Advanced Manufacturing Strategy for Sacubitril: A Cost-Effective Route for Global Supply Chains
Advanced Manufacturing Strategy for Sacubitril: A Cost-Effective Route for Global Supply Chains
The pharmaceutical landscape for heart failure treatment has been revolutionized by the introduction of Entresto, a dual inhibitor combining valsartan and sacubitril. As demand for this life-saving medication surges globally, the pressure on the supply chain to deliver high-purity active pharmaceutical ingredients (APIs) and intermediates efficiently has never been greater. Patent CN111943862A discloses a groundbreaking preparation method for sacubitril, the key component of Entresto, which addresses critical bottlenecks in traditional manufacturing. This novel approach utilizes 4-bromobiphenyl as a starting raw material, diverging significantly from established protocols that rely on expensive chiral precursors. By streamlining the synthetic pathway, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers seeking to optimize their production portfolios.
The structural complexity of sacubitril, chemically known as 4-(((2S,4R)-1-(1,1'-biphenyl-4-yl)-5-ethoxy-4-methyl-5-oxopentan-2-yl)amino)-4-oxobutanoic acid, presents unique challenges in stereochemical control and functional group manipulation. The patented method overcomes these hurdles through a clever sequence of Grignard reactions, Mitsunobu couplings, and selective hydrolysis. This technical breakthrough not only simplifies the operational route but also aligns with the industry's push towards greener and more cost-effective chemical processes. For procurement managers and R&D directors alike, understanding the nuances of this synthesis is essential for securing a competitive edge in the cardiovascular drug market.
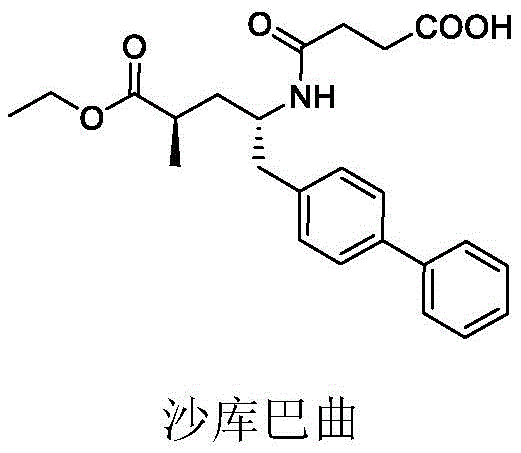
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of sacubitril was predominantly governed by routes described in patents such as US5217996 and WO2008031567. These conventional methods typically initiate with chiral amino alcohols, which are inherently expensive and often require multi-step synthesis themselves before they can even enter the main reaction sequence. The reliance on Wittig reactions and chiral hydrogenation introduces significant complexity, requiring specialized catalysts and stringent reaction conditions that are difficult to maintain on a large scale. Furthermore, the cumulative yield losses across these numerous steps result in a final product with a prohibitively high cost basis, limiting the ability of manufacturers to offer competitive pricing in a price-sensitive generic market.
Another critical drawback of the traditional approach is the difficulty in controlling impurities and stereochemistry throughout the lengthy process. Each additional transformation step increases the risk of racemization or the formation of hard-to-remove byproducts, necessitating rigorous and costly purification protocols. For supply chain heads, this translates to longer lead times and higher inventory costs due to the need for extensive quality control testing. The dependency on scarce chiral starting materials also creates a vulnerability in the supply chain, where any disruption in the availability of these precursors can halt entire production lines, posing a significant risk to supply continuity for major pharmaceutical clients.
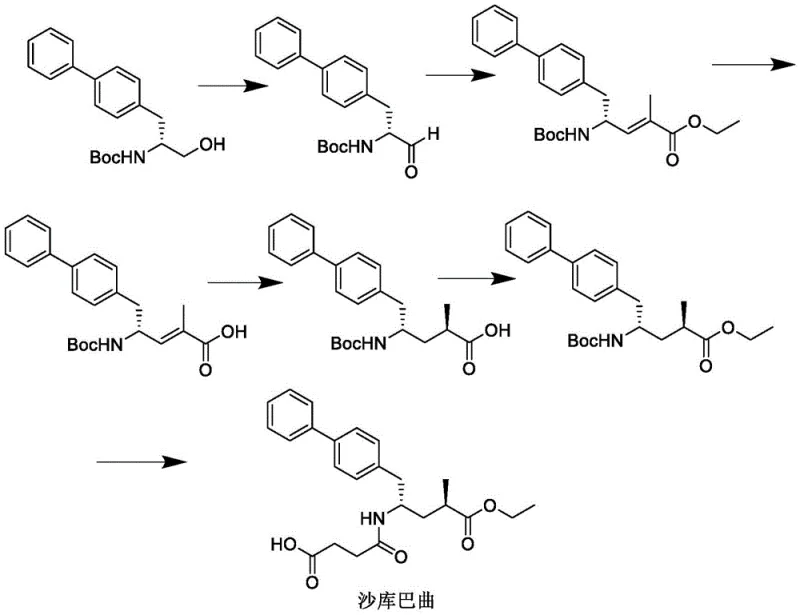
The Novel Approach
In stark contrast, the method disclosed in CN111943862A reinvents the synthesis by starting from 4-bromobiphenyl, a commodity chemical that is abundant and inexpensive. This strategic shift eliminates the need for pre-synthesized chiral amino alcohols, effectively cutting out several upstream steps and their associated costs. The new route employs a Grignard reaction with (S)-epichlorohydrin to establish the necessary chiral center early in the process, followed by a Mitsunobu reaction to introduce the nitrogen functionality. This streamlined sequence reduces the total number of operational units required, thereby minimizing equipment footprint and energy consumption while maximizing overall throughput efficiency.
The simplicity of this novel approach extends to the reaction conditions, which are mild and amenable to standard industrial reactor setups. Unlike the high-pressure hydrogenation required in older methods, this process utilizes liquid-phase reactions at atmospheric pressure and moderate temperatures, significantly enhancing operational safety and reducing capital expenditure on specialized high-pressure equipment. The result is a robust manufacturing protocol that delivers high yields at each stage, as evidenced by the experimental data showing yields consistently above 85%. For a reliable pharmaceutical intermediate supplier, adopting this route means being able to offer high-purity sacubitril at a fraction of the traditional cost, fundamentally changing the economics of Entresto production.
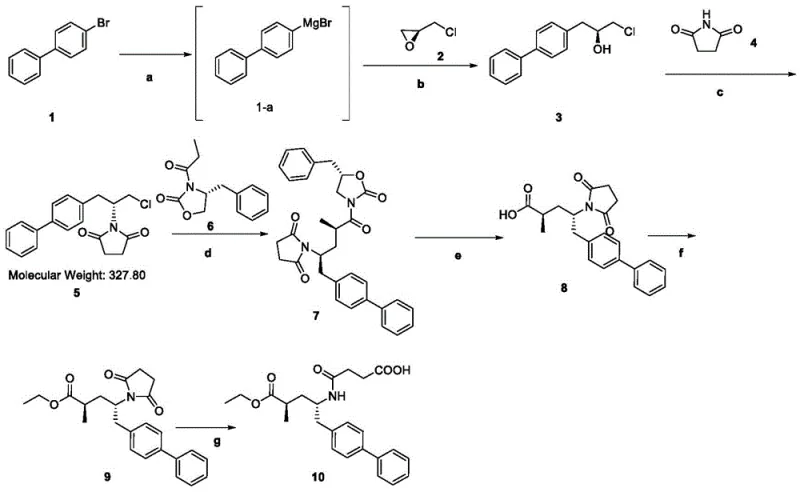
Mechanistic Insights into Grignard and Mitsunobu Coupling Strategies
The cornerstone of this synthesis is the initial formation of the chiral backbone via a copper-catalyzed Grignard reaction. In step 'a' and 'b', 4-bromobiphenyl is converted into a Grignard reagent, which then reacts with (S)-epichlorohydrin in the presence of cuprous iodide. The use of a copper catalyst is critical here, as it facilitates the regioselective opening of the epoxide ring, ensuring that the Grignard reagent attacks the less hindered carbon atom. This precision is vital for maintaining the integrity of the chiral center introduced by the epichlorohydrin, setting the stereochemical trajectory for the entire molecule. The subsequent hydrolysis and workup procedures are optimized to recover the chlorohydrin intermediate (Compound 3) with exceptional purity, laying a solid foundation for downstream transformations.
Following the establishment of the carbon skeleton, the synthesis proceeds through a Mitsunobu reaction in step 'c', where the hydroxyl group of Compound 3 is displaced by succinimide. This reaction, mediated by triphenylphosphine and DIAD, is highly efficient for inverting stereochemistry or introducing nitrogen nucleophiles under mild conditions. The resulting imide is then alkylated with a specific electrophile (Compound 6) in the presence of titanium tetrachloride, a Lewis acid that activates the electrophile for nucleophilic attack. This step constructs the complex carbon-nitrogen framework of the target molecule with high fidelity. Finally, the oxidative cleavage of the imide ring using hydrogen peroxide and lithium hydroxide reveals the free amine and carboxylic acid functionalities, completing the core structure of sacubitril with minimal side reactions.
How to Synthesize Sacubitril Efficiently
The synthesis of sacubitril via this patented route involves a series of well-defined chemical transformations that convert simple starting materials into the complex target molecule. The process begins with the generation of a Grignard reagent from 4-bromobiphenyl, followed by its reaction with a chiral epoxide to set the stereochemistry. Subsequent steps involve nitrogen insertion via Mitsunobu chemistry, chain elongation, and finally, functional group deprotection and esterification. Each step has been optimized for yield and purity, making the overall sequence highly attractive for commercial adoption. The detailed standardized synthesis steps below outline the specific conditions and reagents required to replicate this high-efficiency pathway in a production environment.
- Preparation of Compound 3 via Grignard reaction of 4-bromobiphenyl with (S)-epichlorohydrin using CuI catalyst.
- Mitsunobu reaction of Compound 3 with succinimide to form Compound 5, followed by alkylation with Compound 6 to yield Compound 7.
- Oxidative cleavage and hydrolysis of Compound 7 to Compound 8, followed by esterification and final ring-opening to obtain Sacubitril.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route represents a strategic opportunity to drastically reduce the cost of goods sold (COGS) for sacubitril production. By shifting away from expensive chiral amino alcohol starting materials to commodity chemicals like 4-bromobiphenyl, manufacturers can achieve substantial cost savings on raw material procurement. The elimination of complex chiral hydrogenation steps further reduces the dependency on precious metal catalysts, which are not only costly but also subject to volatile market pricing. This fundamental restructuring of the bill of materials allows for a more predictable and stable cost base, enabling companies to offer more competitive pricing to their downstream pharmaceutical partners without sacrificing margin.
- Cost Reduction in Manufacturing: The streamlined nature of this process directly translates to lower operational expenditures. With fewer reaction steps and simpler workup procedures, the consumption of solvents, energy, and labor is significantly reduced compared to conventional methods. The high yields reported in the patent examples mean that less raw material is wasted, improving the overall atom economy of the process. Furthermore, the avoidance of high-pressure equipment lowers maintenance costs and insurance premiums, contributing to a leaner and more profitable manufacturing operation that can withstand market fluctuations.
- Enhanced Supply Chain Reliability: Sourcing 4-bromobiphenyl and other basic reagents is far more reliable than procuring specialized chiral intermediates, which often have limited suppliers and long lead times. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Additionally, the robustness of the chemical reactions, which tolerate standard industrial conditions, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery schedules demanded by global pharmaceutical companies, thereby strengthening the supplier-client relationship.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations that are common in fine chemical plants, such as reflux, extraction, and crystallization. This ease of scale-up allows manufacturers to rapidly increase production capacity to meet surging demand without requiring massive capital investment in new infrastructure. Moreover, the reduction in step count and the use of less hazardous reagents contribute to a smaller environmental footprint. The simplified waste stream makes effluent treatment more manageable, helping companies comply with increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sacubitril synthesis method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is key for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of this supply source.
Q: What are the primary cost advantages of this new Sacubitril synthesis route?
A: The primary advantage lies in the use of 4-bromobiphenyl as a starting material, which is significantly cheaper and more readily available than the chiral amino alcohols required in conventional methods. Additionally, the process eliminates the need for expensive chiral hydrogenation catalysts and complex multi-step precursor syntheses, drastically reducing raw material and operational costs.
Q: How does this method ensure high chiral purity for pharmaceutical applications?
A: The method utilizes (S)-epichlorohydrin in the initial Grignard step to establish chirality early in the synthesis. Subsequent steps, particularly the Mitsunobu reaction and alkylation, proceed with high stereoselectivity. Experimental data from the patent indicates that the intermediate Compound 7 and final product achieve 100% chiral purity without the need for complex chiral resolution steps.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route is specifically designed for industrial scalability. It employs standard unit operations such as reflux, extraction, and crystallization, avoiding sensitive conditions like ultra-low temperatures or high-pressure hydrogenation. The reported yields for individual steps are consistently high (ranging from 85% to 95%), ensuring robust throughput for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sacubitril Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis route for the global heart failure medication market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for this process, including Grignard chemistry and sensitive hydrolysis steps, while adhering to the most stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to guarantee that every batch of sacubitril meets the highest international standards for pharmaceutical intermediates.
We invite forward-thinking pharmaceutical companies and procurement leaders to collaborate with us to leverage this cost-effective technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain needs. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments that demonstrate how our optimized manufacturing capabilities can drive value and security into your Entresto supply chain. Let us help you secure a sustainable and economical source for this critical life-saving ingredient.
