Industrial Scale-Up of Dabigatran Intermediates: A Technical Breakthrough for Global Supply Chains
Industrial Scale-Up of Dabigatran Intermediates: A Technical Breakthrough for Global Supply Chains
The pharmaceutical industry constantly seeks robust methodologies for producing high-value anticoagulants, and the synthesis of Dabigatran and its prodrug Dabigatran Etexilate remains a critical area of focus for generic manufacturers and innovators alike. Patent CN103025715A introduces a transformative approach to preparing the key diamino intermediate required for these active pharmaceutical ingredients, addressing long-standing issues related to purification and scalability. This technical insight report analyzes the novel process which utilizes inorganic base-assisted catalytic hydrogenation and a unique hydrobromide salt intermediate to bypass chromatographic purification. By shifting from complex organic amine promoters to simple inorganic carbonates or phosphates, the disclosed method offers a pathway to significantly cleaner reaction profiles and improved operational efficiency. For R&D directors and supply chain managers, understanding this shift is vital for optimizing production costs and ensuring consistent quality in the final API.
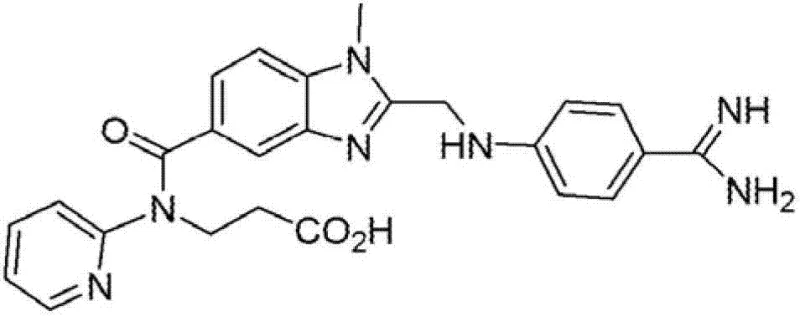
The core value proposition of this patent lies in its ability to produce the critical intermediate, ethyl N-(3-amino-4-methylaminobenzoyl)-N-(2-pyridyl)-3-aminopropanoate, with exceptional purity levels exceeding 99% without the need for column chromatography. Traditional methods often struggle with the reduction of the nitro precursor, leading to incomplete reactions or difficult-to-remove byproducts that necessitate expensive purification steps. The new methodology described in the patent leverages the formation of a crystalline hydrobromide salt of the nitro precursor, which acts as a highly effective purification checkpoint before the reduction step even begins. This strategic intervention ensures that the hydrogenation catalyst operates on a substrate of known high quality, minimizing side reactions and maximizing the yield of the desired diamino product. Such process intensification is exactly what modern pharmaceutical manufacturing requires to remain competitive in a cost-sensitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dabigatran intermediates has relied on protocols described in earlier patents such as WO98/37075 and WO2006/000353, which utilize catalytic hydrogenation in the presence of organic amines or require chromatographic purification of the nitro precursor. These conventional approaches present significant bottlenecks when attempted at an industrial scale, primarily due to the difficulty in completely removing the organic amine additives and the inherent inefficiency of chromatography for multi-kilogram batches. The reliance on chromatography not only drives up the cost of goods sold through solvent consumption and silica gel usage but also introduces variability in batch-to-batch purity which is unacceptable for regulatory compliance. Furthermore, the hydrogenation steps in prior art often suffer from incomplete conversion, leaving residual nitro compounds that are genotoxic impurities of high concern, thereby complicating the safety profile of the final drug substance.
In addition to purification challenges, the use of tertiary amines like triethylamine or DBU in the hydrogenation step creates downstream processing headaches, as these bases can coordinate with metal catalysts or form salts that are difficult to separate from the product stream. The environmental footprint of these older methods is also substantial, given the large volumes of waste solvents generated during the purification phases. For a procurement manager, these factors translate into higher raw material costs, longer lead times due to complex work-ups, and increased waste disposal fees. The inability to simply filter and dry the intermediate means that production throughput is limited by the capacity of purification equipment rather than reaction kinetics, creating a significant constraint on supply chain agility and responsiveness to market demand.
The Novel Approach
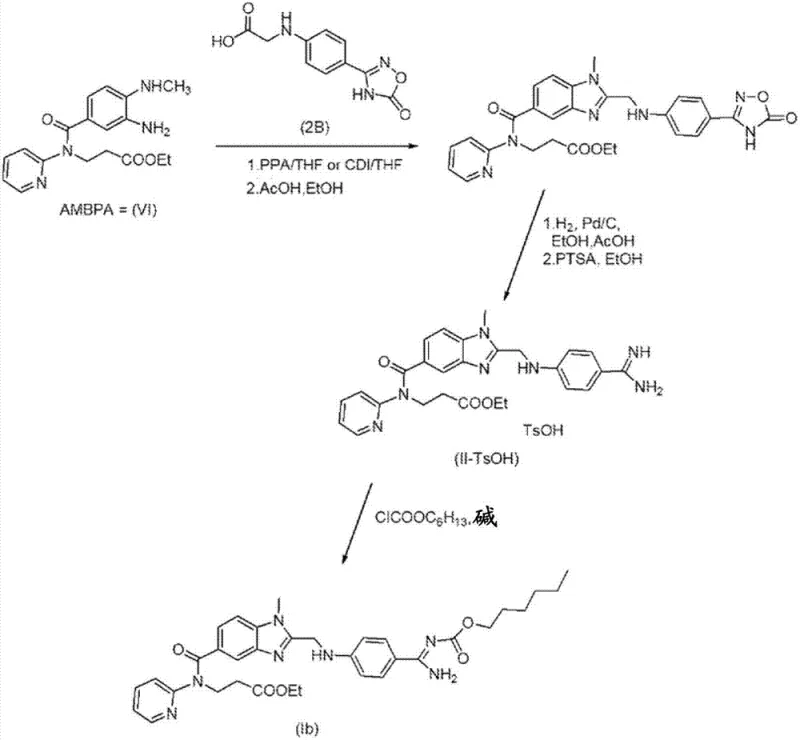
The innovative process detailed in CN103025715A fundamentally reengineers the synthesis by introducing an inorganic base during the catalytic hydrogenation of the nitro compound, effectively eliminating the need for problematic organic amines. This switch to inorganic bases such as potassium carbonate or potassium phosphate not only reduces raw material costs but also simplifies the reaction work-up, as these salts are easily removed by filtration or aqueous washes. The patent further discloses a novel solid form of the nitro precursor, specifically the hydrobromide salt (VII-HBr), which can be isolated in high purity through crystallization, thereby obviating the need for chromatography entirely. This crystalline intermediate serves as a robust starting material for the reduction step, ensuring that the hydrogenation proceeds to completion with minimal formation of side products.
By integrating the salt formation and inorganic base hydrogenation, the new method achieves a level of process robustness that is ideal for commercial scale-up. The reaction conditions are mild, typically operating at temperatures between 50-60°C and pressures around 4 bar, which are well within the safety limits of standard industrial hydrogenation reactors. The resulting diamino intermediate is obtained in high yield and purity, ready for direct conversion into the benzimidazole core of Dabigatran without further purification. This streamlined workflow represents a paradigm shift in how this critical anticoagulant intermediate is manufactured, offering a clear path to cost reduction in pharmaceutical manufacturing while enhancing the reliability of the supply chain for this essential medication.
Mechanistic Insights into Inorganic Base-Assisted Catalytic Hydrogenation
The mechanistic advantage of using inorganic bases in the hydrogenation of nitro-aromatic compounds lies in their ability to neutralize acidic byproducts and maintain a favorable pH environment for the catalyst without interfering with the metal surface. Unlike organic amines which can adsorb onto the palladium catalyst and poison its activity, inorganic carbonates and phosphates remain largely in the solution phase or as suspended solids, allowing for unhindered access of hydrogen and the substrate to the active sites. This results in a faster reaction rate and more complete conversion of the nitro group to the amine, effectively suppressing the accumulation of hydroxylamine intermediates which are often precursors to azo-type impurities. The patent specifies the use of solvents like ethyl acetate, which provides excellent solubility for the organic substrate while being compatible with the heterogeneous catalyst system.
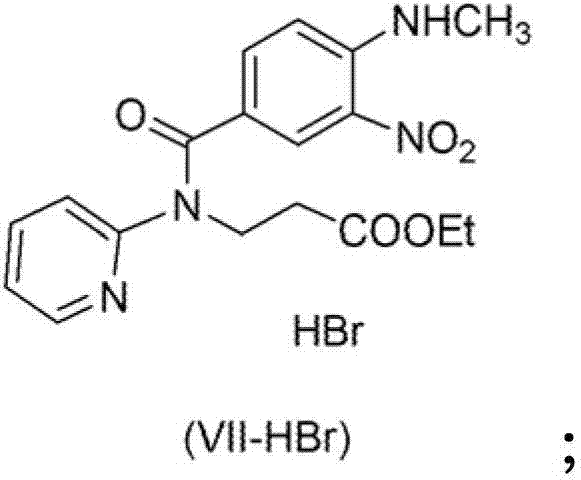
Furthermore, the isolation of the nitro precursor as a hydrobromide salt (VII-HBr) plays a crucial role in impurity control by leveraging the differential solubility of the salt versus potential organic impurities. The crystallization of this salt from solvents like tetrahydrofuran or ethanol allows for the rejection of non-basic impurities and unreacted starting materials in the mother liquor. When this high-purity salt is subsequently neutralized to the free base for hydrogenation, the input material quality is assured, which directly correlates to the purity of the final diamino product. This two-stage purification strategy, combining salt crystallization followed by selective catalytic reduction, creates a powerful barrier against impurity propagation, ensuring that the final intermediate meets the stringent specifications required for API synthesis.
How to Synthesize Dabigatran Intermediate Efficiently
The synthesis of the key diamino intermediate involves a sequence of coupling, salt formation, and catalytic hydrogenation that has been optimized for industrial feasibility. The process begins with the acylation of an aminopyridine derivative followed by the precise formation of the hydrobromide salt to secure high purity. Subsequent hydrogenation under controlled pressure and temperature with an inorganic base promoter yields the target diamino compound in excellent quality. Detailed standardized operating procedures for each step, including specific solvent ratios and catalyst loadings, are essential for replicating the high yields reported in the patent literature. The following guide outlines the critical stages of this optimized synthetic route.
- Preparation of Nitro Compound (VII) followed by precipitation as Hydrobromide Salt (VII-HBr) to ensure high purity without chromatography.
- Catalytic hydrogenation of the free base of (VII) in the presence of an inorganic base such as K2CO3 or K3PO4 using Pd/C catalyst.
- Isolation of the diamino intermediate (VI) through filtration and solvent removal, achieving high purity suitable for downstream coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain leaders, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere technical elegance. The elimination of chromatographic purification steps drastically reduces the consumption of silica gel and organic solvents, leading to substantial cost savings in raw materials and waste management. Additionally, the use of inexpensive inorganic bases instead of specialized organic amines lowers the bill of materials, while the improved reaction kinetics shorten the overall cycle time per batch. These efficiencies translate into a more competitive cost structure for the final API, allowing manufacturers to offer better pricing to healthcare systems without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The removal of chromatography is the single most impactful cost-saving measure in this process, as it eliminates a low-throughput, high-waste operation that traditionally bottlenecks production. By replacing this with a crystallization step, the process becomes continuous-flow friendly and scalable, reducing labor costs and equipment occupancy time. Furthermore, the switch to inorganic bases reduces the cost of reagents and simplifies the quenching and extraction phases, further driving down the operational expenditure associated with each kilogram of intermediate produced.
- Enhanced Supply Chain Reliability: The robustness of the inorganic base hydrogenation method ensures consistent batch-to-batch quality, reducing the risk of production failures or out-of-specification results that can disrupt supply. The ability to isolate stable crystalline intermediates allows for inventory buffering, decoupling the upstream synthesis from downstream coupling reactions and providing greater flexibility in production scheduling. This stability is crucial for maintaining a reliable supply of Dabigatran intermediates, especially in a market where demand for anticoagulants remains steady and predictable.
- Scalability and Environmental Compliance: The process operates at moderate pressures and temperatures, making it safe and easy to scale from pilot plant to multi-ton commercial production using standard reactor infrastructure. The reduction in solvent usage and the avoidance of toxic organic amines align with green chemistry principles, simplifying environmental permitting and reducing the carbon footprint of the manufacturing site. This sustainability angle is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals and regulatory expectations regarding environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for technology transfer teams and process engineers who are evaluating the feasibility of adopting this method for commercial production. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: Why is the inorganic base hydrogenation method superior to traditional amine-based methods?
A: The use of inorganic bases like potassium carbonate eliminates the need for expensive and toxic organic amines, simplifies the work-up procedure through easy filtration, and ensures complete conversion of the nitro group, thereby reducing impurity profiles significantly.
Q: How does the hydrobromide salt form (VII-HBr) improve the manufacturing process?
A: Converting the nitro intermediate into its hydrobromide salt allows for isolation via crystallization rather than chromatography. This solid form exhibits excellent filtration properties, leading to higher overall yields and easier handling on an industrial scale.
Q: What are the typical reaction conditions for the key hydrogenation step?
A: The hydrogenation is typically conducted at moderate temperatures between 50-60°C and pressures around 4 bar using a Pd/C catalyst in solvents like ethyl acetate, ensuring safety and scalability for large batch production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-demand pharmaceutical intermediates like those for Dabigatran. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate we produce meets the highest international standards for safety and efficacy. Our commitment to process excellence means we can deliver the cost and quality advantages described in this technical insight directly to your supply chain.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our optimized production methods. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics rather than theoretical projections. Let us help you secure a sustainable and cost-effective supply of critical anticoagulant intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
