Advanced Manufacturing of Mirtazapine Intermediates via Optimized Hydrolysis and Cyclization
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex antidepressant agents, and Patent CN1680374A represents a significant technological leap in the manufacturing of mirtazapine. This pivotal intellectual property discloses a novel synthetic and crystallization method that fundamentally alters the production landscape for piperazine ring-containing compounds. Unlike traditional methodologies that rely on excessive reagent consumption and hazardous solvent systems, this invention introduces a streamlined approach characterized by optimized base hydrolysis ratios and improved purification protocols. The core innovation lies in the preparation of the critical intermediate, 1-(3-carboxypyridyl-2)-4-methyl-2-phenyl-piperazine, achieved through a controlled hydrolysis process that drastically reduces the molar equivalent of base required. 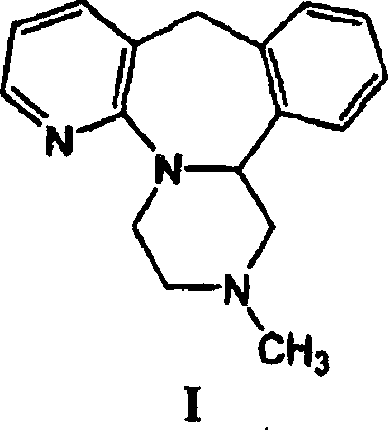 By refining these reaction parameters, the patent addresses long-standing inefficiencies in yield and waste management, positioning this technology as a cornerstone for modern, cost-effective API intermediate production.
By refining these reaction parameters, the patent addresses long-standing inefficiencies in yield and waste management, positioning this technology as a cornerstone for modern, cost-effective API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the standard industrial synthesis of mirtazapine was heavily reliant on the methods disclosed in U.S. Patent No. 4,062,848, which presented substantial operational and economic drawbacks for large-scale manufacturers. The conventional route necessitated extremely harsh reaction conditions, specifically requiring a massive excess of potassium hydroxide—up to 25 moles of base for every single mole of the nitrile substrate. This stoichiometric imbalance not only inflated raw material costs but also created a significant burden on waste treatment facilities due to the generation of vast quantities of alkaline effluent that required careful neutralization. Furthermore, the reaction kinetics were sluggish, often demanding prolonged heating periods of up to 24 hours to reach completion, thereby tying up reactor capacity and reducing overall plant throughput. 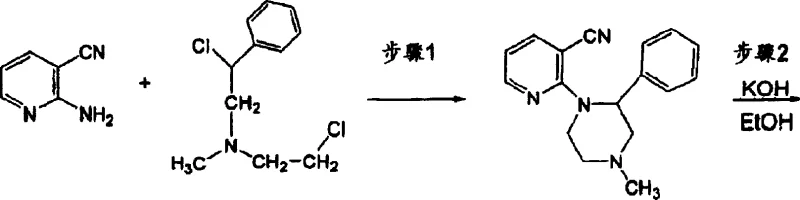 Compounding these issues, the final purification step relied on volatile and highly flammable solvents like ether and petroleum ether, posing severe safety risks and complicating the scale-up process for commercial facilities aiming for strict environmental and safety compliance.
Compounding these issues, the final purification step relied on volatile and highly flammable solvents like ether and petroleum ether, posing severe safety risks and complicating the scale-up process for commercial facilities aiming for strict environmental and safety compliance.
The Novel Approach
In stark contrast to the legacy processes, the methodology outlined in CN1680374A introduces a sophisticated optimization of the hydrolysis step that serves as a game-changer for production efficiency. The inventors discovered that the hydrolysis of 1-(3-cyanopyridyl-2)-4-methyl-2-phenyl-piperazine could be effectively conducted with a significantly reduced base ratio, capped at approximately 12 moles of base per mole of substrate, with preferred embodiments operating between 9 to 12 moles. This reduction is not merely a minor adjustment but a fundamental process intensification that allows for higher concentrations and faster reaction rates without compromising yield. Additionally, the patent proposes the use of more robust and manageable solvent systems for the final recrystallization, such as mixtures of ethanol and water or toluene and hexane, which eliminate the hazards associated with ether. 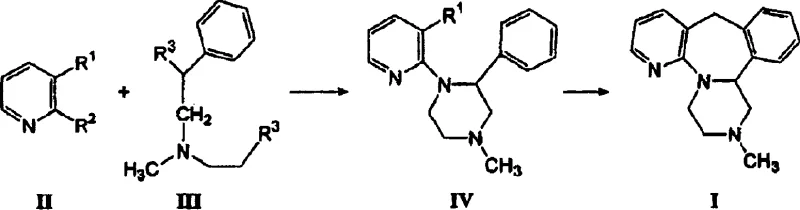 By integrating these improvements, the novel approach delivers a synthesis route that is inherently safer, faster, and more economically viable, directly addressing the pain points of high operational expenditure and environmental liability found in the prior art.
By integrating these improvements, the novel approach delivers a synthesis route that is inherently safer, faster, and more economically viable, directly addressing the pain points of high operational expenditure and environmental liability found in the prior art.
Mechanistic Insights into Optimized Base Hydrolysis and Cyclization
The chemical elegance of this patented process is rooted in the precise control of the hydrolysis mechanism, where the conversion of the nitrile group to the carboxylic acid is accelerated under high-temperature conditions with minimized base loading. The reaction typically proceeds at temperatures of at least 130°C, often reaching between 130°C and 150°C, frequently under elevated pressure (3-4 atm) to maintain the solvent in a liquid phase and enhance kinetic energy. Under these conditions, the nucleophilic attack of the hydroxide ion on the nitrile carbon is sufficiently rapid to drive the reaction to completion within a mere 2 to 8 hours, a fraction of the time required by older methods. The use of polar aprotic solvents like DMSO or DMF, or even alcoholic solvents like ethanol, facilitates the solubility of both the organic substrate and the inorganic base, creating a homogeneous or semi-homogeneous reaction medium that maximizes molecular collisions and reaction efficiency.
Following the hydrolysis and subsequent reduction to the hydroxymethyl intermediate, the final ring-closure step to form the tetracyclic mirtazapine structure is mediated by strong acid catalysis. The patent highlights the efficacy of sulfuric acid, polyphosphoric acid (PPA), or phosphoryl chloride as dehydrating agents to facilitate the intramolecular cyclization. This acid-catalyzed dehydration is critical for forming the seven-membered azepine ring fused to the pyridine and piperazine systems. The mechanism involves the protonation of the hydroxyl group, converting it into a good leaving group (water), followed by an electrophilic aromatic substitution or a related cyclization pathway where the electron-rich aromatic ring attacks the activated benzylic position. The choice of sulfuric acid is particularly advantageous due to its dual role as both a catalyst and a dehydrating agent, ensuring high conversion rates while minimizing the formation of side products that could complicate the impurity profile of the final API.
How to Synthesize Mirtazapine Efficiently
The practical implementation of this synthesis route requires careful attention to reaction parameters to maximize the benefits of the patented technology. The process begins with the hydrolysis of the nitrile precursor using the optimized base ratio, followed by isolation of the acid intermediate. This intermediate is then reduced, typically using lithium aluminum hydride or similar reducing agents, to generate the key hydroxymethyl-piperazine species. The final and most critical step involves the acid-mediated cyclization, where the linear precursor is folded into the complex tetracyclic structure of mirtazapine. For R&D teams looking to replicate or license this technology, understanding the specific solvent interactions and temperature gradients is essential for achieving the reported high yields and purity levels. The detailed standardized synthesis steps see the guide below.
- Hydrolyze 1-(3-cyanopyridyl-2)-4-methyl-2-phenyl-piperazine using potassium hydroxide (KOH) at a molar ratio of up to 12: 1 at temperatures exceeding 130°C.
- Isolate the intermediate 1-(3-carboxypyridyl-2)-4-methyl-2-phenyl-piperazine and reduce it to the hydroxymethyl derivative using LiAlH4 or similar reducing agents.
- Perform ring closure using sulfuric acid to form crude mirtazapine, followed by recrystallization using ethanol/water or toluene/hexane solvent systems.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN1680374A offers tangible strategic advantages that extend far beyond simple chemical yield improvements. The primary value proposition lies in the substantial reduction of raw material consumption, specifically the drastic cut in potassium hydroxide usage, which directly translates to lower input costs per kilogram of produced intermediate. Furthermore, the shift away from hazardous ether-based crystallization to safer alcohol-water or hydrocarbon systems significantly reduces the regulatory burden and insurance costs associated with handling volatile organic compounds. This process intensification allows manufacturing partners to achieve higher throughput with existing infrastructure, effectively increasing capacity without the need for capital-intensive expansion of reactor farms.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the optimized stoichiometry of the hydrolysis step. By reducing the potassium hydroxide requirement from 25 equivalents to roughly 12 equivalents, the process eliminates nearly half of the base-related raw material costs. More importantly, this reduction dramatically decreases the volume of alkaline waste generated, which in turn lowers the costs associated with waste neutralization, disposal, and environmental compliance reporting. The elimination of expensive and hazardous solvents like ether further contributes to cost savings by reducing solvent recovery expenses and minimizing loss due to evaporation.
- Enhanced Supply Chain Reliability: The robustness of the new solvent systems, such as ethanol, water, and toluene, ensures a more stable and reliable supply chain compared to the volatility of ether markets. These common industrial solvents are widely available from multiple global suppliers, reducing the risk of supply disruptions due to regional shortages or logistical bottlenecks. Additionally, the shortened reaction time—from 24 hours down to single-digit hours—allows for faster batch turnover, enabling manufacturers to respond more agilely to fluctuating market demands and reducing the lead time for delivering high-purity pharmaceutical intermediates to downstream API producers.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns perfectly with modern green chemistry principles by minimizing waste generation at the source. The reduced chemical load simplifies the effluent treatment process, making it easier for facilities to meet stringent environmental discharge regulations. The use of non-peroxide-forming solvents enhances operational safety during scale-up, allowing for the safe transition from pilot plant batches to multi-ton commercial production runs. This scalability ensures that the supply of mirtazapine intermediates can be reliably expanded to meet global pharmaceutical needs without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optimized synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of the technology for potential licensees and manufacturing partners. Understanding these nuances is crucial for making informed decisions about process adoption and supply chain integration.
Q: How does the new hydrolysis method reduce waste compared to prior art?
A: The novel process reduces the potassium hydroxide requirement from 25 moles per mole of nitrile (in US Patent 4,062,848) to a maximum of 12 moles. This drastic reduction minimizes the volume of alkaline waste requiring neutralization, significantly lowering environmental disposal costs and simplifying downstream processing.
Q: What are the safety advantages of the new crystallization solvents?
A: Unlike the conventional method which relies on highly flammable ether and petroleum ether, the improved process utilizes safer solvent systems such as ethanol/water or toluene/hexane. These solvents are easier to handle on a large industrial scale, reducing fire hazards and improving overall operational safety profiles.
Q: Can this synthesis route be scaled for commercial production?
A: Yes, the process is designed for scalability. By shortening reaction times from 24 hours to approximately 2-8 hours and utilizing robust solvent systems like toluene and ethanol, the method supports efficient commercial scale-up while maintaining high purity standards suitable for API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mirtazapine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing processes in the competitive landscape of antidepressant therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1680374A are fully realized in practical, industrial applications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of mirtazapine intermediate we produce adheres to the highest international quality standards required by regulatory bodies worldwide.
We invite pharmaceutical companies and procurement specialists to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can improve your bottom line. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable manufacturing strategy available in the market.
