Advanced Rare Earth Catalysis for Efficient Boronic Ester Manufacturing
Introduction to Next-Generation Boronic Ester Synthesis
The landscape of organoboron chemistry is undergoing a significant transformation driven by the urgent need for more sustainable and efficient synthetic methodologies in the pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is detailed in Chinese Patent CN108164555B, which discloses a novel synthesis method for boronic esters utilizing beta-diimine divalent rare earth borohydride complexes. This technology represents a paradigm shift from traditional harsh conditions to a highly active, mild catalytic system capable of facilitating the hydroboration of aldehydes with unprecedented efficiency. For global supply chain leaders and R&D directors, this innovation offers a compelling pathway to enhance the production of critical intermediates used extensively in Suzuki-Miyaura cross-coupling reactions. The core breakthrough involves the utilization of specific rare earth metals, such as ytterbium or samarium, coordinated within a robust beta-diimine ligand framework, creating a catalyst that operates with remarkable turnover frequencies.
The strategic importance of this patent lies in its ability to address long-standing bottlenecks in fine chemical manufacturing, particularly regarding reaction time and catalyst loading. Unlike conventional methods that often require elevated temperatures or stoichiometric amounts of reagents, this rare earth catalytic system achieves quantitative yields in mere minutes under ambient conditions. This capability not only accelerates the development timeline for new drug candidates but also provides a scalable solution for the commercial production of high-purity pharmaceutical intermediates. As the industry moves towards greener chemistry principles, the adoption of such rare earth-based catalytic systems positions forward-thinking manufacturers at the forefront of technological excellence, ensuring reliability and compliance with increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of boronic esters has relied on methods that present substantial operational and economic challenges for large-scale manufacturing. Traditional routes often involve the reaction of boron trichloride with alcohols or phenols, a process that, while theoretically quantitative, is plagued by the hazardous nature and limited availability of boron trichloride. Alternative approaches utilizing boric acid or borax require high temperatures and complex equilibrium management, leading to difficult product separation and significant energy consumption. Furthermore, the use of transition metal catalysts for hydroboration reactions, while effective, introduces the risk of heavy metal contamination in the final product, necessitating costly and time-consuming purification steps to meet the rigorous purity specifications demanded by the pharmaceutical industry. These legacy methods often suffer from poor atom economy and generate substantial waste streams, conflicting with modern sustainability goals.
The Novel Approach
In stark contrast, the methodology disclosed in the patent introduces a highly efficient catalytic cycle driven by beta-diimine divalent rare earth complexes. This novel approach enables the direct hydroboration of aldehydes with pinacol borane or catechol borane under exceptionally mild conditions, specifically at room temperature.  The reaction kinetics are remarkably fast, typically reaching completion within 5 to 15 minutes, which drastically reduces reactor occupancy time and increases throughput capacity. By employing a catalyst loading as low as 0.01 to 0.1 mol%, this system minimizes the introduction of extraneous materials into the reaction matrix, thereby simplifying the downstream purification process. This method effectively bypasses the need for harsh reagents and high-energy inputs, offering a streamlined route to diverse boronic ester derivatives that is both economically viable and environmentally superior.
The reaction kinetics are remarkably fast, typically reaching completion within 5 to 15 minutes, which drastically reduces reactor occupancy time and increases throughput capacity. By employing a catalyst loading as low as 0.01 to 0.1 mol%, this system minimizes the introduction of extraneous materials into the reaction matrix, thereby simplifying the downstream purification process. This method effectively bypasses the need for harsh reagents and high-energy inputs, offering a streamlined route to diverse boronic ester derivatives that is both economically viable and environmentally superior.
Mechanistic Insights into Rare Earth Catalyzed Hydroboration
The efficacy of this synthesis method is rooted in the unique electronic and steric properties of the beta-diimine divalent rare earth borohydride complex. 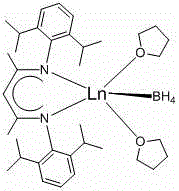 The catalyst features a central lanthanide ion, specifically ytterbium (Yb) or samarium (Sm), stabilized by a bulky beta-diimine ligand with 2,6-diisopropylphenyl substituents. This structural arrangement creates a highly Lewis acidic metal center that is perfectly tuned to activate the boron-hydrogen bond of the borane reagent while simultaneously coordinating the carbonyl oxygen of the aldehyde substrate. The resulting transition state facilitates a concerted hydride transfer mechanism that is both rapid and highly selective. The presence of the tetrahydrofuran (THF) molecules in the coordination sphere further modulates the reactivity, ensuring stability during storage while allowing for facile ligand exchange during the catalytic cycle. This precise molecular engineering allows the catalyst to maintain high activity even at ultra-low concentrations, a feature that is critical for cost-effective industrial application.
The catalyst features a central lanthanide ion, specifically ytterbium (Yb) or samarium (Sm), stabilized by a bulky beta-diimine ligand with 2,6-diisopropylphenyl substituents. This structural arrangement creates a highly Lewis acidic metal center that is perfectly tuned to activate the boron-hydrogen bond of the borane reagent while simultaneously coordinating the carbonyl oxygen of the aldehyde substrate. The resulting transition state facilitates a concerted hydride transfer mechanism that is both rapid and highly selective. The presence of the tetrahydrofuran (THF) molecules in the coordination sphere further modulates the reactivity, ensuring stability during storage while allowing for facile ligand exchange during the catalytic cycle. This precise molecular engineering allows the catalyst to maintain high activity even at ultra-low concentrations, a feature that is critical for cost-effective industrial application.
From an impurity control perspective, the mildness of this catalytic system is a decisive advantage for producing high-purity intermediates. Because the reaction proceeds at room temperature, there is minimal thermal stress on the substrate, which significantly reduces the formation of side products such as aldol condensation byproducts or over-reduced species that are common in high-temperature processes. The chemoselectivity of the rare earth center ensures that sensitive functional groups on the aromatic ring, such as halogens, nitro groups, or methoxy groups, remain intact throughout the transformation. This high fidelity in chemical transformation means that the crude product obtained after solvent removal is of exceptional quality, often requiring only a simple wash with n-hexane to remove residual high-boiling solvents. For R&D teams focused on impurity profiling, this translates to a cleaner reaction profile and a more straightforward path to regulatory approval for new drug substances.
How to Synthesize Boronic Esters Efficiently
Implementing this advanced catalytic technology into a production workflow requires adherence to specific operational protocols to maximize yield and safety. The process is designed to be operationally simple, leveraging standard laboratory and plant equipment without the need for specialized high-pressure or high-temperature reactors. The key to success lies in maintaining strict anhydrous and oxygen-free conditions, as the divalent rare earth species are sensitive to moisture and air. The procedure involves the sequential addition of the catalyst, borane reagent, and aldehyde substrate in an inert solvent environment, followed by a short reaction period. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction environment under strict anhydrous and oxygen-free conditions using an inert gas atmosphere.
- Sequentially mix the beta-diimine rare earth catalyst, borane reagent, and aldehyde substrate in a suitable solvent.
- Allow the reaction to proceed at room temperature for 5-15 minutes, followed by solvent removal and hexane washing to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rare earth catalytic technology offers profound strategic benefits that extend beyond simple reaction efficiency. The primary value proposition is the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures and enhanced supply chain resilience. By eliminating the need for expensive transition metal catalysts and the associated heavy metal scavenging resins, manufacturers can achieve significant cost reduction in fine chemical manufacturing. The ultra-low catalyst loading means that the cost contribution of the catalyst to the final product is negligible, allowing for more competitive pricing structures in a market where margin compression is a constant challenge. Furthermore, the simplicity of the workup procedure reduces the consumption of solvents and consumables, contributing to a leaner and more sustainable production model.
- Cost Reduction in Manufacturing: The economic impact of switching to this rare earth catalytic system is substantial due to the elimination of multiple cost drivers inherent in traditional synthesis. The catalyst loading of 0.01-0.1 mol% is orders of magnitude lower than many conventional catalytic systems, meaning less capital is tied up in catalyst inventory. Additionally, the absence of heavy metals removes the necessity for costly purification steps such as column chromatography or specialized filtration, which are often bottlenecks in production schedules. The energy savings derived from running reactions at room temperature rather than reflux conditions further compound these financial benefits, leading to a lower overall cost of goods sold. This efficiency allows suppliers to offer more attractive pricing while maintaining healthy margins, a critical factor for long-term contracts with major pharmaceutical clients.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex synthesis routes that are prone to variability and failure. This new method enhances reliability by utilizing readily available starting materials such as simple aldehydes and commercially sourced boranes, reducing dependency on exotic or hard-to-source reagents. The robustness of the catalyst across a wide range of substrates, including those with steric hindrance or electron-withdrawing groups, ensures consistent output quality regardless of feedstock variations. The short reaction time of 5 to 15 minutes significantly increases asset utilization, allowing facilities to produce larger volumes in shorter timeframes, thereby reducing lead times for high-purity pharmaceutical intermediates. This agility enables suppliers to respond more rapidly to fluctuating market demands and urgent customer requests.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen complications, but the mild nature of this hydroboration reaction mitigates many scale-up risks. The exothermicity of the reaction is manageable under ambient conditions, reducing the need for complex cooling systems and enhancing process safety. From an environmental standpoint, the atom-efficient nature of the hydroboration reaction combined with the simple aqueous-free workup minimizes waste generation, aligning with green chemistry principles. The avoidance of toxic boron trichloride and the reduction in solvent usage contribute to a smaller environmental footprint, helping companies meet increasingly strict regulatory compliance standards. This sustainability profile is becoming a key differentiator in vendor selection processes for multinational corporations committed to carbon neutrality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rare earth catalytic technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing production lines.
Q: What represents the primary advantage of this rare earth catalyst over traditional transition metals?
A: The primary advantage lies in the exceptionally low catalyst loading required (0.01-0.1 mol%) and the ability to operate at room temperature, which drastically reduces energy consumption and eliminates the need for expensive heavy metal scavenging processes often associated with transition metal catalysts.
Q: Is this synthesis method compatible with sensitive functional groups?
A: Yes, the mild reaction conditions (room temperature) and the specific electronic nature of the beta-diimine ligand system allow for excellent tolerance of various substituents including halogens, nitro groups, and methoxy groups without causing side reactions or decomposition.
Q: How does the workup procedure impact overall production efficiency?
A: The workup is significantly simplified compared to traditional methods; it involves simple solvent removal under reduced pressure followed by washing with n-hexane to remove high-boiling solvents, avoiding complex chromatographic separations and enabling faster batch turnover.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boronic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic technologies is key to maintaining competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN108164555B are fully realized in practical manufacturing settings. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of boronic ester intermediate meets the exacting standards required for API synthesis. Our commitment to technical excellence allows us to navigate the complexities of rare earth chemistry safely and efficiently, delivering products that empower our clients' drug development pipelines.
We invite you to collaborate with us to leverage these cutting-edge synthetic advantages for your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this technology can optimize your supply chain. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a reliable pharmaceutical intermediates supplier dedicated to driving innovation and efficiency in your manufacturing operations.
