Scalable Industrial Synthesis of Pemetrexed Disodium via Alkali-Modified Mesoporous Catalysts
Scalable Industrial Synthesis of Pemetrexed Disodium via Alkali-Modified Mesoporous Catalysts
The pharmaceutical industry is constantly seeking robust manufacturing processes that balance high yield with environmental sustainability, particularly for complex oncology agents like Pemetrexed Disodium. Patent CN112442038B introduces a transformative industrial preparation method that fundamentally alters the catalytic landscape of this synthesis. By replacing traditional homogeneous sodium hydroxide catalysts with a specialized alkali-modified mesoporous molecular sieve, this technology addresses critical bottlenecks in scale-up efficiency and waste management. This innovation is not merely a procedural tweak but a strategic overhaul that ensures consistent product quality while adhering to green chemistry principles. For global supply chains, this represents a pivotal shift towards more reliable and compliant manufacturing of high-value pharmaceutical intermediates.
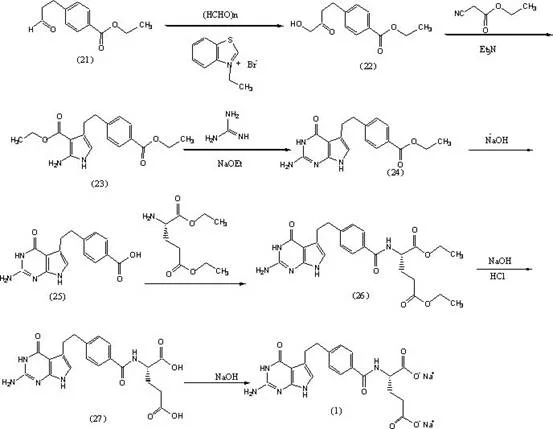
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Pemetrexed Disodium has relied heavily on strong alkaline conditions, typically utilizing aqueous sodium hydroxide solutions for key hydrolysis steps. While effective on a small laboratory scale, these conventional methods exhibit severe drawbacks when transferred to industrial production environments. The primary issue is the instability of yield upon scale-up; data indicates that reaction efficiency can plummet significantly when moving from gram-scale to kilogram-scale batches due to poor heat dissipation and mixing inefficiencies inherent in liquid-liquid systems. Furthermore, the reliance on NaOH generates substantial volumes of alkaline wastewater, creating a heavy environmental burden and increasing operational costs associated with neutralization and disposal. Additionally, traditional purification often requires toxic organic solvents like dimethyl sulfoxide or acetone, which pose safety risks and complicate solvent residue control in the final Active Pharmaceutical Ingredient.
The Novel Approach
The novel approach detailed in the patent leverages the unique properties of alkali-modified mesoporous molecular sieves to overcome these historical limitations. Unlike traditional liquid bases, this heterogeneous catalyst offers a structured surface with tunable pore sizes that facilitate better mass transfer and active site accessibility. This structural advantage allows the reaction to proceed with high selectivity and conversion rates even under the rigorous conditions of industrial reactors. By eliminating the need for bulk sodium hydroxide, the process inherently reduces the generation of saline wastewater, simplifying the downstream environmental compliance workflow. Moreover, the stability of the solid catalyst ensures that the reaction kinetics remain consistent regardless of batch size, effectively solving the yield attenuation problem that plagues conventional scale-up efforts. This method also enables the use of greener solvent systems, such as ethanol and water mixtures, for crystallization, further enhancing the safety profile of the manufacturing process.
Mechanistic Insights into Alkali-Modified Mesoporous Catalysis
The core of this technological breakthrough lies in the specific interaction between the reactant molecules and the engineered surface of the alkali-modified mesoporous molecular sieve. These catalysts are synthesized using soft template methods, creating a framework with uniform mesoporous channels that are chemically modified with organic base groups. This architecture is crucial because it provides a high density of basic active sites that are sterically accessible to the relatively bulky Pemetrexed precursors. During the hydrolysis of the methyl ester intermediate (S-1) to the free acid (S-2), the catalyst functions by activating the carbonyl group through surface basic sites, facilitating nucleophilic attack by water molecules without the need for excessive bulk alkalinity. This localized catalysis minimizes the exposure of the sensitive pyrrolo-pyrimidine core to harsh conditions that could lead to degradation or polymerization.
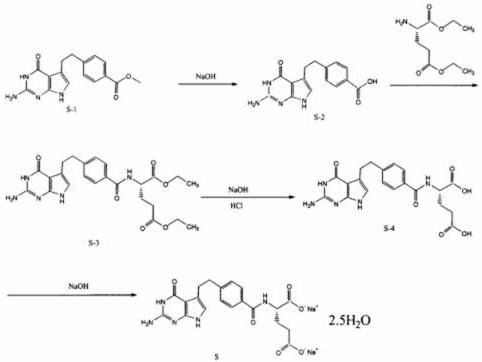
Furthermore, the impurity control mechanism is intrinsically linked to the shape-selective nature of the mesoporous structure. The defined pore dimensions act as a molecular sieve, potentially restricting the formation or diffusion of larger, unwanted by-product molecules that might arise from non-specific base-catalyzed side reactions. In conventional NaOH systems, the high concentration of hydroxide ions in the bulk solution can promote indiscriminate hydrolysis or epimerization, leading to complex impurity profiles that are difficult to separate. In contrast, the confined environment of the molecular sieve channels enforces a degree of regioselectivity, ensuring that hydrolysis occurs primarily at the intended ester linkages. This results in a cleaner reaction crude, which significantly reduces the load on subsequent purification steps and contributes to the high final purity observed in the finished product, meeting stringent pharmacopeial standards for oncology drugs.
How to Synthesize Pemetrexed Disodium Efficiently
The synthesis protocol outlined in the patent provides a clear, four-stage pathway designed for industrial reproducibility. The process begins with the catalytic hydrolysis of the methyl ester precursor, followed by amide coupling with L-glutamic acid derivatives, a second hydrolysis step to reveal the dicarboxylic acid functionality, and finally, salt formation. Each stage is optimized to utilize the recyclable nature of the molecular sieve catalyst, maximizing atom economy. The detailed standardized synthesis steps, including specific temperature controls, molar ratios, and workup procedures required to replicate this high-efficiency route, are provided in the technical guide below.
- Hydrolyze methyl 4-[2-(2-amino-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoate (S-1) using the alkali-modified catalyst to generate the free acid (S-2).
- Condense the resulting acid (S-2) with L-diethyl glutamate using coupling agents to form the diethyl ester intermediate (S-3).
- Perform a second hydrolysis on intermediate (S-3) using the same molecular sieve catalyst to remove ethyl groups and obtain the crude acid (S-4).
- Convert the crude acid (S-4) into the final disodium salt through saponification and recrystallization in an ethanol/water system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology translates directly into enhanced operational resilience and cost predictability. The shift from a consumable liquid base to a durable solid catalyst fundamentally changes the cost structure of the manufacturing process. By mitigating the risks associated with yield loss during scale-up, manufacturers can guarantee more consistent output volumes, reducing the need for safety stock and minimizing the risk of supply shortages. The elimination of hazardous solvents and the reduction of wastewater treatment requirements also lower the overall cost of goods sold, allowing for more competitive pricing strategies in the global market. This process stability is a critical factor for long-term supply agreements, ensuring that partners can rely on uninterrupted delivery schedules.
- Cost Reduction in Manufacturing: The replacement of sodium hydroxide with a reusable molecular sieve catalyst eliminates the recurring cost of purchasing bulk chemicals for every batch. More importantly, the drastic reduction in alkaline wastewater generation removes the significant expense associated with neutralization and specialized waste disposal services. The ability to use simpler solvent systems like ethanol and water for purification further reduces solvent recovery costs compared to complex mixtures involving dimethyl sulfoxide. These cumulative savings create a leaner manufacturing model that protects margins against fluctuating raw material prices.
- Enhanced Supply Chain Reliability: One of the most significant advantages is the stabilization of yield at the industrial scale. Conventional methods often suffer from unpredictable yield drops when moving from pilot to production, leading to planning uncertainties. This new method demonstrates consistent performance regardless of batch size, ensuring that production targets are met reliably. The robustness of the solid catalyst also means fewer process interruptions due to reagent quality variations, providing a more stable and predictable supply of high-purity pharmaceutical intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: As regulatory pressures on pharmaceutical manufacturing intensify, the environmental footprint of a synthesis route becomes a key qualification criterion. This process inherently aligns with green chemistry initiatives by reducing hazardous waste and avoiding toxic solvents. The scalability is proven by the maintenance of high yields in large-scale reactors, demonstrating that the technology is ready for immediate commercial deployment without the need for extensive re-optimization. This compliance readiness accelerates time-to-market for generic versions and ensures long-term viability of the supply chain against evolving environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkali-modified molecular sieve technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering clarity on performance metrics and operational benefits. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing facilities.
Q: How does the alkali-modified molecular sieve catalyst improve yield stability compared to NaOH?
A: Traditional sodium hydroxide catalysis often suffers from significant yield reduction when scaling from laboratory to industrial reactors due to mixing and heat transfer limitations. The alkali-modified mesoporous molecular sieve provides a heterogeneous catalytic surface with optimized pore structures that maintain consistent activity and selectivity even at large scales, preventing the yield drop typically seen in homogeneous base catalysis.
Q: What are the environmental benefits of this synthesis route?
A: By replacing aqueous sodium hydroxide solutions with a solid molecular sieve catalyst, the process eliminates the generation of large volumes of highly alkaline wastewater. This significantly reduces the burden on waste treatment facilities and aligns with stricter environmental regulations regarding effluent discharge in pharmaceutical manufacturing.
Q: Does this method affect the final purity of Pemetrexed Disodium?
A: Yes, the method enhances purity control. The specific pore structure of the catalyst minimizes side reactions and by-product formation. Furthermore, the process allows for effective purification using ethanol/water mixed solvents at normal temperatures, avoiding thermal degradation and achieving high-purity specifications suitable for oncology applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Disodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced catalytic technologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the alkali-modified molecular sieve method are translated into reality with precision. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that validate every batch against the highest international standards. Our infrastructure is designed to handle complex synthetic routes, guaranteeing that the benefits of this novel process are fully realized in the final product delivered to our clients.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your project's success and ensure a steady supply of high-quality Pemetrexed Disodium.
