Advanced Hydrotalcite-Catalyzed Synthesis of Iodoaniline Derivatives for Commercial Scale Manufacturing
Advanced Hydrotalcite-Catalyzed Synthesis of Iodoaniline Derivatives for Commercial Scale Manufacturing
The pharmaceutical and agrochemical industries rely heavily on halogenated aromatic amines as critical building blocks for complex active pharmaceutical ingredients (APIs) and functional materials. Patent CN103467306A introduces a groundbreaking synthetic methodology for producing iodoaniline derivatives that addresses longstanding challenges regarding safety, efficiency, and environmental impact in electrophilic aromatic substitution. This innovation utilizes a specialized zinc-aluminum hydrotalcite intercalated with iodate radicals, functioning as a controlled-release catalyst system within an acidic buffer environment. By shifting away from direct elemental iodine usage or hazardous strong oxidants, this technology offers a robust pathway for generating high-purity intermediates essential for drugs like reserpine and indomethacin. For global supply chain leaders, this patent represents a significant opportunity to optimize manufacturing protocols while adhering to stricter environmental regulations. The following analysis details the mechanistic advantages and commercial viability of adopting this hydrotalcite-mediated iodination strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of iodoanilines has been plagued by significant operational hazards and inefficiencies inherent to traditional electrophilic substitution protocols. Conventional processes typically involve the direct reaction of aniline with elemental iodine in the presence of bases like sodium bicarbonate, a method fraught with the physical challenge of iodine sublimation which leads to substantial raw material loss and equipment corrosion. Furthermore, alternative approaches utilizing strong oxidizing agents such as periodic acid or mixtures of nitric and sulfuric acid introduce severe safety risks, including potential explosions and the generation of toxic waste streams that complicate regulatory compliance. The weak electrophilicity of molecular iodine often necessitates harsh reaction conditions or excessive reagent loading to drive the reaction to completion, resulting in poor atom economy and difficult purification steps. These legacy methods also struggle with selectivity issues, often producing complex mixtures of poly-iodinated byproducts that require energy-intensive separation techniques to achieve pharmaceutical-grade purity. Consequently, procurement managers face inflated costs due to low yields and the expensive disposal of hazardous chemical waste associated with these outdated synthetic routes.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by employing iodate-intercalated zinc-aluminum hydrotalcite as a sophisticated, heterogeneous catalyst system that fundamentally alters the reaction kinetics. Instead of relying on volatile elemental iodine, this approach generates active iodine species in situ through the controlled interaction between slowly released iodate ions from the hydrotalcite layers and added alkali metal iodides in a buffered solution. This mechanism effectively eliminates the sublimation losses associated with solid iodine handling while simultaneously lowering the activation energy required for the electrophilic attack on the aniline ring. The use of a mild acidic buffer, specifically within the pH range of 3.7 to 5.2, provides an optimal environment that stabilizes the reactive intermediates without degrading the sensitive amine functionality of the substrate. Operational simplicity is another hallmark of this novel approach, as the reaction proceeds efficiently at moderate temperatures between 35°C and 75°C, drastically reducing energy consumption compared to high-temperature conventional processes. Ultimately, this technology delivers equivalent or superior yields with a significantly reduced environmental footprint, making it an ideal candidate for modern green chemistry initiatives in fine chemical manufacturing.
Mechanistic Insights into Hydrotalcite-Mediated Electrophilic Iodination
The core innovation of this synthesis lies in the unique structural properties of the zinc-aluminum hydrotalcite, which acts not merely as a support but as a dynamic reservoir for iodate anions within its interlayer galleries. During the preparation phase, iodate radicals are successfully intercalated into the hydrotalcite structure via a co-precipitation method under strict nitrogen protection to prevent carbonate contamination, ensuring a high loading capacity of the active iodine source. When introduced into the acidic buffer solution during the reaction phase, the hydrotalcite lattice undergoes a controlled dissolution or ion-exchange process that releases iodate ions ($IO_3^-$) at a steady, manageable rate rather than all at once. These released iodate ions immediately react with the excess iodide ions ($I^-$) present in the solution from the added alkali metal iodide salt to generate highly reactive electrophilic iodine species, likely in the form of molecular iodine or cationic iodine complexes, directly at the reaction site. This in situ generation ensures that the concentration of active iodine remains optimal for substitution without accumulating to levels that would promote side reactions or pose safety hazards. The layered double hydroxide structure thus serves a dual purpose: it protects the iodate from premature decomposition and facilitates a homogeneous-like reaction environment despite being a heterogeneous solid, thereby enhancing the overall mass transfer efficiency.
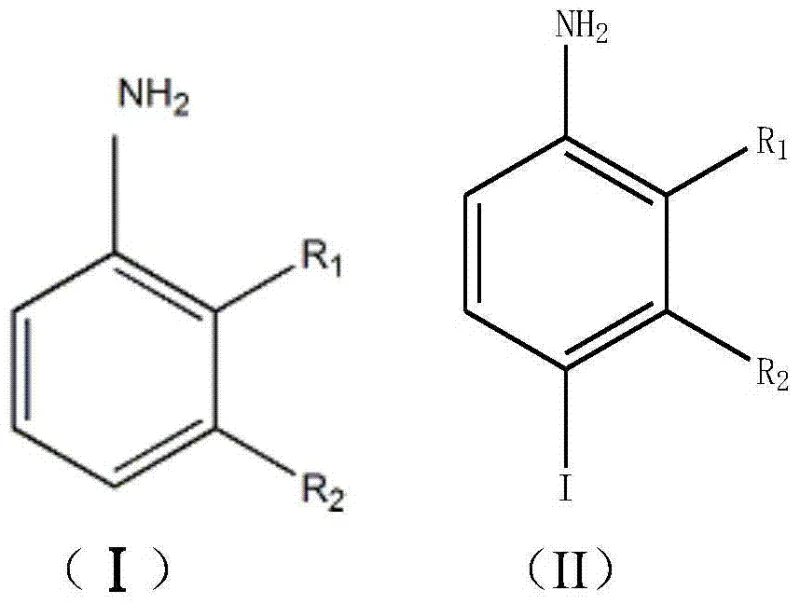
From an impurity control perspective, this mechanistic pathway offers distinct advantages over traditional oxidation methods by minimizing the formation of poly-iodinated byproducts and oxidative degradation products. The buffered pH environment plays a critical role in suppressing the protonation of the aniline amino group, which would otherwise deactivate the ring towards electrophilic attack, while simultaneously preventing the formation of diazonium salts that can occur under strongly acidic conditions. The slow-release nature of the catalyst ensures that the local concentration of the electrophile never exceeds the kinetic capacity of the substrate to react, effectively enforcing a pseudo-first-order kinetic regime that favors mono-iodination. Furthermore, the absence of strong mineral acids or aggressive oxidants like periodic acid means that the aromatic ring and other sensitive functional groups (such as methyl or chloro substituents defined in Formula I) remain intact throughout the process. Post-reaction workup is streamlined because the solid hydrotalcite catalyst can be easily removed via filtration, and any residual unreacted iodine is efficiently quenched using sodium bisulfite during the recrystallization step, yielding a product with exceptional purity profiles suitable for downstream pharmaceutical applications.
How to Synthesize Iodoaniline Derivatives Efficiently
Implementing this synthesis route requires precise adherence to the catalyst preparation and reaction conditions outlined in the patent to ensure reproducibility and maximum yield. The process begins with the meticulous synthesis of the iodate-intercalated hydrotalcite, where maintaining a CO2-free environment and controlling the pH during the double-drop addition of metal nitrates and base is paramount to achieving the correct interlayer spacing and iodine content. Once the catalyst is prepared and characterized, the actual iodination reaction is conducted in a standard reactor equipped with vigorous stirring to maintain suspension of the solid catalyst within the acidic buffer medium. Operators must carefully monitor the temperature profile, keeping it within the preferred range of 35°C to 75°C, and allow sufficient time for the slow-release mechanism to drive the conversion to completion before proceeding to the isolation stage. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the technical guide below for immediate reference by process engineers.
- Prepare iodate-intercalated zinc-aluminum hydrotalcite via a double-drop method under nitrogen atmosphere, ensuring precise pH control between 6.8 and 7.2 during crystallization.
- In an acidic buffer solution (pH 3.7-5.2), combine the prepared hydrotalcite catalyst, alkali metal iodide, and the aniline derivative substrate.
- Stir the reaction mixture vigorously at temperatures between 35°C and 75°C for 2 to 3 hours, followed by filtration, extraction, and recrystallization to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hydrotalcite-based iodination technology translates into tangible strategic benefits that extend far beyond simple reaction yield improvements. By eliminating the reliance on volatile elemental iodine and hazardous strong oxidants, the process inherently reduces the risks associated with raw material storage, handling, and transportation, thereby lowering insurance premiums and safety compliance costs. The enhanced stability of the reagents allows for more flexible inventory management and reduces the likelihood of production stoppages caused by the degradation of sensitive iodine sources. Furthermore, the simplified workup procedure, which avoids complex neutralization steps and extensive washing required by strong acid methods, significantly shortens the overall batch cycle time, increasing the throughput capacity of existing manufacturing facilities without the need for capital-intensive equipment upgrades. These operational efficiencies collectively contribute to a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic value of this process is primarily driven by the drastic improvement in raw material utilization efficiency, as the controlled release mechanism prevents the loss of iodine through sublimation which is a major cost driver in traditional methods. Additionally, the replacement of expensive and dangerous oxidizing agents with stable, reusable hydrotalcite materials significantly lowers the cost of goods sold (COGS) by reducing both reagent expenses and the costs associated with hazardous waste treatment and disposal. The ability to operate at lower temperatures also results in substantial energy savings, further compressing the manufacturing budget and improving the overall margin profile for high-volume production runs. Qualitative analysis suggests that the elimination of corrosion-related maintenance on reactors, caused by free iodine and strong acids, will lead to extended equipment lifecycles and reduced downtime for repairs.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the use of readily available and stable starting materials such as zinc nitrate, aluminum nitrate, and common alkali metal iodides, which are less subject to market volatility compared to specialized oxidizing reagents. The robustness of the catalyst system allows for consistent batch-to-batch quality, reducing the rejection rates of final products and ensuring a steady flow of materials to downstream API manufacturers. Moreover, the safety profile of the process minimizes the risk of regulatory shutdowns or accidents that could disrupt production schedules, providing a more predictable delivery timeline for global customers. This reliability is crucial for long-term contracts where consistent supply of high-purity intermediates is a contractual obligation.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the heterogeneous nature of the catalyst, which simplifies separation and allows for continuous flow processing possibilities in the future. The environmental benefits are profound, as the process generates significantly less toxic waste and avoids the emission of iodine vapors, aligning perfectly with increasingly stringent global environmental regulations and corporate sustainability goals. The use of aqueous buffer systems and the potential for catalyst recycling or safe disposal as non-hazardous solid waste further enhances the green chemistry credentials of the manufacturing site. This alignment with eco-friendly standards not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer among environmentally conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrotalcite-catalyzed synthesis method, derived directly from the patent specifications and experimental data. These answers are designed to provide clarity on the operational parameters, safety profile, and scalability of the technology for potential licensees and manufacturing partners. Understanding these details is essential for conducting a thorough feasibility study and integrating this process into existing production lines. Please refer to the specific technical sections above for deeper mechanistic explanations.
Q: How does the hydrotalcite catalyst improve safety compared to traditional iodination methods?
A: Traditional methods often rely on elemental iodine which sublimes easily or dangerous strong oxidants like periodic acid. The hydrotalcite system slowly releases iodate ions in situ, generating active iodine species gently without the risks of sublimation or explosive oxidants.
Q: What represents the primary cost advantage of this synthesis route for procurement teams?
A: The primary cost advantage stems from significantly improved raw material utilization. By preventing iodine sublimation and avoiding expensive, hazardous oxidants, the process reduces waste disposal costs and lowers the overall consumption of iodine sources.
Q: Is this process suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the process operates under mild conditions (35°C-75°C) and atmospheric pressure using stable solid catalysts. These factors, combined with short reaction times and simple workup procedures involving filtration and recrystallization, make it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iodoaniline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the hydrotalcite-catalyzed synthesis route described in patent CN103467306A for producing high-value iodoaniline derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical clients, guaranteeing that every batch of iodoaniline intermediate meets the highest quality standards. We are committed to leveraging this advanced technology to deliver superior products that enhance your drug development pipeline while maintaining full regulatory compliance.
We invite you to collaborate with us to explore how this innovative synthesis method can optimize your specific supply chain requirements and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your project timelines and secure your supply of critical chemical intermediates.
