Advancing Pharmaceutical Intermediate Production: A Safer, High-Yield Route to Beta-Nitrostyrene Compounds
The pharmaceutical and fine chemical industries are constantly seeking robust manufacturing processes that balance high efficiency with uncompromising safety standards, particularly when handling energetic materials like nitro compounds. Patent CN101765579A introduces a transformative method for producing beta-nitrostyrene compounds, which serve as critical intermediates for therapeutic agents such as baclofen and antispasmodics. This technology addresses the longstanding industrial challenge of thermal runaway risks associated with nitromethane condensations by shifting from traditional ammonium acetate catalysts to a primary amine system. By utilizing benzaldehyde derivatives and nitromethane in an acetic acid solvent, this novel approach achieves exceptional yields within a strictly controlled, industrially safe temperature window. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more reliable pharma intermediate supplier capabilities, offering a pathway to reduce production costs while enhancing operational safety profiles.
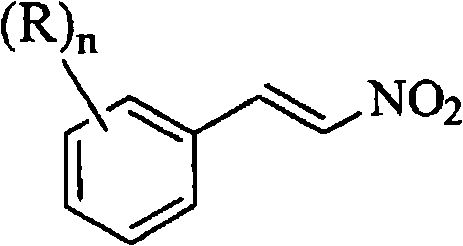
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-nitrostyrene compounds has relied heavily on methods utilizing ammonium acetate as a base, often necessitating heating under reflux conditions. As detailed in prior art such as JP 9-194457 and JP 11-71348, these conventional processes require reaction temperatures approaching the boiling point of the solvent, creating significant thermal hazards. Accelerating Rate Calorimeter (ARC) data cited in the background of CN101765579A reveals a critical safety flaw: at an operating temperature of 95°C, the time to maximum self-reaction rate (runaway) is merely 7.9 hours, which is dangerously close to the typical 12-hour residence time of the reaction. This narrow margin for error poses severe risks for commercial scale-up of complex pharmaceutical intermediates, potentially leading to catastrophic equipment failure or uncontrolled exothermic events. Furthermore, these older methods often struggle with yield consistency, typically capping around 67% to 81%, which generates substantial chemical waste and increases the cost of goods sold.
The Novel Approach
In stark contrast, the methodology disclosed in CN101765579A utilizes a primary amine, preferably benzylamine, as a catalyst within an acetic acid solvent system. This strategic modification allows the condensation reaction between the benzaldehyde derivative and nitromethane to proceed efficiently at a significantly lower temperature range of 70-80°C. By operating well below the boiling point and within a thermally stable window, the process effectively mitigates the risk of runaway reactions, as evidenced by an Adiabatic Decomposition Temperature for 24 hours (ADT24) of 78°C. This means that even under adiabatic conditions, the reaction remains stable for a full day, providing a massive safety buffer for large-scale manufacturing. Additionally, the use of acetic acid simplifies the downstream processing; unlike toluene-based systems, the addition of water to the acetic acid reaction mixture directly induces crystallization of the product, eliminating complex extraction steps and facilitating cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Primary Amine-Catalyzed Condensation
The core of this technological advancement lies in the specific interaction between the primary amine catalyst and the carbonyl group of the benzaldehyde derivative. In the presence of a primary amine such as benzylamine, the reaction likely proceeds through the formation of an imine or Schiff base intermediate, which activates the carbonyl carbon towards nucleophilic attack by the nitromethane anion. This activation lowers the energy barrier for the Henry reaction (nitroaldol condensation) followed by dehydration, allowing the transformation to occur rapidly at moderate temperatures of 70-80°C. The patent specifies that the molar ratio of the primary amine to the benzaldehyde derivative is critical, with an optimal range of 0.2 to 1.5 moles per mole of aldehyde. Using benzylamine at a 1:1 ratio, for instance, drives the reaction to completion within a few hours, whereas reducing the catalyst loading to 0.2 moles extends the reaction time but still maintains viable conversion rates. This mechanistic flexibility allows process chemists to tune the reaction kinetics based on specific production constraints without sacrificing the integrity of the final product.
Beyond kinetics, the choice of acetic acid as the solvent plays a dual role in both reaction mediation and product isolation. Acetic acid acts as a polar protic solvent that stabilizes the transition states involved in the condensation while remaining miscible with water. This property is exploited during the workup phase, where the addition of water at 30-60°C reduces the solubility of the beta-nitrostyrene compound, causing it to precipitate out of the solution as high-purity crystals. This crystallization mechanism is superior to oiling out, a common issue in nitro-compound synthesis that complicates purification. By controlling the cooling rate and water addition speed, manufacturers can achieve a particle size distribution that is ideal for filtration and drying. This level of control over the solid-state properties is essential for meeting the stringent purity specifications required for high-purity OLED material or pharmaceutical applications, ensuring that impurities are excluded from the crystal lattice during formation.
How to Synthesize 4-Chloro-beta-nitrostyrene Efficiently
The synthesis of 4-chloro-beta-nitrostyrene serves as a prime example of the efficacy of this patented process, demonstrating how precise control over reaction parameters leads to superior outcomes. The procedure involves dissolving 4-chlorobenzaldehyde and benzylamine in glacial acetic acid, followed by the controlled dropwise addition of nitromethane while maintaining the temperature between 78°C and 80°C. This specific temperature control is vital to prevent the accumulation of unreacted nitromethane, which could lead to a sudden exotherm if the temperature spikes. Following the addition, the mixture is stirred to ensure complete conversion, after which water is introduced to trigger crystallization. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of reagent quantities and timing.
- Dissolve the benzaldehyde derivative and primary amine (preferably benzylamine) in an acetic acid solvent.
- Heat the solution to a safe reaction temperature range of 70-80°C and slowly add nitromethane.
- After reaction completion, add water to the mixture to induce crystallization of the beta-nitrostyrene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN101765579A offers profound strategic benefits that extend beyond simple yield improvements. The shift to a lower temperature process fundamentally alters the risk profile of the manufacturing facility, reducing the likelihood of unplanned shutdowns due to safety incidents. This enhanced operational stability translates directly into improved supply chain reliability, ensuring consistent delivery schedules for downstream customers who rely on these intermediates for their own API production. Furthermore, the simplified workup procedure, which leverages water-induced crystallization in acetic acid, significantly reduces the consumption of organic solvents like toluene during the isolation phase. This reduction in solvent usage not only lowers raw material costs but also diminishes the volume of hazardous waste requiring treatment, aligning with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The implementation of this primary amine-catalyzed route eliminates the need for high-energy reflux conditions, resulting in substantial energy savings during the heating phase of the batch cycle. Moreover, the dramatic increase in yield, reaching up to 97.1% in optimized examples compared to roughly 67% in older methods, means that less raw material is wasted per kilogram of finished product. This efficiency gain directly impacts the bottom line by lowering the effective cost per unit of the active pharmaceutical ingredient precursor. Additionally, the removal of expensive transition metal catalysts or complex purification columns further streamlines the cost structure, making the final product more competitive in the global market.
- Enhanced Supply Chain Reliability: Safety is the cornerstone of supply continuity, and the thermal stability provided by the ADT24 of 78°C ensures that production can proceed without the constant threat of thermal runaway. This safety margin allows for larger batch sizes and more aggressive production scheduling without compromising plant safety protocols. By mitigating the risk of catastrophic failure, manufacturers can guarantee longer campaign runs and reduce the frequency of maintenance interventions caused by thermal stress on reactor vessels. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing partners to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: The use of acetic acid and water creates a waste stream that is significantly easier to treat compared to mixed organic solvent wastes containing heavy metals or halogenated hydrocarbons. The aqueous mother liquor from the crystallization step can often be processed through standard wastewater treatment facilities with minimal pre-treatment, reducing the environmental footprint of the facility. From a scalability perspective, the exotherm is manageable and predictable, allowing for safe scale-up from pilot plants to multi-ton commercial reactors. This ease of scale-up ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly, supporting the rapid expansion needs of growing biotech and pharma clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of beta-nitrostyrene compounds using this advanced methodology. These insights are derived directly from the experimental data and safety analysis provided in the patent literature, offering clarity on process robustness and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the primary amine method safer than the conventional ammonium acetate method?
A: The primary amine method operates at a lower temperature (70-80°C) compared to the reflux conditions required for ammonium acetate. Patent data indicates an ADT24 of 78°C, ensuring thermal stability and preventing runaway reactions within the operating window.
Q: What represents the key advantage of using acetic acid as a solvent in this process?
A: Acetic acid facilitates a simplified post-reaction workup. Unlike toluene, adding water to the acetic acid reaction mixture directly induces crystallization of the beta-nitrostyrene compound, streamlining isolation and purification.
Q: What yields can be expected from this novel synthesis route?
A: Experimental data from the patent demonstrates significantly improved yields ranging from 80% to 99%, with specific examples achieving up to 97.1%, surpassing the 67-81% yields typical of older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-beta-nitrostyrene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the safety and efficiency benefits of the CN101765579A process are fully realized at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs to verify that every batch of beta-nitrostyrene meets the exacting standards required for pharmaceutical synthesis. Our infrastructure is designed to handle reactive nitro chemistry safely, utilizing advanced calorimetry and process control systems to monitor reaction parameters in real-time.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this primary amine-catalyzed method for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of high-quality intermediates into your drug development pipeline.
