Industrial Scale-Up of Pemetrexed Disodium Using Advanced Molecular Sieve Catalysis
Industrial Scale-Up of Pemetrexed Disodium Using Advanced Molecular Sieve Catalysis
The pharmaceutical industry continuously seeks robust manufacturing processes that balance high yield with environmental sustainability, particularly for critical oncology treatments like Pemetrexed Disodium. Patent CN112174970B introduces a transformative approach to this challenge by replacing traditional homogeneous base catalysts with an alkali-modified microporous-mesoporous molecular sieve. This innovation addresses the persistent issue of yield attenuation during scale-up, a common bottleneck in the commercial production of complex pyrimidine analogs. By leveraging the unique structural properties of modified molecular sieves, manufacturers can achieve consistent reaction kinetics and superior impurity profiles, ensuring a reliable supply of high-purity API intermediates for global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pemetrexed Disodium has relied heavily on aqueous sodium hydroxide solutions for key hydrolysis and saponification steps. While effective on a laboratory scale, this homogeneous catalytic system presents severe drawbacks when transitioned to industrial manufacturing. Data from comparative examples in the patent literature indicates that scaling up NaOH-catalyzed reactions often results in a precipitous drop in yield, falling from approximately 76% in small-scale trials to roughly 64% in larger reactors. Furthermore, the reliance on strong alkaline conditions generates substantial volumes of hazardous wastewater, imposing heavy burdens on effluent treatment facilities and increasing operational costs. The traditional purification protocols also frequently necessitate the use of toxic solvents like dimethyl sulfoxide (DMSO) and thermal crystallization, which can accelerate the oxidation of the sensitive pemetrexed molecule, thereby compromising final product quality and stability.
The Novel Approach
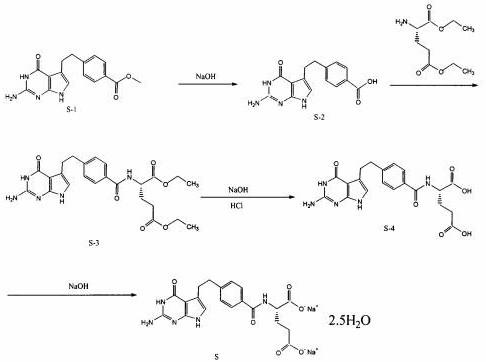
In stark contrast, the novel methodology disclosed in CN112174970B utilizes a specifically engineered alkali-modified microporous-mesoporous molecular sieve as a heterogeneous catalyst. This advanced material facilitates the hydrolysis of methyl 4-[2-(2-amino-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoate (S-1) and subsequent saponification steps with remarkable efficiency. The implementation of this solid catalyst not only stabilizes the reaction yield across different production scales but also fundamentally alters the downstream processing requirements. By eliminating the need for liquid NaOH, the process drastically reduces alkaline waste generation. Moreover, the improved selectivity allows for the substitution of hazardous solvents with safer ethanol-water mixtures for recrystallization, performed at ambient temperatures to preserve chemical integrity.
Mechanistic Insights into Alkali-Modified Molecular Sieve Catalysis
The efficacy of this synthesis route lies in the precise engineering of the catalyst's pore structure and surface chemistry. The molecular sieve is synthesized using a soft template method involving a quaternary ammonium salt template agent, a silicon source such as sodium silicate, and an aluminum source. The subsequent alkali modification creates a hierarchical pore system containing both micropores and mesopores. This dual-pore architecture is critical for accommodating the relatively bulky Pemetrexed intermediates, allowing them to diffuse freely to the active basic sites within the catalyst framework. Unlike traditional zeolites which may suffer from diffusion limitations, this microporous-mesoporous composite ensures that the reactant molecules have unhindered access to the catalytic centers, thereby maintaining high turnover frequencies even in viscous reaction media typical of large-scale batches.
From an impurity control perspective, the heterogeneous nature of the catalyst plays a pivotal role in minimizing side reactions. In homogeneous NaOH systems, the high concentration of free hydroxide ions can promote non-selective hydrolysis or degradation of sensitive functional groups, leading to complex impurity profiles that are difficult to separate. The confined basic sites within the molecular sieve channels provide a more controlled microenvironment, favoring the desired hydrolysis of the ester groups while suppressing unwanted decomposition pathways. This selectivity is further enhanced by the ability to easily separate the solid catalyst from the reaction mixture via filtration, preventing catalyst residues from contaminating the product stream and simplifying the purification workflow significantly.
How to Synthesize Pemetrexed Disodium Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from bench-scale optimization to commercial manufacturing. The process begins with the fixation of the prepared alkali-modified molecular sieve catalyst in a reaction vessel, followed by the addition of the starting ester intermediate. The reaction proceeds under mild heating with continuous stirring until TLC analysis confirms the disappearance of the starting material. Subsequent steps involve standard workup procedures such as pH adjustment and filtration, followed by condensation with L-glutamic acid derivatives. The final saponification and salt formation steps also utilize the molecular sieve catalyst, ensuring consistency throughout the synthetic sequence. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Hydrolyze methyl 4-[2-(2-amino-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoate (S-1) using the molecular sieve catalyst to generate acid (S-2).
- Condense intermediate S-2 with diethyl L-glutamate to obtain the diethyl ester intermediate (S-3).
- Perform degradation and saponification of S-3 using the molecular sieve catalyst to obtain crude Pemetrexed Disodium (S-4), followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this molecular sieve-catalyzed process offers compelling economic and logistical advantages over legacy methods. The primary value driver is the stabilization of production yields at scale, which directly translates to more predictable output volumes and reduced raw material waste. By mitigating the yield drop typically associated with scaling up NaOH reactions, manufacturers can optimize their material balance and reduce the cost of goods sold. Furthermore, the elimination of strong alkaline wastewater significantly lowers the environmental compliance costs associated with effluent treatment, contributing to substantial overall cost reduction in API manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The shift to a heterogeneous catalyst system eliminates the recurring cost of purchasing and handling large volumes of concentrated sodium hydroxide solutions. More importantly, the simplified purification process, which avoids the use of expensive and toxic solvents like DMSO in favor of ethanol and water, leads to significant savings in solvent procurement and recovery costs. The reduction in waste treatment complexity further drives down operational expenditures, making the overall production economics highly favorable for long-term commercial supply.
- Enhanced Supply Chain Reliability: Consistency is the cornerstone of a reliable pharmaceutical supply chain. The robustness of the molecular sieve catalyst ensures that batch-to-batch variability is minimized, even as production volumes increase to meet market demand. This reliability reduces the risk of production delays caused by failed batches or extensive reprocessing, ensuring a steady flow of high-purity Pemetrexed Disodium to downstream formulation partners. The use of readily available and less hazardous raw materials also mitigates supply risks associated with regulated chemical precursors.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with the catalyst demonstrating stable performance from gram-scale experiments to multi-kilogram production runs. The environmental footprint of the manufacturing process is drastically reduced by avoiding the discharge of alkaline wastewater and minimizing the use of volatile organic compounds. This alignment with green chemistry principles not only facilitates regulatory approval in stringent markets but also future-proofs the supply chain against tightening environmental regulations and carbon emission targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific procurement needs. Understanding these nuances is essential for making informed decisions about supplier qualification and process validation.
Q: How does the molecular sieve catalyst improve yield compared to traditional NaOH methods?
A: Traditional NaOH methods suffer from significant yield reduction upon scale-up (dropping from ~76% to ~64% in comparative examples). The alkali-modified molecular sieve maintains high yields (over 80% in initial steps) even at industrial scales by providing stable active sites without the side reactions associated with homogeneous strong bases.
Q: What are the environmental benefits of this new synthesis route?
A: The process eliminates the use of aqueous sodium hydroxide solutions, thereby avoiding the discharge of strong alkaline wastewater. Additionally, it replaces toxic solvents like dimethyl sulfoxide (DMSO) with safer ethanol/water mixtures for purification.
Q: Does this method affect the purity of the final Pemetrexed Disodium product?
A: Yes, it enhances purity control. The heterogeneous catalyst reduces the generation of organic impurities, and the use of ethanol/water recrystallization at normal temperatures prevents thermal oxidation, achieving HPLC purity levels exceeding 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Disodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to ensure the highest standards of quality and efficiency. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the molecular sieve-catalyzed route for Pemetrexed Disodium are seamlessly translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting requirements of global regulatory bodies, providing our partners with absolute confidence in the integrity of their supply chain.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic potential of this technology. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-value chemical solutions tailored to your strategic goals.
