Revolutionizing Mirtazapine Production: A Green Resin-Catalyzed Cyclization Strategy for Scalable API Manufacturing
Revolutionizing Mirtazapine Production: A Green Resin-Catalyzed Cyclization Strategy for Scalable API Manufacturing
The global demand for high-quality antidepressants continues to surge, placing immense pressure on pharmaceutical supply chains to deliver Active Pharmaceutical Ingredients (APIs) with superior purity and environmental compliance. Mirtazapine, a noradrenergic and specific serotonergic antidepressant (NaSSA), remains a cornerstone therapy for major depressive disorder, necessitating robust and scalable manufacturing routes. A pivotal advancement in this domain is detailed in patent CN111187264A, which discloses a novel preparation method for mirtazapine and its critical intermediate. This technology shifts the paradigm from traditional, waste-intensive liquid acid catalysis to a sophisticated heterogeneous solid acid system. By leveraging sulfonic acid resins, this process addresses the chronic pain points of product separation and environmental pollution that have long plagued the industrial synthesis of this tetracyclic compound.
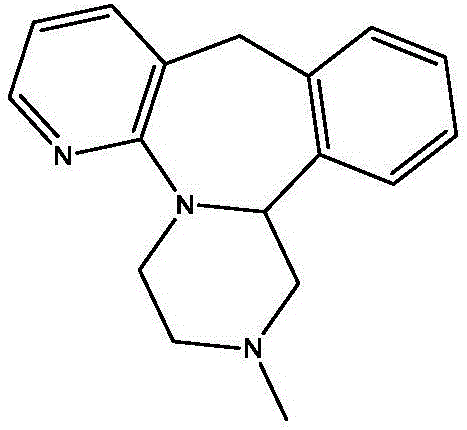
The strategic value of this patent lies in its ability to streamline the final cyclization step, which is historically the most chemically demanding phase of the synthesis. For R&D directors and process chemists, the transition to a resin-catalyzed system offers a cleaner reaction profile with minimized side reactions. For procurement and supply chain leaders, the implications are equally profound: the elimination of massive quantities of corrosive sulfuric acid translates directly into reduced hazardous waste disposal costs and simplified regulatory compliance. As we delve deeper into the technical specifics, it becomes evident that this methodology represents a significant leap forward in green chemistry for psychiatric medication manufacturing, aligning perfectly with the industry's shift towards sustainable and cost-effective production models.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of mirtazapine has relied heavily on the use of concentrated sulfuric acid to effect the critical dehydration cyclization of the piperazine precursor. While chemically effective, this conventional approach presents severe operational and environmental liabilities that are increasingly untenable in modern regulatory landscapes. The process typically requires the substrate to be dissolved or suspended in a large excess of concentrated sulfuric acid, which acts simultaneously as the solvent and the dehydrating agent. This creates a highly corrosive reaction environment that demands specialized, expensive equipment resistant to extreme acid conditions. Furthermore, the quenching and workup of such reactions are notoriously difficult; neutralizing vast volumes of hot, concentrated acid generates substantial amounts of saline waste and heat, posing safety risks and complicating downstream processing. The inability to recycle the sulfuric acid means that every batch produces a fresh load of acidic effluent, driving up waste treatment costs and carbon footprint significantly.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111187264A introduces a heterogeneous catalytic system that fundamentally reengineers the cyclization step. Instead of a liquid acid bath, the process utilizes solid sulfonic acid resins, such as macroporous strong acid polystyrene sulfonic acid resin or perfluorinated sulfonic acid resin (e.g., Nafion-H). This shift allows the reaction to proceed in mild organic solvents like ethylene glycol dimethyl ether, toluene, or chloroform, rather than in neat acid. The solid nature of the catalyst facilitates a straightforward separation protocol; once the reaction reaches completion, the resin can be simply filtered off, leaving the product in the solution or precipitated as a solid salt depending on the specific conditions. This not only simplifies the isolation of the mirtazapine intermediate but also opens the door for catalyst regeneration and reuse. The result is a process that is inherently safer, easier to control, and vastly more environmentally friendly than its predecessors.
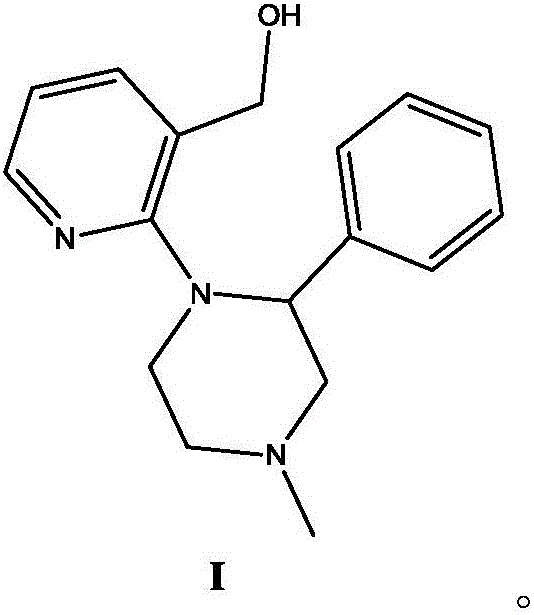
Mechanistic Insights into Sulfonic Acid Resin-Catalyzed Cyclization
The core of this innovation lies in the mechanistic efficiency of the solid acid catalyst in promoting intramolecular dehydration. The precursor, 1-(3-hydroxymethylpyridin-2-yl)-2-phenyl-4-methylpiperazine (Compound I), contains a hydroxymethyl group positioned ortho to the piperazine nitrogen on the pyridine ring. Under the influence of the sulfonic acid groups (-SO3H) anchored on the resin matrix, the hydroxyl group is protonated, converting it into a good leaving group (water). This activation triggers a nucleophilic attack by the adjacent piperazine nitrogen, leading to the formation of the new C-N bond that closes the fourth ring of the mirtazapine skeleton. The porous structure of the resin, particularly the macroporous varieties, provides a high surface area for this interaction, ensuring that the reaction kinetics remain favorable even in non-polar or moderately polar organic solvents. Unlike homogeneous acids which solvate the transition state indiscriminately, the resin surface can offer a unique microenvironment that may enhance selectivity and reduce the formation of polymeric byproducts often seen in harsh acid conditions.
From an impurity control perspective, this heterogeneous system offers distinct advantages. In traditional sulfuric acid methods, the aggressive oxidative and dehydrating nature of the medium can lead to charring, sulfonation of the aromatic rings, or over-degradation of the sensitive piperazine moiety. The milder acidity and controlled activity of the sulfonic acid resin mitigate these risks, resulting in a cleaner crude product profile. The patent data supports this, showing that the intermediate can be isolated as a solid with high purity prior to the final basification step. Furthermore, the subsequent ion exchange step—where the intermediate salt is treated with a mild base like sodium bicarbonate—ensures that any residual acidic species are neutralized and removed into the aqueous phase. This two-stage purification (filtration of resin followed by aqueous workup) effectively scrubs the product of catalyst residues and inorganic salts, yielding mirtazapine that consistently meets high purity specifications (e.g., >98.5%) without the need for complex chromatographic purification.
How to Synthesize Mirtazapine Efficiently
Implementing this resin-catalyzed route requires precise control over reaction parameters to maximize yield and catalyst life. The process begins with the preparation of the substrate, often as an inorganic acid salt like the hydrochloride, which is then suspended or dissolved in a suitable aprotic solvent. The choice of solvent is critical; while toluene and chloroform are effective, ethylene glycol dimethyl ether has shown particular promise in balancing solubility and reaction rate. The addition of the resin must be managed to control exotherms, with temperatures generally maintained between 60°C and 110°C depending on the solvent boiling point and resin type. Monitoring the reaction progress via TLC or HPLC is essential to prevent over-reaction. Once the cyclization is complete, the simplicity of the workup shines: the mixture is cooled, and the solid resin (along with the precipitated intermediate salt in some embodiments) is filtered. The final conversion to the free base is achieved through a gentle ion exchange with aqueous base, followed by standard extraction and recrystallization.
- Dissolve the intermediate compound I (or its inorganic acid salt) in an aprotic organic solvent such as ethylene glycol dimethyl ether or toluene under an inert atmosphere.
- Add the sulfonic acid resin catalyst (e.g., polystyrene sulfonic acid resin) to the reaction mixture and heat to 60-110°C to facilitate the cyclization dehydration.
- Filter the reaction mixture to separate the solid resin and intermediate, then treat the solid with an aqueous base solution to perform ion exchange and isolate the free base mirtazapine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resin-catalyzed technology offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic reduction of operational complexity and associated costs. By eliminating the need for handling and disposing of tons of concentrated sulfuric acid per ton of product, manufacturers can significantly lower their expenditure on hazardous waste management and corrosion-resistant infrastructure maintenance. This shift transforms a high-risk, high-cost unit operation into a manageable, standard organic synthesis step. Moreover, the potential for catalyst recycling introduces a variable cost saving that accumulates over large-scale production campaigns, enhancing the overall margin profile of the API.
- Cost Reduction in Manufacturing: The transition from homogeneous liquid acid to heterogeneous solid resin catalysis fundamentally alters the cost structure of mirtazapine production. Traditional methods incur hidden costs related to the neutralization of vast quantities of acidic waste, the disposal of saline byproducts, and the frequent replacement of equipment degraded by corrosive media. In this novel process, the solid catalyst can theoretically be regenerated and reused multiple times, turning a consumable expense into a capital asset. Additionally, the simplified workup reduces the consumption of auxiliary reagents and solvents required for extensive washing and neutralization, leading to substantial cost savings in raw materials and utility usage without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by regulatory crackdowns on environmental pollutants. Facilities utilizing heavy sulfuric acid processes face increasing scrutiny and potential shutdowns due to waste discharge limits. By adopting this greener resin-based technology, suppliers can future-proof their operations against tightening environmental regulations, ensuring a more stable and uninterrupted supply of mirtazapine intermediates. The ease of operation also reduces the risk of batch failures due to process upsets, providing buyers with greater confidence in delivery schedules and consistent quality. This reliability is crucial for pharmaceutical companies managing just-in-time inventory for critical antidepressant medications.
- Scalability and Environmental Compliance: Scaling up chemical processes involving concentrated sulfuric acid presents significant engineering challenges, particularly regarding heat dissipation and mixing efficiency in viscous, corrosive media. The resin-catalyzed method, operating in standard organic solvents with a solid catalyst, is inherently more scalable using conventional reactor setups. The reduction in hazardous waste generation aligns perfectly with global sustainability goals and corporate ESG (Environmental, Social, and Governance) mandates. This environmental compliance not only mitigates legal risks but also enhances the brand reputation of the supply chain partners, making them preferred vendors for multinational pharmaceutical corporations committed to green sourcing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is vital for technical teams evaluating the feasibility of technology transfer or scale-up. The answers are derived directly from the experimental data and claims presented in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: How does the sulfonic acid resin method improve upon traditional concentrated sulfuric acid cyclization?
A: Traditional methods utilize large volumes of concentrated sulfuric acid as both solvent and catalyst, generating significant acidic waste and complicating product isolation. The resin-catalyzed method employs a heterogeneous solid acid that can be easily filtered off, drastically reducing waste treatment costs and simplifying the workup procedure.
Q: Can the sulfonic acid resin catalyst be recycled in this process?
A: Yes, one of the primary advantages of using macroporous strong acid polystyrene sulfonic acid resin or perfluorinated sulfonic acid resin is their potential for regeneration and reuse, which significantly lowers the long-term operational expenditure compared to homogeneous liquid acids.
Q: What purity levels can be achieved with this resin-catalyzed route?
A: Experimental data from the patent indicates that following the resin-catalyzed cyclization and subsequent recrystallization, mirtazapine with purity exceeding 98.5% to 99.0% can be consistently obtained, meeting stringent pharmacopoeia standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mirtazapine Supplier
The technological advancements described in patent CN111187264A highlight the continuous evolution of pharmaceutical manufacturing towards greener and more efficient processes. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of these developments, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex heterogeneous catalysis and multi-step syntheses with the utmost precision. We understand that consistency is key in the pharmaceutical industry; therefore, our rigorous QC labs enforce stringent purity specifications for all intermediates and APIs, ensuring that every batch of mirtazapine we produce meets or exceeds international pharmacopoeia standards.
We invite global partners to collaborate with us to optimize their supply chains using this advanced manufacturing technology. Whether you require custom synthesis of the key piperazine intermediate or full-scale production of the final API, our technical team is ready to assist. Please contact our Customized Cost-Saving Analysis department to discuss how we can tailor this resin-catalyzed process to your specific volume requirements. We encourage potential clients to reach out to our technical procurement team to request specific COA data and route feasibility assessments, ensuring a transparent and data-driven partnership.
