Advanced Synthesis of Hydrocortisone Butyrate for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously demands more efficient and purer synthetic routes for critical dermatological active ingredients, and patent CN101812108B presents a significant breakthrough in the manufacturing of Hydrocortisone Butyrate (CAS: 13609-67-1). This specific technical disclosure outlines a novel two-step methodology that starts from the readily available Hydrocortisone Acetate, bypassing the complex and impurity-prone orthoester pathways traditionally employed in the seventies. By leveraging specific catalytic conditions and precise pH control mechanisms, this process achieves high selectivity at the C17 position while preserving the integrity of the C21 acetate group until the final hydrolysis step. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages of this route is crucial for ensuring long-term supply chain stability and cost efficiency in topical corticosteroid production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 17-esters of pregnane steroids relied heavily on the formation of 17,21-cyclic ortho esters as a protective strategy to differentiate the hydroxyl groups. As illustrated in the structural representation of these intermediates, the conventional pathway involves reacting hydrocortisone with original acid triethyl to form a cyclic structure, followed by selective acid hydrolysis to open the ring at the 21-position. However, this legacy technology suffers from inherent thermodynamic and kinetic drawbacks, primarily because the acid-catalyzed ring-opening process often lacks perfect regioselectivity. Consequently, a significant portion of the 21-ester isomer is frequently generated alongside the desired 17-ester, creating a mixture of compounds with remarkably similar polarities that are notoriously difficult to separate via standard recrystallization techniques. This lack of separation efficiency forces manufacturers to perform multiple purification cycles, which drastically reduces the overall yield and increases the consumption of solvents and energy, thereby inflating the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
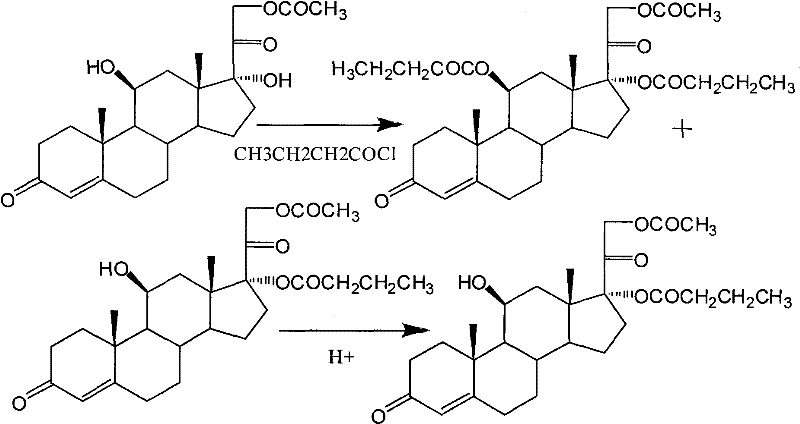
In stark contrast to the orthoester dependency, the innovative route described in the patent utilizes a direct selective acylation strategy that fundamentally alters the reaction landscape. By employing Hydrocortisone Acetate as the starting material and reacting it with butyryl chloride in the presence of specific nucleophilic catalysts, the process achieves direct esterification at the C17-alpha-hydroxyl group without needing cyclic protection. This approach effectively circumvents the steric hindrance issues typically associated with the beta-acetone side chain, which usually prevents direct esterification. Furthermore, the subsequent selective hydrolysis step allows for the precise removal of the C21-acetate group under mild alkaline conditions, yielding the target Hydrocortisone 17-alpha-butyrate with superior purity profiles. This streamlined workflow not only simplifies the operational complexity but also enhances the commercial scale-up of complex pharmaceutical intermediates by reducing the number of unit operations required.
Mechanistic Insights into DMAP-Catalyzed Selective Acylation
The core of this synthetic advancement lies in the sophisticated management of steric and electronic effects during the acylation phase. The C17-alpha-hydroxyl group in pregnane derivatives is sterically hindered by the adjacent beta-acetone side chain and the angular methyl groups, making it significantly less reactive than the primary C21-hydroxyl or the C11-hydroxyl. To overcome this, the protocol employs catalysts such as 4-dimethylaminopyridine (DMAP) or pyridinium tribromide hydrohalogenic acid salts, which act as potent nucleophilic accelerators. These catalysts form highly reactive acyl-pyridinium intermediates with butyryl chloride, which are sufficiently electrophilic to attack the hindered C17 position even at low temperatures ranging from 0°C to 10°C. This low-temperature regime is critical for suppressing side reactions and maintaining the stability of the sensitive steroid backbone while ensuring that the acylation occurs predominantly at the desired secondary alcohol site.
Another brilliant aspect of this mechanism is the in-situ control of impurities through pH manipulation. During the acylation reaction, a minor amount of 11-beta-butyrate byproduct may form due to the reactivity of the C11-hydroxyl group. However, the patent reveals that this 11-beta-ester linkage is chemically unstable under acidic conditions. By adjusting the reaction mixture pH to below 5, preferably between 1 and 3 using hydrochloric or organic acids, the unstable 11-beta-butyrate undergoes rapid hydrolysis back to the free hydroxyl form. This self-correcting mechanism ensures that the final intermediate, Hydrocortisone 17-alpha-butyrate-21-acetate, is obtained with minimal 11-ester contamination, thereby simplifying downstream purification and ensuring high-purity OLED material grade standards are met for pharmaceutical applications.
How to Synthesize Hydrocortisone Butyrate Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and purity. The process begins with the dissolution of Hydrocortisone Acetate in a suitable organic solvent system, followed by cooling to sub-ambient temperatures to manage exothermicity. The addition of butyryl chloride must be controlled slowly to maintain the reaction temperature between 0°C and 10°C, ensuring optimal selectivity. Following the acylation, the critical pH adjustment step is performed to eliminate 11-ester impurities before isolation. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and workup procedures, are outlined in the guide below for technical reference.
- Perform selective acylation of Hydrocortisone Acetate with butyryl chloride using DMAP catalyst at 0-10°C to form the 17-butyrate-21-acetate intermediate.
- Adjust pH to below 5 using acid to hydrolyze unstable 11-beta-butyrate impurities formed during the reaction.
- Conduct selective hydrolysis of the 21-acetate group using mild inorganic base at -20 to 0°C to yield pure Hydrocortisone Butyrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers substantial strategic benefits beyond mere technical elegance. The elimination of the orthoester formation step removes a major bottleneck in production throughput, allowing for faster batch turnover and reduced equipment occupancy time. Furthermore, the ability to remove impurities via pH adjustment rather than extensive chromatography or repeated recrystallization translates directly into lower solvent consumption and waste generation. This aligns perfectly with modern green chemistry initiatives and reduces the environmental compliance burden on manufacturing facilities, making it a highly attractive option for sustainable specialty chemical sourcing.
- Cost Reduction in Manufacturing: The streamlined two-step process significantly lowers production costs by eliminating the need for expensive orthoester reagents and reducing the number of purification cycles. By avoiding the formation of hard-to-separate 21-ester isomers, the overall yield is improved, and the loss of valuable starting material is minimized. Additionally, the use of common reagents like butyryl chloride and inorganic bases ensures that raw material costs remain stable and predictable, facilitating better budget planning for large-scale cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Relying on robust chemistry that tolerates minor variations in reaction conditions enhances the reliability of supply. The method uses widely available solvents such as methylene dichloride and methanol, reducing the risk of raw material shortages that can plague specialized reagent supply chains. The robustness of the pH-controlled impurity removal step ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results and ensuring reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations like liquid-liquid extraction and crystallization that are easily transferred from pilot to commercial scale. The reduction in solvent usage and the avoidance of heavy metal catalysts or complex protecting group chemistry simplify waste treatment protocols. This makes the process environmentally friendlier and easier to permit in regulated jurisdictions, supporting the long-term commercial scale-up of complex polymer additives and pharmaceutical products alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of Hydrocortisone Butyrate. These answers are derived directly from the patented technical data to provide accurate guidance for potential partners and technical teams evaluating this route for their own production needs or procurement strategies.
Q: How does this method improve purity compared to orthoester routes?
A: By avoiding the formation of 17,21-cyclic ortho esters, this method eliminates difficult separation steps and reduces 21-ester impurities significantly.
Q: What catalysts are used for selective C17 acylation?
A: The process utilizes 4-dimethylaminopyridine (DMAP) or pyridinium tribromide hydrohalogenic acid salts to overcome steric hindrance at the C17 position.
Q: How are 11-beta impurities controlled during synthesis?
A: Unstable 11-beta-butyrate byproducts are selectively hydrolyzed back to hydroxyl groups by adjusting the reaction pH to below 5 during workup.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrocortisone Butyrate Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent to production requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of temperature and pH required for this synthesis is maintained perfectly at every scale. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of Hydrocortisone Butyrate meets the highest global pharmacopoeia standards.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your product quality while driving down overall costs.
