Scalable Production of Optically Pure Binaphthyl NN-Dioxide Ligands for Asymmetric Catalysis
Scalable Production of Optically Pure Binaphthyl NN-Dioxide Ligands for Asymmetric Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for highly efficient chiral ligands that can induce precise stereocontrol in complex organic transformations. Patent CN111732542B discloses a groundbreaking preparation method for a novel optically pure binaphthyl NN-dioxide ligand, addressing critical gaps in the diversity of chiral skeletons available for industrial application. This technology creatively integrates an axial chiral binaphthyl framework into the NN-dioxide ligand architecture, significantly expanding the toolbox for chemists working in pharmaceutical and fine chemical synthesis. The structural integrity and electronic properties of this ligand, as depicted in the formula below, make it a superior candidate for coordinating with various transition metals to facilitate green and efficient asymmetric reactions.
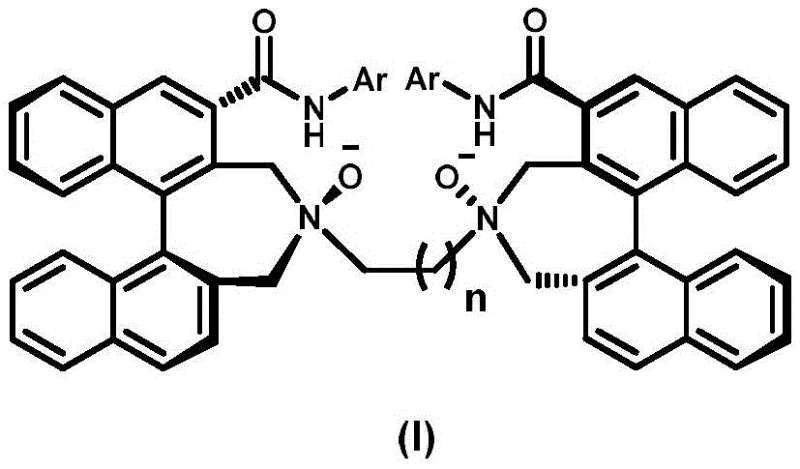
For R&D directors and procurement specialists seeking a reliable chiral ligand supplier, this patent represents a significant advancement in process chemistry. The synthesis route is designed to maintain high optical purity while utilizing robust reaction conditions that are amenable to scale-up. By introducing specific substituents on the aromatic rings, such as 2,4,6-trimethylphenyl or 2,4,6-triisopropylphenyl groups, the steric environment around the catalytic center can be finely tuned. This level of customization is essential for optimizing reaction outcomes in the manufacturing of high-value active pharmaceutical ingredients (APIs) and advanced materials, ensuring that the supply chain can meet the rigorous specifications required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing chiral nitrogen-oxygen ligands often suffer from limited structural diversity and cumbersome purification processes that hinder commercial viability. Many existing methods rely on racemic mixtures that require expensive and wasteful resolution steps to isolate the desired enantiomer, drastically increasing the cost of goods sold and extending lead times for project timelines. Furthermore, conventional ligands frequently lack the necessary rigidity or electronic tunability to support a broad scope of asymmetric transformations, limiting their utility in complex multi-step syntheses. The inability to effectively control the axial chirality in earlier generations of binaphthyl derivatives often resulted in inconsistent catalytic performance, posing a significant risk for process chemists aiming to establish robust manufacturing protocols for sensitive intermediates.
The Novel Approach
The methodology outlined in patent CN111732542B overcomes these historical barriers through a meticulously designed seven-step synthetic sequence that prioritizes yield and stereochemical fidelity. By starting with optically pure (S)-3-bromobinaphthyl secondary amine, the process inherently preserves chirality, eliminating the need for downstream resolution and ensuring a consistent supply of high-purity material. The introduction of a palladium-catalyzed carbonylation step allows for the efficient construction of the carbon skeleton under relatively mild conditions, while the subsequent oxidation to the N-oxide functionality is achieved with high selectivity. This novel approach not only enriches the library of available chiral ligands but also provides a scalable pathway that aligns with the principles of green chemistry, reducing solvent waste and energy consumption compared to legacy methods.
Mechanistic Insights into Pd-Catalyzed Carbonylation and N-Oxidation
The core of this synthetic innovation lies in the strategic application of transition metal catalysis and selective oxidation to build the complex ligand architecture. The second step of the synthesis utilizes a palladium catalyst system, specifically Pd(OAc)2 coupled with 1,3-bis(diphenylphosphino)propane, to facilitate the benzyloxycarbonylation of the bromo-intermediate. This transformation is critical as it installs the ester functionality required for subsequent amidation, proceeding through a oxidative addition and migratory insertion mechanism that is highly sensitive to ligand sterics and electronic properties. The careful selection of solvents, such as a mixture of dimethyl sulfoxide and benzyl alcohol, ensures optimal solubility of the bulky binaphthyl species while maintaining the activity of the palladium center, resulting in a clean conversion that minimizes the formation of homocoupling byproducts.
Following the assembly of the backbone, the final oxidation step employs m-chloroperoxybenzoic acid (m-CPBA) to convert the tertiary amines into the active N-oxide moieties. This reaction is performed at low temperatures, typically around 0 degrees Celsius, to prevent over-oxidation or degradation of the sensitive chiral axis. The resulting N-O dipole possesses strong polarity, which is fundamental to the ligand's ability to act as an electron donor in metal complexes. This mechanistic precision ensures that the final product exhibits the desired electrophilic and nucleophilic characteristics, enabling it to coordinate effectively with metals like scandium or copper to drive asymmetric catalytic cycles with high enantioselectivity, a crucial factor for the synthesis of single-enantiomer drugs.
How to Synthesize Optically Pure Binaphthyl NN-Dioxide Ligand Efficiently
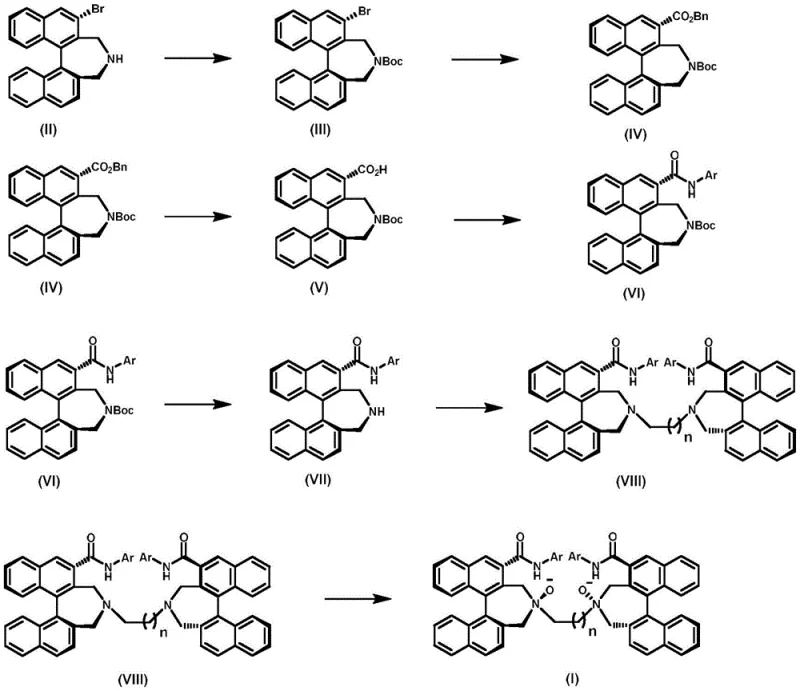
The preparation of this high-value ligand follows a logical progression of protection, functionalization, and oxidation steps that have been optimized for reproducibility and yield. The process begins with the protection of the secondary amine to prevent side reactions during the carbonylation phase, followed by the key palladium-catalyzed step that builds molecular complexity. Subsequent hydrogenation and amidation steps install the necessary aromatic amide groups, which are critical for defining the steric pocket of the final catalyst. The route concludes with a disubstitution reaction to link the two halves of the molecule and a final oxidation to activate the ligand. For a detailed visualization of this entire synthetic pathway, refer to the reaction scheme provided below, which outlines the transformation from the starting bromo-amine to the final N,N'-dioxide product.
- Protect (S)-3-bromobinaphthyl secondary amine with Boc anhydride to form the first intermediate.
- Perform palladium-catalyzed benzyloxycarbonylation to introduce the ester functionality.
- Execute catalytic hydrogenation to remove the benzyl group, followed by condensation with aromatic amines.
- Conduct acidolysis to deprotect the amine, followed by disubstitution with dibromoalkane.
- Finalize the synthesis by oxidizing the tertiary amines to N-oxides using m-CPBA.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this patented synthesis route offers substantial benefits in terms of cost stability and material availability. The reliance on readily available starting materials, such as substituted aromatic amines and standard protecting group reagents, mitigates the risk of raw material shortages that often plague the production of exotic chiral ligands. Furthermore, the high yields reported in the experimental examples, particularly in the final oxidation and initial protection steps, indicate a process with excellent atom economy. This efficiency translates directly into reduced waste generation and lower disposal costs, aligning with the sustainability goals of modern chemical manufacturing facilities while ensuring a consistent supply of critical reagents for R&D and production teams.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps significantly lowers the overall production cost by avoiding the loss of fifty percent of the material typically associated with racemic separations. Additionally, the use of standard heterogeneous catalysts like palladium on carbon for hydrogenation allows for easy catalyst recovery and recycling, further driving down the cost of goods. The high purity achieved directly from the synthesis reduces the need for extensive downstream purification, saving both time and expensive chromatographic resources during the commercial scale-up of complex ligands.
- Enhanced Supply Chain Reliability: By utilizing a linear synthesis strategy with robust intermediates, the manufacturing process is less susceptible to bottlenecks caused by unstable reagents or sensitive reaction conditions. The ability to store stable intermediates, such as the Boc-protected species, provides flexibility in production scheduling, allowing manufacturers to build inventory buffers against market fluctuations. This reliability is crucial for long-term contracts where consistent quality and delivery timelines are paramount for maintaining the continuity of downstream pharmaceutical or agrochemical production lines.
- Scalability and Environmental Compliance: The reaction conditions described, such as moderate temperatures and standard atmospheric pressures for most steps, are inherently safer and easier to scale in large-scale reactors compared to high-pressure or cryogenic alternatives. The solvent systems employed, primarily consisting of dichloromethane, ethyl acetate, and alcohols, are well-understood within the industry and can be efficiently recovered and recycled using standard distillation infrastructure. This facilitates compliance with increasingly stringent environmental regulations regarding volatile organic compound emissions and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel ligand series. These insights are derived directly from the patent specifications and are intended to assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing workflows. Understanding the specific handling requirements and potential applications of the N,N'-dioxide motif is essential for maximizing the value of this chiral tool in asymmetric synthesis projects.
Q: What is the optical purity of the synthesized binaphthyl ligand?
A: The patented process ensures high optical purity, leveraging the chiral integrity of the starting (S)-3-bromobinaphthyl secondary amine throughout the seven-step sequence, resulting in a final product with specific rotation values consistent with high enantiomeric excess.
Q: Can this ligand be used for metal-catalyzed asymmetric reactions?
A: Yes, the N,N'-dioxide structure features strong polarity and rigidity, making it an excellent electron donor capable of forming complex catalysts with metals such as scandium, copper, nickel, and lanthanides for diverse asymmetric transformations.
Q: What are the key advantages of this synthesis route over conventional methods?
A: This method introduces an axial chiral binaphthyl skeleton effectively, utilizes targeted catalyst matching to improve yield and purity, and avoids complex resolution steps by starting from optically pure precursors, ensuring a streamlined production workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Binaphthyl NN-Dioxide Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in the development of next-generation therapeutics and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ligand meets the exacting standards required for sensitive catalytic applications, providing our partners with the confidence to innovate without supply chain constraints.
We invite you to collaborate with us to optimize your catalytic processes and achieve significant cost efficiencies in your manufacturing operations. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral chemistry can accelerate your project timelines and enhance your competitive advantage in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
