Scalable Manufacturing of Novel Optically Pure Binaphthyl NN-Dioxide Ligands for Advanced Asymmetric Catalysis
The pharmaceutical and fine chemical industries are constantly seeking advanced chiral ligands to drive efficiency in asymmetric synthesis, a field revolutionized by the Nobel Prize-winning work on chiral catalysis. Patent CN111732542A introduces a groundbreaking methodology for preparing novel optically pure binaphthyl NN-dioxide ligands, which serve as critical components in high-performance catalytic systems. This technology addresses the persistent demand for ligands that combine rigidity with tunable electronic properties, specifically by integrating an axial chiral binaphthyl skeleton into the NN-dioxide framework. The innovation lies not just in the molecular architecture but in the robust, multi-step synthetic pathway that ensures high optical purity and structural integrity. For R&D directors and procurement specialists, understanding this patent provides a strategic advantage in sourcing next-generation catalysts that can unlock new reactivity profiles in drug substance manufacturing and specialty chemical production.
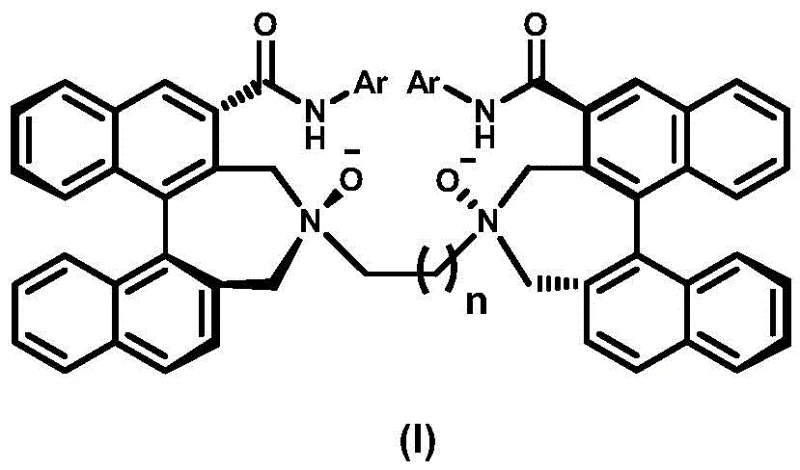
Traditional methods for synthesizing chiral nitrogen-oxide ligands often struggle with limited structural diversity or rely on resolution processes that inherently cap maximum yield at 50%. Conventional approaches frequently lack the rigid axial chirality necessary for inducing high enantioselectivity in complex metal-catalyzed transformations, leading to suboptimal reaction outcomes and difficult downstream purification. Furthermore, many existing ligands suffer from stability issues under harsh reaction conditions or require prohibitively expensive starting materials that hinder commercial viability. The reliance on less defined backbones often results in flexible structures that fail to maintain the precise spatial arrangement required for effective substrate differentiation during catalysis, thereby limiting their utility in industrial applications where consistency is paramount.
The novel approach detailed in this patent overcomes these limitations by employing a systematic construction of the ligand scaffold starting from readily available chiral binaphthyl amines. By utilizing a sequence of Boc protection, palladium-catalyzed carbonylation, and controlled amidation, the process builds complexity while preserving the critical stereocenters. The introduction of the NN-dioxide motif is achieved through a late-stage oxidation strategy that minimizes side reactions, ensuring the final product retains high optical purity. This method effectively expands the library of available chiral ligands, offering a modular platform where the aromatic substituents (Ar) can be varied to fine-tune steric and electronic properties for specific catalytic challenges, thus providing a versatile solution for diverse asymmetric synthesis needs.
Mechanistic Insights into Palladium-Catalyzed Carbonylation and N-Oxidation
The core of this synthetic strategy relies on a sophisticated palladium-catalyzed carbonylation step to install the carboxylic acid precursor, which is essential for subsequent amide bond formation. In this mechanism, the aryl bromide moiety of the protected binaphthyl intermediate undergoes oxidative addition to the Pd(0) species, followed by CO insertion to form an acyl-palladium complex. This intermediate is then trapped by benzyl alcohol to yield the ester, which is subsequently hydrogenated to the free acid. This sequence is critical because it allows for the functionalization of the sterically hindered binaphthyl core under relatively mild conditions, avoiding the racemization that might occur with harsher nucleophilic substitutions. The careful selection of ligands for the palladium catalyst, such as 1,3-bis(diphenylphosphino)propane, ensures high turnover and selectivity, which is vital for maintaining the economic feasibility of the process on a larger scale.
Impurity control is meticulously managed throughout the synthesis, particularly during the final oxidation step where the tertiary amines are converted to N-oxides. The use of m-chloroperoxybenzoic acid (mCPBA) at low temperatures (0°C) is a deliberate choice to prevent over-oxidation or degradation of the sensitive amide linkages and the chiral backbone. The polarity of the resulting nitrogen-oxygen dipole is crucial for the ligand's function, acting as both an electron donor and acceptor to coordinate with metal centers like scandium or copper. By strictly controlling the stoichiometry and reaction time, the process minimizes the formation of N,N'-dioxide impurities or unreacted amine precursors, ensuring that the final isolated ligand meets the stringent purity specifications required for high-value asymmetric catalysis applications in the pharmaceutical sector.
How to Synthesize Optically Pure Binaphthyl NN-Dioxide Ligand Efficiently
The synthesis of this high-value chiral ligand involves a convergent seven-step sequence that transforms simple chiral amines into complex dimeric structures. The process begins with the protection of the amine functionality to prevent unwanted side reactions during the carbonylation phase, followed by the strategic installation of the carboxylic acid handle. Subsequent coupling with bulky aromatic amines introduces the necessary steric bulk, while the dimerization step links two chiral units via an alkyl chain to create the final C2-symmetric or pseudo-symmetric framework. The detailed standardized synthesis steps below outline the specific reagents, temperatures, and workup procedures required to achieve the high yields and purity reported in the patent documentation, serving as a practical guide for laboratory scale-up.
- Protect (S)-3-brominated binaphthyl secondary amine with di-tert-butyl dicarbonate to form the Boc-protected intermediate.
- Perform palladium-catalyzed carbonylation followed by hydrogenation to convert the bromo group into a carboxylic acid functionality.
- Condense the acid with aromatic amines, remove the protecting group, dimerize using dibromoalkanes, and finalize with mCPBA oxidation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers significant strategic benefits regarding cost structure and supply continuity. The process utilizes commodity chemicals such as di-tert-butyl dicarbonate, benzyl alcohol, and standard palladium catalysts, which are widely available from multiple global suppliers, thereby reducing the risk of single-source bottlenecks. By avoiding the need for cryogenic conditions or exotic reagents in most steps, the manufacturing protocol is inherently more energy-efficient and safer to operate, translating to lower operational expenditures. The modular nature of the synthesis also allows for flexibility in sourcing; if a specific aromatic amine becomes scarce, the chemistry is robust enough to accommodate alternative substituents without requiring a complete process redesign, ensuring long-term supply chain resilience for critical catalytic materials.
- Cost Reduction in Manufacturing: The elimination of complex chiral resolution steps significantly lowers the cost of goods sold, as the chirality is derived directly from the inexpensive starting binaphthyl amine rather than through wasteful separation processes. Furthermore, the use of heterogeneous catalysts like Pd/C in the hydrogenation step facilitates easy catalyst recovery and recycling, reducing the consumption of precious metals. The high atom economy of the carbonylation and amidation steps minimizes waste generation, which in turn reduces the costs associated with waste disposal and environmental compliance, contributing to a leaner and more profitable manufacturing model for high-purity chiral ligands.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable intermediates allows for the strategic stocking of key precursors, buffering against market volatility in raw material prices. The synthetic route avoids the use of highly unstable or hazardous reagents that often require special shipping and handling protocols, simplifying logistics and reducing lead times for international shipments. Additionally, the robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-validation, providing supply chain leaders with the flexibility to diversify production geography and mitigate regional disruption risks effectively.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like ethyl acetate and dichloromethane which are well-understood in large-scale chemical engineering and can be efficiently recovered and recycled. The absence of heavy metal contaminants in the final product, due to efficient purification steps like column chromatography and crystallization, simplifies the regulatory filing process for downstream pharmaceutical customers. This alignment with green chemistry principles, such as catalytic efficiency and waste minimization, ensures that the manufacturing facility remains compliant with increasingly stringent environmental regulations, safeguarding the license to operate for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel ligand class. These answers are derived directly from the experimental data and claims within the patent specification, providing clarity on the feasibility and performance characteristics of the technology. Understanding these details is essential for technical teams evaluating the integration of this ligand into their existing catalytic processes or for procurement teams assessing the vendor's technical capability.
Q: What is the key structural advantage of this novel binaphthyl NN-dioxide ligand?
A: The ligand incorporates an axial chiral binaphthyl rigid skeleton into the NN-dioxide framework, significantly expanding the diversity of chiral backbones available for asymmetric catalysis and enhancing stereocontrol in metal-complex formation.
Q: What are the critical reaction conditions for the final oxidation step?
A: The final nitrogen oxidation is conducted at 0°C using m-chloroperoxybenzoic acid (mCPBA) in dichloromethane, ensuring selective formation of the N-oxide dipole without degrading the sensitive chiral backbone.
Q: Is this synthesis suitable for large-scale production of chiral intermediates?
A: Yes, the route utilizes standard reagents like Pd/C for hydrogenation and common solvents like ethyl acetate and dichloromethane, avoiding exotic catalysts and facilitating straightforward scale-up for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Binaphthyl NN-Dioxide Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced chiral ligands play in accelerating drug discovery and optimizing fine chemical synthesis. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of our chiral intermediates meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality assurance means that clients can rely on us for consistent supply of high-performance materials that drive their own R&D success.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific asymmetric catalysis needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized manufacturing processes can reduce your overall material costs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral chemistry can become a cornerstone of your supply chain strategy for next-generation therapeutic agents.
