Optimizing Capecitabine Production Through Advanced Catalytic Glycosylation And Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for oncology therapeutics, particularly for oral prodrugs like Capecitabine which offer improved patient compliance over intravenous 5-FU. A pivotal advancement in this domain is documented in patent CN103193842A, which introduces a highly efficient synthesis method leveraging trifluoromethanesulfonic acid catalysis. This innovation addresses critical bottlenecks in nucleoside chemistry by enabling the direct utilization of alpha and beta isomer mixtures of 5-deoxy-triacetyl ribose without prior separation. For global supply chain stakeholders, this represents a paradigm shift from labor-intensive purification to streamlined catalytic conversion, ensuring a more reliable reliable Active Pharmaceutical Ingredients (APIs) supplier capability. The technical implications extend beyond mere yield improvements, fundamentally altering the cost structure and scalability potential of producing high-purity high-purity Oncology APIs for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Capecitabine has been hindered by the stringent stereochemical requirements of the glycosylation step. Conventional protocols typically mandate the use of pure beta-isomer 5-deoxy-triacetyl ribose to ensure the correct anomeric configuration in the final nucleoside product. Obtaining this pure beta-isomer necessitates complex and costly separation processes from the naturally occurring alpha/beta mixture, often involving repeated crystallizations or chromatographic techniques that drastically reduce overall mass efficiency. Furthermore, the discarded alpha-isomer represents a significant waste of raw materials, inflating the cost of goods sold and creating unnecessary environmental burden through solvent consumption. These inefficiencies compound at the commercial scale, where even minor yield losses translate into substantial financial deficits and extended lead times for commercial scale-up of complex Pharmaceutical Intermediates. The reliance on high-purity starting materials also introduces supply chain fragility, as any disruption in the availability of the specific beta-isomer can halt entire production lines.
The Novel Approach
The methodology outlined in the referenced patent circumvents these historical constraints by employing a powerful trifluoromethanesulfonic acid catalyst system that tolerates and actively utilizes the isomeric mixture. Instead of discarding the alpha-isomer, the catalytic conditions promote its in-situ conversion to the thermodynamically favored beta-configuration during the reaction progress. This dynamic kinetic resolution effectively doubles the utility of the ribose starting material, transforming a previously wasteful byproduct into a valuable reactant. By eliminating the pre-reaction purification step, the process significantly reduces operational complexity, solvent usage, and processing time. This approach not only enhances the economic viability of the synthesis but also aligns with modern green chemistry principles by minimizing waste generation. For procurement teams, this translates to a more resilient supply chain capable of cost reduction in Active Pharmaceutical Ingredients (APIs) manufacturing without compromising the stringent quality standards required for oncology medications.
Mechanistic Insights into Trifluoromethanesulfonic Acid-Catalyzed Glycosylation
The core of this technological breakthrough lies in the exceptional Lewis acidity of trifluoromethanesulfonic acid (TfOH), which activates the anomeric center of the ribose derivative for nucleophilic attack by 5-flucytosine. Unlike milder acids that might struggle to drive the reaction to completion with mixed isomers, TfOH provides the necessary energy to overcome the activation barrier for both alpha and beta forms. Mechanistically, the acid coordinates with the acetoxy groups, facilitating the formation of an oxocarbenium ion intermediate that is highly susceptible to nucleophilic substitution. Crucially, the acidic environment also catalyzes the mutarotation or anomerization of the alpha-glycoside to the beta-glycoside, ensuring that the final product distribution heavily favors the biologically active configuration. This dual function of promoting coupling and correcting stereochemistry is what allows the process to bypass the need for isomerically pure starting materials, representing a sophisticated application of physical organic chemistry to industrial problems.
Impurity control in this synthesis is managed through precise modulation of reaction parameters and workup procedures. The use of TfOH allows for milder temperature profiles compared to traditional harsh Lewis acids like tin tetrachloride, thereby reducing the formation of degradation byproducts and polymeric tars. Following the glycosylation, the subsequent steps involving pentyl chloroformate addition and alkaline hydrolysis are optimized to remove protecting groups without affecting the sensitive fluoropyrimidine moiety. The patent specifies rigorous monitoring via HPLC to ensure intermediate purity before proceeding, which is critical for preventing the carryover of impurities into the final API. This meticulous attention to reaction kinetics and purification ensures that the final Capecitabine meets the strict regulatory specifications for residual solvents and related substances, a key concern for R&D Director stakeholders evaluating technology transfer feasibility.
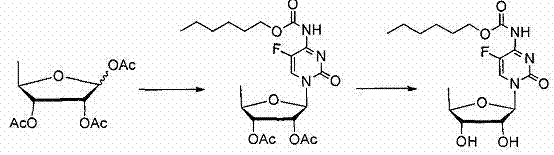
How to Synthesize Capecitabine Efficiently
Implementing this synthesis route requires careful adherence to the specified stoichiometric ratios and temperature controls to maximize the benefits of the catalytic system. The process begins with the silylation of 5-flucytosine to enhance its nucleophilicity, followed by the critical glycosylation step where the mixed ribose is introduced under controlled cooling to manage the exotherm. Detailed standard operating procedures regarding the addition rates of trifluoromethanesulfonic acid and the subsequent quenching protocols are essential for maintaining safety and reproducibility at scale. Operators must ensure that the reaction mixture is monitored closely to determine the exact endpoint, preventing over-reaction which could lead to decomposition. The following guide outlines the critical operational phases derived from the patent data to assist technical teams in replicating this high-efficiency pathway.
- Glycosylation Reaction: React 5-flucytosine with a mixture of alpha and beta 5-deoxy-1,2,3-triacetoxy-D-ribose using trifluoromethanesulfonic acid as a catalyst to form the nucleoside intermediate.
- Intermediate Isolation: Perform aqueous workup and crystallization to isolate 2',3'-diacetoxy-5'-deoxy-5-fluorocytidine with high purity.
- Carbamate Formation and Deprotection: React the intermediate with pentyl chloroformate followed by alkaline hydrolysis to yield the final Capecitabine API.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this catalytic methodology offers profound advantages that extend well beyond the laboratory bench. By removing the dependency on pre-separated beta-isomer ribose, manufacturers can source cheaper, readily available mixed-isomer raw materials, thereby insulating the production cost from the volatility of specialized reagent markets. This simplification of the bill of materials reduces the number of vendors required and minimizes the risk of supply disruptions caused by the unavailability of niche intermediates. Furthermore, the reduction in unit operations—specifically the elimination of the isomer separation step—shortens the overall manufacturing cycle time, allowing for faster turnover and improved responsiveness to market demand fluctuations. These factors collectively contribute to a more agile and cost-effective supply chain architecture.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic reduction in raw material costs achieved by utilizing the alpha/beta ribose mixture directly. Traditional methods incur significant expenses in purifying the beta-isomer and disposing of the alpha-waste, costs that are entirely eliminated in this new process. Additionally, the reduced number of processing steps lowers utility consumption, labor hours, and solvent recovery costs, leading to a substantially lower cost of goods sold. This efficiency allows for more competitive pricing strategies in the generic oncology market while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: Relying on mixed-isomer starting materials broadens the supplier base, as these commodities are more commonly produced and stocked than their purified counterparts. This diversification reduces the risk of single-source dependency and ensures a continuous flow of materials even during periods of market tightness. The robustness of the catalytic reaction also means that production schedules are less prone to delays caused by difficult purification bottlenecks, ensuring consistent on-time delivery performance for downstream partners.
- Scalability and Environmental Compliance: The streamlined nature of the synthesis makes it inherently easier to scale from pilot batches to multi-ton commercial production without encountering the nonlinear challenges often associated with complex separations. Fewer solvent exchanges and crystallization steps result in a smaller environmental footprint, simplifying waste management and regulatory compliance. This alignment with sustainability goals is increasingly important for multinational corporations seeking to reduce the carbon intensity of their pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent documentation to provide clarity for decision-makers. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential return on investment for adopting this catalytic route in your manufacturing portfolio.
Q: How does the new catalytic method reduce raw material costs compared to traditional synthesis?
A: Traditional methods require expensive purification to isolate the beta-isomer of 5-deoxy-triacetyl ribose. The patented method utilizes the crude alpha/beta mixture directly, as the catalyst facilitates in-situ conversion, significantly lowering raw material procurement costs.
Q: What is the role of trifluoromethanesulfonic acid in this synthesis?
A: Trifluoromethanesulfonic acid acts as a potent Lewis acid catalyst that promotes the glycosidic bond formation between 5-flucytosine and the ribose derivative. Crucially, it also catalyzes the anomerization of the alpha-isomer to the desired beta-configuration, improving overall yield.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process eliminates complex chromatographic separation steps and uses robust reaction conditions compatible with standard industrial reactors. The simplified workflow enhances scalability and reduces the environmental footprint associated with solvent-intensive purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable, and compliant synthesis routes for high-value oncology APIs like Capecitabine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced catalytic methods are fully realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international pharmacopoeia standards. Our commitment to process excellence allows us to offer a stable supply of high-quality intermediates and APIs, supporting our partners in bringing life-saving medications to patients faster and more affordably.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain strategy. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the specific economic benefits applicable to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the superior performance of our manufacturing capabilities. Let us collaborate to engineer a more resilient and cost-effective future for your pharmaceutical projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
