Advanced N-Butyllithium Catalysis for High-Purity Boronic Ester Manufacturing
Advanced N-Butyllithium Catalysis for High-Purity Boronic Ester Manufacturing
The landscape of organoboron chemistry is undergoing a significant transformation driven by the need for more efficient and economically viable synthetic routes. Patent CN111320644B introduces a groundbreaking methodology for the preparation of boronic acid esters through a hydroboration reaction that leverages the high activity of commercial n-butyllithium. This innovation addresses long-standing challenges in the catalytic hydroboration of imines, a critical transformation for generating amine precursors and complex pharmaceutical intermediates. Unlike traditional methods that often rely on expensive, specialized catalysts or harsh reaction conditions, this novel approach utilizes a readily available commercial reagent to achieve exceptional yields exceeding 90% under mild conditions. For R&D directors and process chemists, this represents a paradigm shift towards atom-economic synthesis that simplifies workflow while maintaining rigorous purity standards. The ability to operate at room temperature with short reaction times of just 1 to 2 hours significantly enhances the throughput potential for large-scale manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydroboration of unsaturated C=N bonds has presented substantial technical hurdles compared to the more facile hydroboration of carbonyl groups. Existing catalytic systems reported in prior art frequently depend on main group element complexes involving magnesium, calcium, sodium, or zinc, which are often costly to synthesize and difficult to handle on an industrial scale. Furthermore, many of these conventional protocols necessitate elevated temperatures and extended reaction times to drive the conversion to completion, leading to increased energy consumption and potential thermal degradation of sensitive functional groups. Some catalytic systems suffer from low turnover numbers or poor substrate tolerance, limiting their applicability to a narrow range of simple imines. These inefficiencies create bottlenecks in the supply chain for high-purity pharmaceutical intermediates, where consistency and cost-effectiveness are paramount. The reliance on bespoke catalysts also introduces supply chain vulnerabilities, as the availability of these specialized reagents can be inconsistent, thereby jeopardizing production schedules for critical drug substances.
The Novel Approach
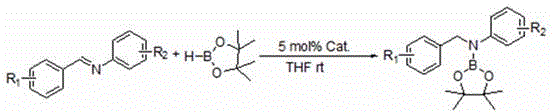
In stark contrast to these legacy methods, the technology disclosed in patent CN111320644B utilizes commercial n-butyllithium as a highly active catalyst, fundamentally altering the economic and operational profile of the reaction. This novel approach enables the hydroboration of imines with pinacolborane to proceed smoothly at room temperature, eliminating the energy costs associated with heating and cooling cycles. The reaction is remarkably fast, typically reaching completion within 1 to 2 hours, which drastically improves reactor utilization rates and overall plant capacity. As illustrated in the reaction scheme below, the process converts various substituted imines directly into the corresponding boronic esters with yields reaching up to 99% under optimized conditions. This level of efficiency not only reduces raw material waste but also simplifies the downstream purification process, as fewer side products are generated. The use of tetrahydrofuran (THF) as a standard solvent further enhances the practicality of this method, making it easily adaptable to existing manufacturing infrastructure without requiring specialized equipment modifications.
Mechanistic Insights into N-Butyllithium Catalyzed Hydroboration
The efficacy of this catalytic system lies in the unique ability of n-butyllithium to activate the boron-hydrogen bond of pinacolborane, facilitating its addition across the carbon-nitrogen double bond of the imine substrate. Mechanistically, the organolithium species acts as a Lewis base or initiates a deprotonation-reprotonation cycle that lowers the activation energy for the hydroboration step. This activation allows the reaction to proceed under neutral and mild conditions, preserving the integrity of other sensitive functional groups that might be present on the aromatic rings. The catalytic cycle is robust enough to tolerate a wide variety of electronic environments on the imine backbone, ensuring consistent performance regardless of the substituent nature. For process chemists, understanding this mechanism is crucial for optimizing reaction parameters such as stoichiometry and addition order to maximize yield and minimize impurity formation. The simplicity of the catalytic species also means that there are no complex ligand dissociation steps that could slow down the turnover frequency, resulting in the rapid kinetics observed in the experimental data.
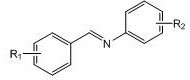
Furthermore, the substrate scope of this reaction is exceptionally broad, accommodating both electron-withdrawing and electron-donating groups on the aromatic rings of the imine. As shown in the general structure below, substituents such as halogens (fluorine, chlorine, bromine), methyl, and methoxy groups at various positions (ortho, meta, para) do not inhibit the catalytic activity. This universality is a significant advantage for the synthesis of diverse pharmaceutical intermediates, where structural variation is often required during lead optimization phases. The high chemoselectivity of the system ensures that the boron adds specifically to the imine carbon without affecting other reducible groups, leading to a clean impurity profile. This selectivity reduces the burden on purification teams, allowing for simpler crystallization or distillation steps to achieve the stringent purity specifications required for GMP manufacturing. The ability to handle such a diverse range of substrates with a single, inexpensive catalyst makes this technology highly versatile for contract development and manufacturing organizations.
How to Synthesize Boronic Esters Efficiently
The implementation of this synthesis route is straightforward and designed for scalability, requiring standard laboratory or plant equipment capable of handling inert atmospheres. The process begins with the preparation of the reaction vessel under anhydrous and oxygen-free conditions to prevent premature decomposition of the organolithium catalyst. Following the addition of the imine substrate and solvent, the borane reagent is introduced, followed by the precise dosing of the n-butyllithium catalyst solution. The reaction mixture is then stirred at ambient temperature, allowing the catalytic cycle to proceed to completion within a short timeframe. Upon completion, the reaction is conveniently quenched by exposure to air, which terminates the catalytic activity and simplifies the workup procedure. For detailed operational parameters and safety guidelines regarding the handling of pyrophoric reagents, please refer to the standardized synthesis steps provided below.
- Under an inert atmosphere, add the imine substrate and anhydrous THF solvent to a dried reaction vessel.
- Introduce pinacolborane to the mixture, followed by the addition of the n-butyllithium catalyst (4-5 mol%).
- Stir the reaction at room temperature for 1-2 hours, then expose to air to quench and remove solvent under reduced pressure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this n-butyllithium catalyzed process offers transformative benefits that directly impact the bottom line and operational resilience. The primary advantage stems from the substitution of expensive, custom-synthesized catalysts with a commodity chemical that is available in bulk quantities from multiple global suppliers. This shift significantly reduces the raw material cost per kilogram of the final product, enhancing the overall margin profile for high-volume manufacturing campaigns. Additionally, the elimination of complex catalyst preparation steps shortens the overall production cycle time, allowing facilities to respond more rapidly to fluctuating market demands. The mild reaction conditions also contribute to lower utility costs, as there is no need for extensive heating or cryogenic cooling systems, further driving down the operational expenditure. These factors combined create a more robust and cost-effective supply chain for critical boronic ester intermediates used in drug discovery and development.
- Cost Reduction in Manufacturing: The utilization of commercial n-butyllithium eliminates the need for purchasing or synthesizing proprietary transition metal or main group catalysts, which often carry high price tags and long lead times. By leveraging a widely available reagent, manufacturers can achieve substantial cost savings on the bill of materials while avoiding the supply risks associated with single-source specialty chemicals. The high atom economy of the reaction ensures that minimal raw material is wasted, contributing to a more sustainable and economically efficient process. Furthermore, the simplified workup procedure reduces the consumption of solvents and purification media, lowering the total cost of goods sold. These cumulative savings make the process highly competitive for the commercial production of generic pharmaceutical ingredients and fine chemicals.
- Enhanced Supply Chain Reliability: Relying on commodity reagents like n-butyllithium and pinacolborane significantly mitigates the risk of supply disruptions that can occur with specialized catalytic systems. Since these materials are produced at a massive scale for various industries, their availability is generally stable, ensuring continuous operation of manufacturing lines. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures. This reliability is crucial for maintaining consistent inventory levels and meeting strict delivery commitments to downstream pharmaceutical clients. Consequently, supply chain managers can plan production schedules with greater confidence, knowing that the core chemistry is supported by a secure and diversified supply base.
- Scalability and Environmental Compliance: The mild nature of this hydroboration reaction facilitates seamless scale-up from laboratory benchtop to multi-ton commercial production without the need for complex engineering controls. Operating at room temperature reduces the thermal load on reactors, minimizing the risk of runaway reactions and enhancing overall process safety. The use of THF, a common and recyclable solvent, aligns well with modern green chemistry principles and simplifies waste management protocols. The high selectivity of the reaction results in fewer byproducts, reducing the volume of hazardous waste generated and lowering disposal costs. These environmental and safety advantages not only ensure compliance with increasingly stringent regulations but also improve the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this hydroboration technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a reliable foundation for process evaluation. Understanding these details is essential for technical teams assessing the feasibility of integrating this method into their existing workflows. The responses cover key aspects such as catalyst loading, reaction scope, and operational parameters to ensure a comprehensive understanding of the technology's capabilities.
Q: What is the optimal catalyst loading for this hydroboration reaction?
A: According to patent CN111320644B, the optimal catalyst dosage is between 4 mol% and 5 mol% of n-butyllithium relative to the imine substrate.
Q: Does this method support substrates with electron-withdrawing groups?
A: Yes, the catalytic system demonstrates excellent universality, successfully processing imines with various substituents including halogens, methyl, and methoxy groups with yields up to 99%.
Q: What are the reaction conditions required for this synthesis?
A: The reaction proceeds efficiently at room temperature within 1 to 2 hours using tetrahydrofuran (THF) as the solvent, eliminating the need for harsh heating conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boronic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our technical team has thoroughly evaluated the n-butyllithium catalyzed hydroboration process and confirmed its potential for delivering high-purity boronic esters with exceptional efficiency. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to leveraging this innovative chemistry to provide our clients with superior quality intermediates that accelerate their drug development timelines.
We invite you to collaborate with us to explore how this cost-effective synthesis route can optimize your supply chain and reduce your overall manufacturing expenses. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating the tangible economic benefits of this technology. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. Together, we can drive innovation and efficiency in the production of next-generation pharmaceutical intermediates.
