Advanced Mirtazapine Manufacturing: Novel Cyclization and Crystallization Technologies for Commercial Scale
Introduction to Advanced Mirtazapine Synthesis Technologies
The pharmaceutical industry constantly seeks more efficient pathways for producing critical antidepressants like mirtazapine, a tetracyclic compound widely recognized for its efficacy in treating major depressive disorder. Patent CN1679586A introduces a significant technological leap in the synthesis of piperazine ring-containing compounds, specifically addressing the bottlenecks found in earlier manufacturing methods. This patent details novel approaches for preparing key intermediates such as 1-(3-carboxypyridyl-2)-4-methyl-2-phenyl-piperazine and the final active pharmaceutical ingredient through optimized hydrolysis and cyclization reactions. By fundamentally altering the stoichiometry of base-catalyzed reactions and introducing safer crystallization protocols, this technology offers a robust framework for modern API production. The implications for industrial chemistry are profound, as the shift away from excessive reagent usage directly correlates with reduced environmental impact and lower operational expenditures. For stakeholders in the fine chemical sector, understanding these mechanistic improvements is essential for evaluating supply chain resilience and production feasibility.
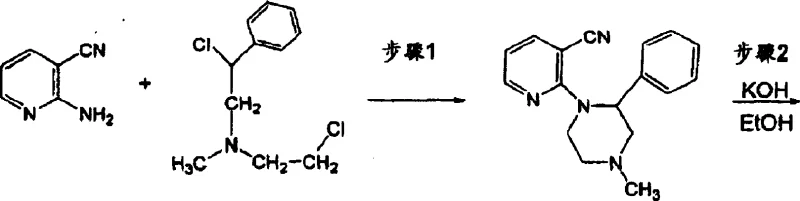
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of mirtazapine intermediates relied heavily on methods disclosed in older patents, such as U.S. Patent No. 4,062,848, which presented significant operational challenges for large-scale manufacturers. The conventional process for synthesizing the carboxylic acid intermediate involved the hydrolysis of a nitrile precursor under extremely harsh alkaline conditions. Specifically, the prior art required a massive excess of potassium hydroxide, utilizing up to 25 moles of base for every single mole of nitrile substrate. This extreme stoichiometric imbalance not only drove up raw material costs but also created severe downstream processing issues. The resulting reaction mixture was highly basic, necessitating complex and costly neutralization steps to handle the substantial volume of alkaline waste generated. Furthermore, these reactions were kinetically sluggish, often requiring heating for extended periods of up to 24 hours to reach completion, thereby tying up reactor capacity and reducing overall plant throughput. Additionally, the final purification of mirtazapine in these older methods relied on hazardous solvent systems like diethyl ether and petroleum ether, which pose significant safety risks regarding flammability and explosion potential in an industrial setting.
The Novel Approach
In stark contrast to the inefficient legacy processes, the methodology described in CN1679586A offers a streamlined and chemically elegant solution that drastically improves process metrics. The inventors discovered that the hydrolysis of the nitrile intermediate could be achieved effectively with a significantly reduced amount of base, lowering the molar ratio of potassium hydroxide to nitrile to about 12:1 or even less, preferably between 9:1 and 12:1. This reduction in reagent load simplifies the work-up procedure, minimizes waste generation, and shortens the reaction time to a mere 2 to 8 hours depending on temperature conditions. Alternatively, the patent discloses an entirely different synthetic route that bypasses the nitrile hydrolysis step altogether by reacting 2-amino-3-hydroxymethylpyridine directly with N-methyl-1-phenyl-2,2'-iminodiethyl chloride. This direct cyclization approach forms the piperazine intermediate in fewer steps, utilizing common solvents like dichloroethane and concluding with an acid-catalyzed ring closure using sulfuric acid. This dual-pathway innovation provides manufacturers with flexible options to optimize their specific production lines for either cost or speed, representing a substantial advancement over the rigid and wasteful protocols of the past.
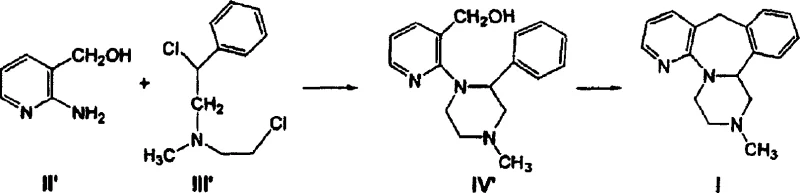
Mechanistic Insights into Base-Catalyzed Hydrolysis and Acid Cyclization
The core chemical innovation in this patent lies in the optimization of the hydrolysis mechanism and the introduction of a robust acid-catalyzed cyclization pathway. In the optimized hydrolysis route, the reaction proceeds by heating the nitrile intermediate with aqueous alkali in a polar solvent system, potentially including alcohols like ethanol or aprotic solvents like DMSO. The key mechanistic advantage here is the ability to drive the conversion of the cyano group to the carboxylate anion efficiently without the need for a vast excess of hydroxide ions, which traditionally served to push the equilibrium but at a high cost. By maintaining temperatures above 130°C, potentially under autogenous pressure of 3 to 4 atmospheres, the activation energy barrier is overcome more rapidly, allowing the reaction to proceed to completion in a fraction of the time required by prior art. This suggests a more favorable kinetic profile where the rate-determining step is accelerated by thermal energy rather than brute-force reagent concentration. On the alternative pathway, the mechanism involves a nucleophilic substitution where the amine group of the pyridine derivative attacks the chloro-ethyl groups of the imino-diethyl chloride, forming the piperazine ring. Subsequent treatment with strong dehydrating acids like concentrated sulfuric acid facilitates the intramolecular electrophilic aromatic substitution or dehydration necessary to close the seven-membered azepine ring, yielding the final tetracyclic mirtazapine structure with high fidelity.
Impurity control is another critical aspect addressed by these mechanistic refinements, particularly concerning the formation of side products during the harsh conditions of traditional synthesis. In the conventional 25:1 base method, the prolonged exposure to high concentrations of hot alkali increases the likelihood of degradation of the sensitive pyridine and piperazine moieties, leading to complex impurity profiles that are difficult to separate. The novel method's shorter reaction time and lower base concentration inherently limit the window for such degradation reactions to occur, resulting in a cleaner crude product profile. Furthermore, the acid-cyclization route allows for precise control over the ring-closing step; by selecting specific dehydrating agents like polyphosphoric acid or phosphorus oxychloride, manufacturers can tune the reaction environment to minimize polymerization or charring, which are common issues when using non-specific strong acids at high temperatures. The patent also highlights the importance of pH control during work-up, recommending adjustment to pH 6-7 to precipitate the intermediate cleanly, thereby avoiding the co-precipitation of inorganic salts or soluble organic impurities that might remain in highly acidic or basic solutions. This attention to physicochemical parameters ensures that the final API meets stringent purity specifications required for regulatory approval.
How to Synthesize Mirtazapine Efficiently
The synthesis of mirtazapine according to this patent involves precise control over reaction parameters to maximize yield and purity while minimizing hazards. The process generally begins with the formation of the piperazine core, either through the optimized hydrolysis of a nitrile precursor or the direct condensation of an amino-alcohol with a chloro-amine. Detailed operational procedures involve dissolving reactants in appropriate solvents such as dichloroethane or ethanol, followed by heating to reflux temperatures to drive the reaction to completion. The subsequent cyclization step requires careful addition of strong acids like sulfuric acid under controlled cooling to manage the exotherm, followed by a heating phase to effect ring closure. Finally, the crude product is subjected to a specialized recrystallization protocol using safe solvent mixtures to isolate the pure drug substance. For a comprehensive understanding of the specific temperatures, mixing rates, and isolation techniques required to replicate these results in a GMP environment, please refer to the standardized synthesis guide below.
- React 2-amino-3-hydroxymethylpyridine with N-methyl-1-phenyl-2,2'-iminodiethyl chloride in a solvent like dichloroethane to form the piperazine intermediate.
- Perform cyclization by adding the intermediate to concentrated sulfuric acid at controlled temperatures (e.g., 10-60°C) to close the azepine ring.
- Purify the crude mirtazapine via recrystallization using safe solvent systems such as ethanol and water mixtures instead of hazardous ethers.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the technologies described in CN1679586A offers transformative benefits for procurement strategies and supply chain management within the pharmaceutical sector. The most immediate impact is seen in the drastic reduction of raw material consumption, particularly regarding the base catalyst. By lowering the requirement for potassium hydroxide from 25 moles to roughly 12 moles per mole of substrate, manufacturers can achieve significant cost savings on bulk chemicals, which translates directly to a lower cost of goods sold (COGS). This efficiency gain is compounded by the reduction in waste disposal costs; handling and neutralizing large volumes of concentrated alkaline waste is a major expense in chemical manufacturing, and cutting this volume nearly in half represents a substantial operational improvement. Moreover, the shift away from hazardous solvents like diethyl ether towards safer alternatives such as ethanol, water, and toluene mitigates the risks associated with storage and handling of flammable materials. This not only lowers insurance and safety compliance costs but also enhances the overall reliability of the supply chain by reducing the likelihood of production stoppages due to safety incidents or regulatory scrutiny regarding volatile organic compound emissions.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the optimization of reagent stoichiometry and the simplification of downstream processing. Eliminating the need for a massive excess of base means that less money is spent on purchasing potassium hydroxide, and significantly less resources are allocated to the neutralization and disposal of the resulting salt waste. The shorter reaction times, dropping from 24 hours to as little as 2 to 8 hours, allow for increased batch turnover, meaning the same reactor infrastructure can produce more product over a given period, effectively amortizing fixed capital costs over a larger output volume. Additionally, the use of common, inexpensive solvents like ethanol and water for crystallization replaces costly and regulated solvents like ether, further driving down the variable costs associated with each kilogram of API produced. These cumulative efficiencies create a leaner manufacturing model that is highly competitive in the global generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the reliance on readily available, commodity-grade raw materials rather than specialized or hazardous reagents. Potassium hydroxide, ethanol, and sulfuric acid are produced globally in massive quantities, ensuring that supply disruptions are unlikely to impact production schedules. The robustness of the new crystallization method, which tolerates a wider range of solvent ratios and conditions compared to the finicky ether precipitation of the past, adds another layer of reliability. Manufacturers are less likely to face batch failures due to minor variations in solvent quality or environmental conditions, leading to more consistent delivery timelines for customers. This predictability is crucial for pharmaceutical companies managing tight inventory levels and just-in-time manufacturing schedules, as it reduces the need for excessive safety stock and buffer inventory.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is often fraught with challenges, particularly when dealing with exothermic reactions and hazardous solvents. The novel methods described herein are inherently more scalable because they operate under milder conditions and use solvents with higher flash points and lower toxicity profiles. The replacement of ether with ethanol/water systems eliminates the explosion hazards that often limit the size of batch reactors in older facilities, allowing for larger batch sizes and more efficient use of plant capacity. From an environmental standpoint, the reduction in alkaline waste and the use of greener solvents align with increasingly strict global environmental regulations, such as REACH in Europe and various EPA guidelines in the US. This proactive compliance reduces the risk of future regulatory fines and facilitates easier permitting for facility expansions, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel mirtazapine synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on how these methods differ from standard industry practices. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of suppliers utilizing this IP. The focus remains on practical application, safety improvements, and the tangible benefits observed in pilot and production-scale examples provided within the intellectual property.
Q: How does the new hydrolysis method reduce waste compared to prior art?
A: The novel method reduces the molar ratio of potassium hydroxide from 25:1 to approximately 12:1 or lower, significantly decreasing the volume of alkaline waste requiring neutralization and disposal.
Q: What solvents are recommended for safe mirtazapine crystallization?
A: The patent recommends replacing hazardous ether and petroleum ether with safer, scalable solvent systems like ethanol/water mixtures, toluene, or hexane, which improve operational safety and yield.
Q: Can this process be scaled for industrial production?
A: Yes, the use of common solvents like ethanol and the reduction in extreme reaction conditions (lower base load, shorter times) make the process highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mirtazapine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes like those described in CN1679586A requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of reduced base usage and safer solvents are fully realized in actual manufacturing runs. Our facilities are equipped with state-of-the-art rigorous QC labs capable of monitoring the complex impurity profiles associated with piperazine and azepine chemistry, guaranteeing that every batch meets stringent purity specifications required by global regulatory bodies. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to process validation ensures that the cost-saving and efficiency gains promised by this patent are delivered reliably to our partners.
We invite pharmaceutical companies and procurement leaders to engage with us to explore how these optimized synthesis routes can enhance your supply chain security and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data from our recent batches and to discuss detailed route feasibility assessments for your upcoming projects. By leveraging our expertise in mirtazapine intermediate synthesis, you can secure a stable, high-quality supply of this critical antidepressant while optimizing your operational budget.
