Scalable Manufacturing Of Novel Binaphthyl NN-Dioxide Ligands For Asymmetric Catalysis
Scalable Manufacturing Of Novel Binaphthyl NN-Dioxide Ligands For Asymmetric Catalysis
In the rapidly evolving landscape of asymmetric synthesis, the demand for highly efficient chiral ligands has never been more critical for the production of enantiomerically pure pharmaceuticals and advanced materials. Patent CN111732542A introduces a groundbreaking methodology for the preparation of novel optically pure binaphthyl NN-dioxide ligands, representing a significant leap forward in ligand design. This technology addresses the persistent challenge of balancing structural rigidity with electronic tunability, which is essential for achieving high enantioselectivity in metal-catalyzed reactions. By integrating an axial chiral binaphthyl skeleton with polar nitrogen-oxygen dipoles, this invention expands the diversity of available chiral frameworks beyond traditional phosphine or simple amine ligands. For R&D directors and procurement specialists, understanding the nuances of this synthesis is key to securing a reliable supply of high-performance catalytic components.
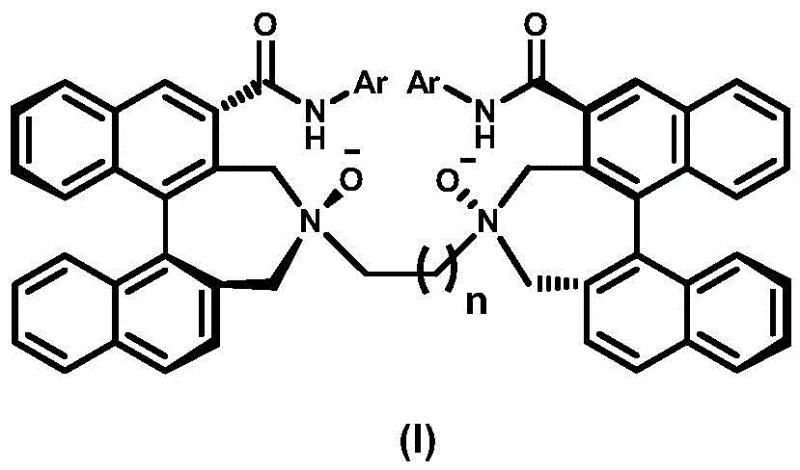
The strategic value of this patent lies not just in the molecular architecture but in the robustness of the synthetic pathway described. Unlike many academic protocols that rely on air-sensitive reagents or impractical cryogenic conditions, the disclosed method utilizes standard organic transformations that are amenable to kilogram-scale manufacturing. The process begins with readily available chiral building blocks and proceeds through a series of well-defined intermediates, ensuring consistent quality and batch-to-batch reproducibility. This level of process control is vital for industrial applications where supply chain continuity and cost predictability are paramount. As we delve deeper into the technical specifics, it becomes clear that this ligand class offers a versatile platform for developing next-generation catalysts for complex bond-forming reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral ligands for asymmetric catalysis has often been hindered by synthetic complexity and limited structural diversity. Traditional approaches frequently rely on rigid scaffolds that lack the necessary electronic flexibility to adapt to different substrates, or conversely, flexible backbones that fail to provide sufficient stereochemical induction. Furthermore, many existing N-oxide ligands suffer from difficult purification processes, often requiring extensive chromatography that drives up costs and limits scalability. The reliance on expensive transition metals without efficient recovery systems also poses environmental and economic challenges. In many cases, the inability to easily modify the steric environment around the coordinating atoms restricts the ligand's utility across a broad range of catalytic transformations, forcing chemists to screen dozens of suboptimal candidates.
The Novel Approach
The methodology outlined in patent CN111732542A overcomes these hurdles through a modular synthetic strategy that decouples the installation of the chiral backbone from the tuning of the electronic environment. By employing a palladium-catalyzed carbonylation step early in the sequence, the process efficiently installs functional handles that allow for subsequent diversification. The introduction of bulky aromatic groups via amide coupling provides precise steric control, while the final oxidation step generates the crucial N-oxide dipoles responsible for strong metal coordination. This approach not only streamlines the synthesis but also enhances the overall yield and purity of the final ligand. The result is a family of ligands that combine the rigidity of the binaphthyl core with the tunable electronics of the N-oxide moiety, offering a superior alternative for demanding asymmetric syntheses.
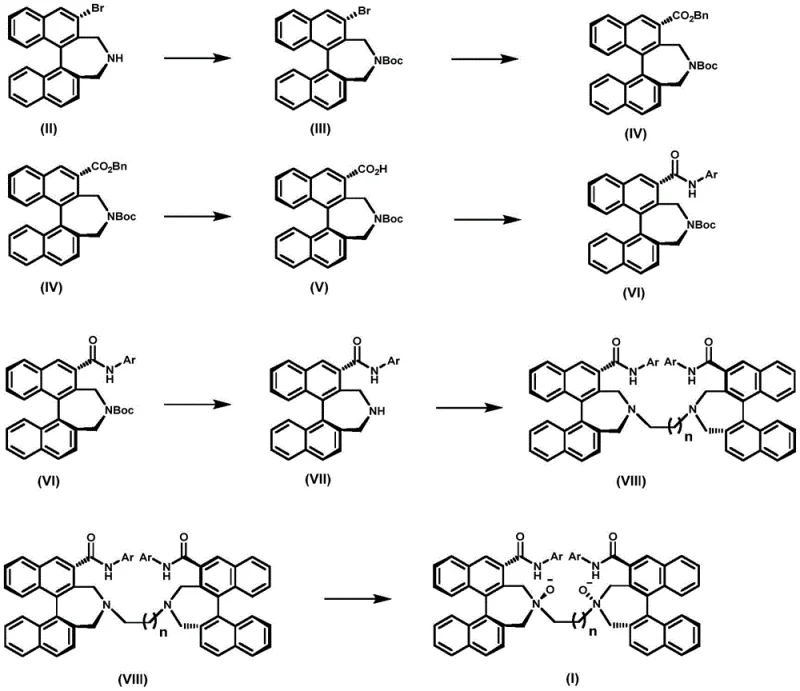
Mechanistic Insights into Binaphthyl NN-Dioxide Ligand Synthesis
The core of this innovation lies in the sophisticated manipulation of the binaphthyl scaffold to create a highly defined chiral pocket. The synthesis initiates with the protection of the secondary amine, followed by a critical palladium-catalyzed carbonylation that converts the aryl bromide into a benzyl ester. This step is mechanistically significant as it preserves the axial chirality of the binaphthyl system while introducing a versatile carbonyl functionality. Subsequent hydrogenation and amide coupling steps are carefully optimized to prevent racemization, ensuring that the optical purity of the starting material is transferred quantitatively to the final product. The use of specific condensing agents and bases during the amide formation minimizes side reactions, which is crucial for maintaining high purity levels required in pharmaceutical intermediate manufacturing.
The final transformation involves the oxidation of the tertiary amine nitrogens to N-oxides using meta-chloroperoxybenzoic acid (m-CPBA). Mechanistically, this creates a strong N→O dipole that acts as a potent electron donor to Lewis acidic metal centers such as scandium, copper, or lanthanides. The rigidity of the binaphthyl backbone ensures that the two N-oxide groups are held in a fixed spatial arrangement, creating a C2-symmetric environment that effectively discriminates between enantiotopic faces of a substrate. This structural feature is what enables the ligand to induce high enantioselectivity in reactions like aldol condensations or Friedel-Crafts alkylations. Understanding this mechanism allows process chemists to appreciate why this specific ligand architecture outperforms simpler analogues in complex catalytic cycles.
How to Synthesize Optically Pure Binaphthyl NN-Dioxide Ligand Efficiently
The synthesis of these advanced ligands requires precise control over reaction parameters to ensure optimal yield and stereochemical integrity. The patented route involves seven distinct chemical transformations, each designed to build complexity while maintaining purity. From the initial Boc protection to the final oxidative workup, every step has been validated to minimize impurity formation. For laboratories looking to replicate or scale this chemistry, adherence to the specified stoichiometry and temperature profiles is essential. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a foundational reference for process development teams aiming to implement this technology.
- Protect the (S)-3-brominated binaphthyl secondary amine with Boc anhydride, followed by Pd-catalyzed benzyloxycarbonylation to install the ester group.
- Perform catalytic hydrogenation to remove the benzyl group, then couple with aromatic amines using T3P/DMAP to form the amide intermediate.
- Deprotect the amine, alkylate with dibromoalkanes to dimerize, and finally oxidize the tertiary amines with m-CPBA to yield the NN-dioxide ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel ligand synthesis offers tangible benefits in terms of cost efficiency and supply security. The reliance on commercially available starting materials, such as substituted anilines and dibromoalkanes, mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Furthermore, the synthetic route avoids the use of exotic reagents or proprietary catalysts that command premium pricing, thereby significantly reducing the overall cost of goods sold. The robustness of the chemistry means that production can be scaled from gram to kilogram quantities without the need for specialized equipment, ensuring a steady flow of material for downstream catalytic applications.
- Cost Reduction in Manufacturing: The elimination of cryogenic conditions and the use of standard solvent systems like dichloromethane and ethyl acetate drastically simplify the manufacturing infrastructure requirements. By avoiding expensive chiral resolution steps through the use of enantiopure starting materials, the process achieves substantial cost savings. Additionally, the high yields reported in the patent examples indicate minimal waste generation, which further lowers the effective cost per kilogram of the active ligand. This economic efficiency makes the ligand viable for large-scale industrial processes where margin compression is a constant concern.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for flexible production scheduling, as intermediates can be stockpiled at various stages to buffer against demand fluctuations. The use of stable intermediates reduces the risk of degradation during storage and transport, ensuring that the material arrives at the customer's site with guaranteed potency. This stability is a critical factor for global supply chains where long lead times and variable storage conditions are common. By partnering with a supplier capable of executing this robust chemistry, companies can secure a long-term source of high-quality chiral ligands.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing catalytic amounts of palladium which can be recovered and recycled, thus minimizing heavy metal waste. The final purification steps rely on standard chromatography or crystallization techniques that are easily adaptable to continuous manufacturing setups. This scalability ensures that the technology can grow alongside the customer's production needs, from pilot plant trials to full commercial tonnage. Moreover, the reduced solvent intensity and waste profile align with increasingly stringent environmental regulations, facilitating smoother regulatory approvals for the final drug substance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these binaphthyl NN-dioxide ligands. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this ligand class into their catalytic workflows.
Q: What is the key structural advantage of this binaphthyl NN-dioxide ligand?
A: The ligand features a rigid axial chiral binaphthyl backbone combined with polar N-oxide dipoles, offering superior stereocontrol and tunable electronic properties for metal catalysis.
Q: How does this synthesis method improve scalability compared to traditional routes?
A: The route utilizes robust reactions like Pd-catalyzed carbonylation and standard acid-base workups, avoiding cryogenic conditions and enabling easier purification via column chromatography.
Q: Can the aryl groups on the ligand be customized for specific reactions?
A: Yes, the synthetic pathway allows for the introduction of various sterically hindered aromatic amines (e.g., mesityl, triisopropylphenyl), allowing fine-tuning of the catalyst pocket.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Binaphthyl NN-Dioxide Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful implementation of advanced catalytic technologies requires more than just access to a patent; it demands a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to factory floor is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of ligand meets the highest standards of optical and chemical purity. Our commitment to quality assurance means that you can rely on our materials to deliver consistent performance in your most sensitive asymmetric reactions.
We invite you to engage with our technical procurement team to discuss how this novel ligand technology can optimize your specific catalytic processes. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to our manufactured ligands. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us collaborate to accelerate your development timelines and secure a competitive advantage in the marketplace through superior chiral catalysis.
