electronic factors during the hydrolysis of the cyclic orthoester intermediate. When the cyclic ester is dissolved in a polar solvent like methanol and treated with ammonium chloride, the ammonium ions likely interact with the oxygen atoms of the ester linkage, stabilizing the transition state that leads to the 17α-ester. The presence of aluminum chloride or other Lewis acids further activates the carbonyl group, facilitating nucleophilic attack by water molecules in a highly regioselective manner. This mechanistic pathway ensures that the hydrolysis proceeds with a strong preference for the thermodynamically favored or kinetically accessible 17α-position, effectively suppressing the formation of the 21-ester isomer which is often the major by-product in non-catalyzed or poorly catalyzed reactions. The result is a clean reaction profile that minimizes the generation of structural analogs that are difficult to remove.
Furthermore, the choice of solvent plays a critical role in maintaining the integrity of the intermediate and facilitating the selective transformation. Solvents such as dioxane and dimethyl sulfoxide (DMSO) provide an optimal dielectric environment that supports the dissolution of the steroid substrate while remaining inert to the acidic conditions required for cyclization. The subsequent switch to methanol or acetone for the hydrolysis step ensures that the directional reagent is fully soluble and effective. This careful orchestration of solvent polarity and reagent compatibility prevents side reactions such as elimination or rearrangement of the steroid skeleton, which are common pitfalls in steroid chemistry. By controlling these variables, the process achieves a total recovery of the 17α-ester product approaching theoretical limits, with the 21-ester impurity consistently controlled below detectable or negligible levels, ensuring a robust and reproducible manufacturing protocol.
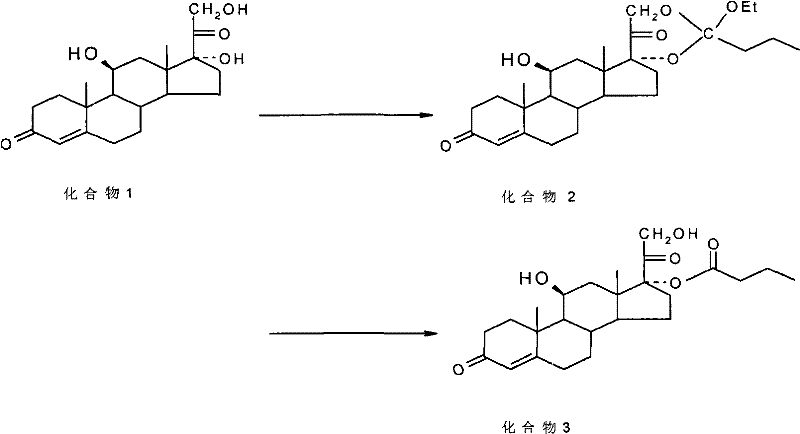
How to Synthesize Hydrocortisone Butyrate Efficiently
The synthesis of Hydrocortisone Butyrate via this patented route involves a streamlined sequence that is amenable to large-scale production. The process initiates with the dissolution of hydrocortisone in a cyclic ester solvent, followed by the addition of an orthoester and an acid catalyst to generate the cyclic intermediate. After isolation and neutralization, the intermediate undergoes selective hydrolysis in the presence of ammonium chloride to yield the final product. This methodology not only simplifies the operational workflow but also enhances safety by avoiding the use of highly corrosive reagents in the critical selectivity-determining step. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Dissolve the 17α,21-dihydroxy steroid in a cyclic ester solvent like dioxane, add an orthoester and acid catalyst to form the cyclic ester intermediate.
- Neutralize the reaction mixture with saturated sodium bicarbonate solution and wash to isolate the crude cyclic ester.
- Dissolve the cyclic ester in methanol, add saturated aqueous ammonium chloride and a hydrolyzing agent like aluminum chloride to selectively obtain the 17α-ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits in terms of cost efficiency and supply reliability. The ability to produce high-purity intermediates with minimal by-product formation translates directly into reduced raw material consumption and lower waste disposal costs. By eliminating the need for complex purification trains to remove 21-ester impurities, manufacturers can significantly shorten production cycles and reduce the burden on quality control laboratories. This efficiency gain is crucial for maintaining competitive pricing in the global market for topical corticosteroids, where margin pressures are constant. Moreover, the use of common, commercially available solvents and catalysts ensures that the supply chain remains resilient against fluctuations in the availability of exotic reagents.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substantial increase in yield and the drastic reduction in impurity levels. By achieving near-quantitative conversion to the desired 17α-ester, the process minimizes the loss of expensive starting materials like hydrocortisone. Additionally, the suppression of the 21-ester by-product removes the necessity for costly chromatographic separations or multiple recrystallization steps, which are often the most expensive part of steroid processing. The use of recoverable solvents like dioxane further contributes to cost savings by allowing for solvent recycling, thereby lowering the overall variable cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures consistent batch-to-batch quality, which is vital for maintaining uninterrupted supply to downstream formulation partners. The reliance on stable and widely available reagents such as ammonium chloride and aluminum chloride reduces the risk of supply disruptions associated with specialized catalysts. Furthermore, the mild reaction conditions reduce equipment corrosion and maintenance requirements, leading to higher plant availability and uptime. This reliability allows supply chain planners to forecast production volumes with greater confidence, ensuring that inventory levels are sufficient to meet market demand without excessive safety stock.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers significant advantages over traditional acid hydrolysis routes. The avoidance of large quantities of strong mineral acids reduces the generation of acidic wastewater, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The process is designed to be scalable from pilot plant to commercial production without significant modification, facilitating rapid technology transfer. The high atom economy and reduced solvent usage align with green chemistry initiatives, enhancing the corporate sustainability profile of manufacturers who adopt this technology for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this steroidal esterification process. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential licensees and manufacturing partners.
Q: What is the key advantage of using ammonium chloride in this hydrolysis process?
A: Ammonium chloride acts as a directional reagent that significantly shifts the hydrolysis equilibrium towards the desired 17α-ester while suppressing the formation of the unwanted 21-ester isomer, thereby enhancing overall product purity.
Q: Which solvents are compatible with the cyclic ester formation step?
A: The process allows for flexibility in solvent selection, including dioxane, benzene, toluene, DMF, acetonitrile, or DMSO, enabling manufacturers to optimize based on cost and recovery capabilities.
Q: How does this method compare to traditional sulfuric acid hydrolysis?
A: Unlike traditional sulfuric acid hydrolysis which often yields a poor ratio of 17α-ester to 21-ester (e.g., 7:3), this novel method achieves high conversion rates with minimal 21-ester impurity, drastically reducing downstream purification burdens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrocortisone Butyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and selective synthesis routes in the production of high-value steroid derivatives. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101891797A are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Hydrocortisone Butyrate or related 17α-ester meets the highest international standards for potency and impurity control.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to obtain specific COA data and route feasibility assessments, and let us help you optimize your production of high-purity steroidal intermediates.
