Advanced Purification Technology for High-Purity Pemetrexed Disodium: Scaling Commercial Production
Advanced Purification Technology for High-Purity Pemetrexed Disodium: Scaling Commercial Production
The pharmaceutical industry constantly faces the challenge of producing active pharmaceutical ingredients (APIs) that meet stringent regulatory standards for purity and impurity profiles. Patent CN102206218B introduces a groundbreaking method for purifying pemetrexed disodium, a critical antifolate agent used in the treatment of malignant pleural mesothelioma and non-small cell lung cancer. This technology addresses the persistent issue of organic and inorganic impurities that often plague conventional synthesis routes, offering a robust solution for reliable API intermediate supplier networks. By combining a salting-out technique with an organic solvent-water mixed solvent crystallization method, this process effectively controls impurity levels without the need for harsh heating conditions. The innovation lies in its ability to reduce the number of organic impurities significantly while ensuring the maximum single impurity content falls below the strict 0.1% identification threshold mandated by drug administration authorities. For R&D directors and procurement managers, this represents a pivotal shift towards more efficient and compliant manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification strategies for pemetrexed disodium have historically relied on either mixed solvent crystallization or simple salting-out techniques, both of which possess inherent drawbacks that compromise final product quality. Mixed solvent crystallization methods often require heating the aqueous solution to temperatures between 40°C and 70°C followed by cooling, a thermal cycle that unfortunately accelerates the hydrolysis and oxidation of the sensitive pemetrexed molecule. This thermal stress leads to the formation of degradation products, increasing both the variety and content of organic impurities beyond acceptable limits for bulk drug registration. Furthermore, standard salting-out processes, while effective at removing water-soluble inorganic salts, struggle to eliminate organic impurities with lower polarity than the target compound. Consequently, crude products purified by these singular methods frequently fail to meet the rigorous comparative quality standards set by original research and development enterprises, necessitating costly and time-consuming reprocessing steps.
The Novel Approach
The novel approach detailed in the patent overcomes these deficiencies by integrating two complementary purification mechanisms into a seamless, room-temperature workflow. By first employing a salting-out step using sodium chloride, the process efficiently precipitates the product while leaving highly water-soluble impurities in the mother liquor. This is immediately followed by a recrystallization step using a water-miscible organic solvent, such as ethanol, which targets the removal of less polar organic contaminants that the salting-out step missed. Crucially, this entire sequence is conducted at normal temperatures (5°C to 39°C), completely eliminating the thermal degradation pathways associated with traditional heating methods. This dual-action strategy ensures that the final purified pemetrexed disodium not only meets but often exceeds the purity benchmarks of imported reference preparations, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Salting-Out and Mixed Solvent Crystallization
The core of this purification technology relies on the precise manipulation of solubility parameters through ionic strength adjustment and solvent polarity modulation. In the first stage, the addition of sodium chloride increases the ionic strength of the aqueous phase, effectively "salting out" the pemetrexed disodium due to its reduced solubility in high-salt environments. This step is meticulously controlled by maintaining the pH between 7.5 and 8.5 and the sodium chloride concentration between 8% and 20%, conditions that optimize the selective precipitation of the target molecule while keeping soluble impurities in solution. The subsequent filtration and washing with a matching salt solution further strip away surface-adhered inorganic contaminants. The second stage involves redissolving the intermediate cake and introducing an organic solvent like ethanol to reach a concentration of 70% to 90%. This drastic change in solvent polarity forces the pemetrexed disodium to crystallize again, but this time, the lattice formation excludes organic impurities that have different solubility profiles in the alcohol-water mixture.
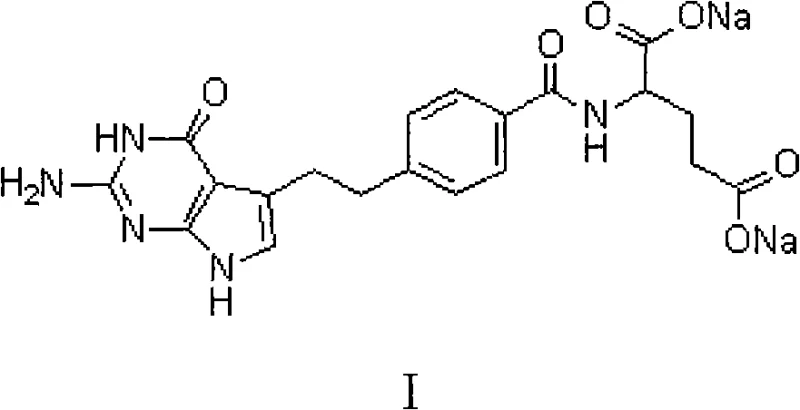
Impurity control is further enhanced by the strict avoidance of heating, which preserves the chemical integrity of the pyrrolo-pyrimidine core structure shown in the molecular diagram. The patent data demonstrates that this method can reduce the total number of organic impurities from as many as 15 distinct peaks in the crude material down to just 4 in the purified product. Moreover, the maximum single impurity content is driven down from levels exceeding 0.18% to below 0.07%, comfortably satisfying the identification threshold requirements. This level of control is achieved without introducing new contaminants; the chloride test confirms that residual sodium chloride from the salting-out step is effectively removed during the second crystallization and washing phases. For technical teams, understanding this mechanism is vital for scaling the commercial scale-up of complex pharmaceutical intermediates, as it provides a clear roadmap for maintaining batch-to-batch consistency.
How to Synthesize Pemetrexed Disodium Efficiently
Implementing this purification protocol requires careful attention to solvent ratios and crystallization times to maximize yield and purity. The process begins with dissolving the crude pemetrexed disodium in water, followed by the controlled addition of sodium chloride solution to induce the first crystallization. After filtering and washing, the intermediate solid is redissolved and treated with ethanol to trigger the final purification crystallization. This standardized approach minimizes variability and ensures that the stringent purity specifications are met consistently. The detailed operational parameters, including specific concentrations and stirring times, are critical for reproducing the high-quality results observed in the patent embodiments.
- Dissolve the pemetrexed disodium crude product in water, adjust pH to 7.5-8.5, and add sodium chloride solution to reach 8-20% concentration for initial crystallization.
- Filter the resulting crystals and wash the filter cake with 8-20% sodium chloride solution to remove water-soluble impurities effectively.
- Redissolve the filter cake in water, add a water-miscible organic solvent (like ethanol) to reach 70-90% concentration, and allow to crystallize at room temperature before final drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology offers substantial strategic benefits beyond mere technical compliance. The elimination of heating steps translates directly into significant energy savings and reduced operational complexity, as there is no need for specialized heating and cooling infrastructure or the extended cycle times associated with thermal cycling. Furthermore, the solvents and reagents used—primarily water, sodium chloride, and ethanol—are commodity chemicals that are readily available globally, mitigating the risk of supply chain disruptions often associated with exotic or hazardous reagents. This accessibility ensures a stable supply of raw materials, reducing lead time for high-purity pharmaceutical intermediates and allowing for more predictable production scheduling. The simplicity of the process also facilitates easier technology transfer between manufacturing sites, enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the simplification of unit operations and the use of inexpensive, non-hazardous reagents. By avoiding the need for expensive chromatography columns or complex extraction systems often required to meet impurity thresholds, the capital expenditure for purification equipment is drastically lowered. Additionally, the room-temperature operation reduces utility costs associated with steam generation and chilled water circulation, contributing to a leaner manufacturing cost structure. The high recovery rates observed in the patent embodiments suggest that material loss is minimized, further improving the overall economic efficiency of the production line.
- Enhanced Supply Chain Reliability: Relying on common industrial solvents like ethanol and standard salts like sodium chloride ensures that the manufacturing process is not vulnerable to the volatility of the specialty chemical market. This stability allows for long-term contracting and inventory planning, securing the continuity of supply for critical oncology medications. The robustness of the method against variations in crude input quality (effective for crude content ranging from 80% to 99.5%) means that suppliers can source raw materials from a broader range of vendors without compromising the final product quality, thereby diversifying the supply base and reducing dependency on single sources.
- Scalability and Environmental Compliance: The absence of toxic organic solvents and heavy metal catalysts simplifies waste treatment protocols, aligning the process with increasingly strict environmental regulations. Ethanol and saline waste streams are far easier to treat and dispose of compared to halogenated solvents or heavy metal residues, reducing the environmental footprint and associated compliance costs. The straightforward nature of the crystallization steps makes the process highly scalable from pilot plant to full commercial production, allowing manufacturers to respond quickly to market demand surges without extensive process re-validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating the feasibility of this method for their own production lines. Understanding these nuances is essential for making informed decisions about process adoption and vendor qualification.
Q: Why is the combination of salting-out and mixed solvent crystallization superior to single methods?
A: Single methods often fail to remove all impurity types; salting-out targets water-soluble inorganic salts, while mixed solvent crystallization targets less polar organic impurities. Combining them ensures comprehensive impurity removal below the 0.1% identification threshold required for drug registration.
Q: What is the critical temperature range for this purification process?
A: The process is designed to operate at normal temperatures, specifically between 5°C and 39°C. Avoiding high-temperature heating steps is crucial to prevent hydrolysis and oxidation of the pemetrexed disodium molecule, which would otherwise generate new degradation impurities.
Q: Which organic solvents are suitable for the second crystallization step?
A: While ethanol is the preferred solvent due to cost and efficacy balance, the method is also compatible with acetonitrile and dimethylformamide. The key requirement is that the solvent must be miscible with water to facilitate the controlled precipitation of the high-purity product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Disodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality intermediates that adhere to the most rigorous global standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the required impurity profiles. Our capability to implement sophisticated purification technologies, such as the salting-out and crystallization method described, positions us as a strategic partner for pharmaceutical companies seeking to optimize their supply chains.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall manufacturing expenses. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate our quality and technical competence firsthand. Let us collaborate to bring life-saving medications to patients faster and more efficiently.
