Advanced Synthesis of Simvastatin via Optimized Silylation and Methylation Strategies
Advanced Synthesis of Simvastatin via Optimized Silylation and Methylation Strategies
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective methodologies for the production of high-value active pharmaceutical ingredients (APIs), particularly for cardiovascular therapeutics like statins. Patent CN101381356A introduces a transformative approach to the preparation of simvastatin, a potent HMG-CoA reductase inhibitor widely utilized in the management of hyperlipidemia and the prevention of cerebrovascular diseases. This technical disclosure outlines a novel synthetic route that strategically addresses the limitations of conventional methods by optimizing the protection and methylation steps. By employing a specific composite solvent system and leveraging the unique reactivity of trialkylsilyl protecting groups, the process achieves a significant simplification of the synthetic pathway. For R&D directors and process chemists, this represents a critical opportunity to enhance purity profiles while simultaneously reducing the operational complexity associated with traditional simvastatin manufacturing. The following analysis details the mechanistic advantages and commercial implications of adopting this refined methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of simvastatin has been plagued by several inherent inefficiencies that impact both yield and overall production economics. Traditional routes often rely on tert-butyldimethylsilyl chloride (TBDMSCl) for hydroxyl protection, a reagent that is not only expensive but also necessitates harsh acidic conditions for subsequent deprotection. These strong acidic environments can compromise the integrity of the sensitive lactone ring or lead to unwanted side reactions, thereby diminishing the final product quality. Furthermore, conventional protocols typically require rigorous washing and purification steps between the protection and methylation stages to remove excess reagents and byproducts. These intermediate operations increase the risk of premature deprotection, where the silyl groups are inadvertently removed before methylation is complete, leading to a complex mixture of impurities that are difficult to separate. Additionally, alternative enzymatic or hydrolytic methods often suffer from long reaction times, sometimes extending up to 60 hours, and require extreme pH conditions that further degrade product stability. These cumulative factors result in lower overall yields, often hovering around 40%, and generate substantial waste streams that complicate environmental compliance.
The Novel Approach
In stark contrast, the methodology disclosed in CN101381356A offers a streamlined solution that elegantly bypasses these traditional bottlenecks through intelligent solvent engineering and reagent selection. The core innovation lies in the substitution of the costly TBDMS group with a trimethylsilyl (TMS) group, facilitated by the use of trimethylchlorosilane (TMSCl), which is far more economical. Crucially, the process utilizes a composite solvent system comprising tetrahydrofuran (THF) and cyclohexane for the silylation reaction. This specific solvent ratio ensures that upon completion of the protection step, cooling the reaction mixture causes insoluble impurities to precipitate completely, allowing for their removal via simple filtration. This eliminates the need for alkali or water washing prior to methylation, which traditionally triggers premature deprotection. Consequently, the filtered solution containing the lovastatin amide bis(trimethyl)silyl ether can be directly subjected to methylation. Perhaps most remarkably, the subsequent aqueous workup following methylation induces the automatic脱落 (falling off) of the protecting groups, effectively merging the deprotection and isolation steps into a single operation. This integration drastically reduces the number of unit operations and minimizes material handling losses.
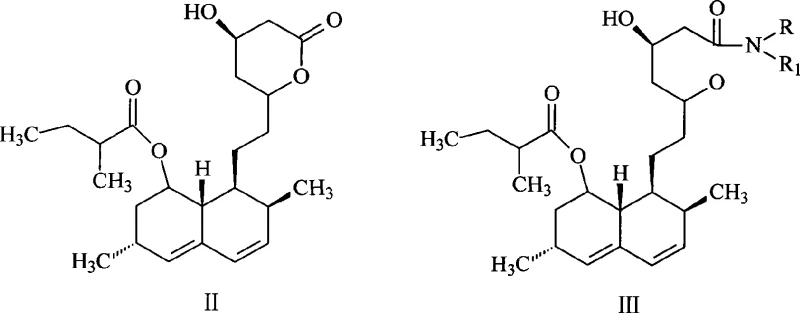
Mechanistic Insights into Trialkylsilyl Protection and Methylation
The success of this synthetic route hinges on the precise control of silyl ether stability and reactivity under specific solvent conditions. In the protection phase, the use of imidazole as a catalyst in the THF-cyclohexane mixture promotes the efficient formation of the disiloxane derivative (Structure IV). The thermodynamic properties of this solvent blend are critical; they maintain the silylated intermediate in solution during the reaction at elevated temperatures (40-90°C) but facilitate the crystallization of imidazole hydrochloride and other salts upon cooling. This physical separation technique prevents the introduction of water or base that would otherwise hydrolyze the TMS groups. Following filtration, the methylation step employs a strong base, generated in situ from a secondary amine like pyrrolidine and an alkyllithium reagent such as n-butyllithium, to deprotonate the alpha-carbon of the side chain ester. The resulting enolate is then reacted with methyl iodide to introduce the requisite methyl group, transforming the 2-methylbutyrate side chain into a 2,2-dimethylbutyrate moiety. The stability of the TMS groups during this low-temperature (-50 to -10°C) methylation is sufficient to prevent side reactions, yet they remain labile enough to be cleaved spontaneously upon the addition of water during the quenching phase. This "self-immolative" behavior of the protecting group under workup conditions is a key mechanistic feature that distinguishes this process from prior art.
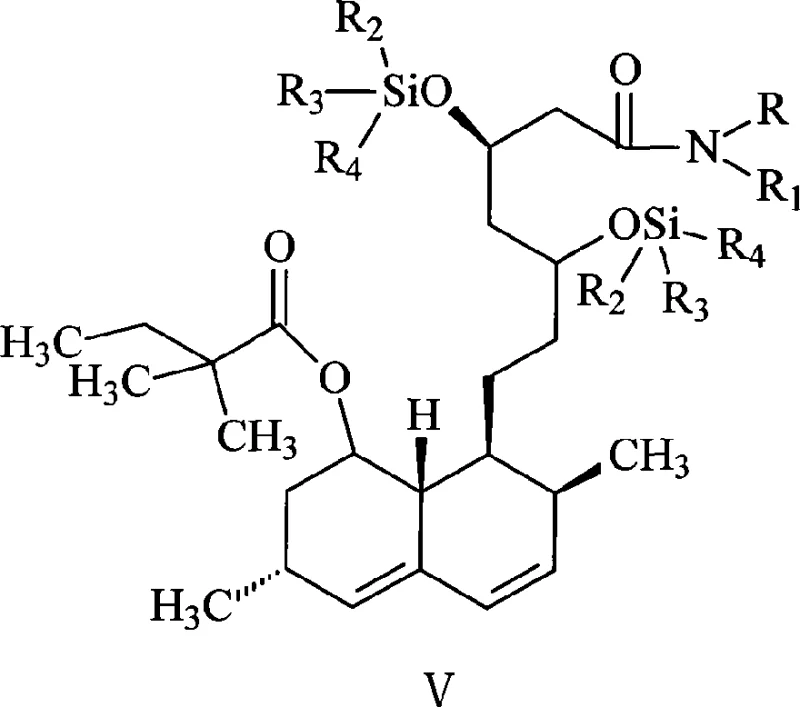
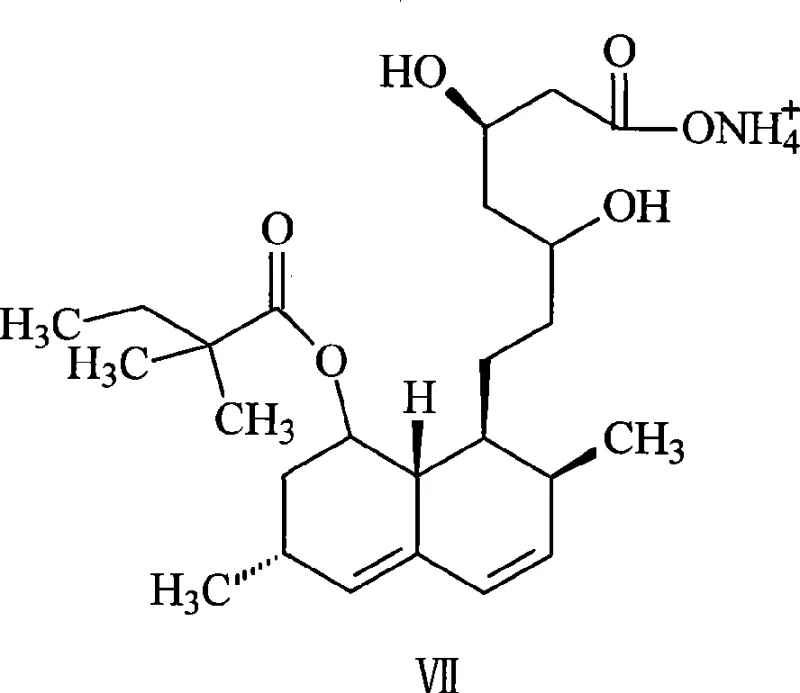
Furthermore, the control of impurities is intrinsically built into this mechanism. By avoiding the harsh acidic deprotection steps required by TBDMS groups, the process mitigates the risk of lactone ring opening or rearrangement, which are common degradation pathways in statin synthesis. The direct transition from the methylated intermediate to the free hydroxy amide (Structure VI) via aqueous wash ensures that the product is isolated in a form ready for the subsequent hydrolysis step without requiring additional purification columns or recrystallizations at this stage. This mechanistic elegance translates directly into a cleaner crude product profile, reducing the burden on downstream purification processes such as activated carbon treatment and final recrystallization. For quality assurance teams, this means a more consistent impurity profile and a higher probability of meeting stringent pharmacopoeial specifications for related substances and residual solvents.
How to Synthesize Simvastatin Efficiently
The implementation of this synthesis requires strict adherence to the specified reaction parameters to maximize yield and purity. The process begins with the ring-opening of lovastatin using an alkylamine, followed by the critical silylation in the THF-cyclohexane system. Detailed operational guidelines regarding temperature gradients, molar ratios of silylating agents, and filtration protocols are essential for reproducibility. The subsequent methylation must be conducted under inert atmosphere with precise temperature control to manage the exotherm and ensure complete conversion. Finally, the cyclization of the ammonium salt back to the lactone form requires careful pH buffering to avoid epimerization. For a comprehensive, step-by-step technical guide including exact reagent quantities and safety precautions, please refer to the standardized operating procedure below.
- Convert lovastatin to lovastatin amide using alkylamines such as n-butylamine at elevated temperatures.
- Protect hydroxyl groups using trimethylchlorosilane in a THF-cyclohexane composite solvent to form the disiloxane intermediate.
- Perform alpha-methylation using alkyllithium and methyl iodide, followed by aqueous workup which automatically removes protecting groups.
- Hydrolyze the amide to form the ammonium salt, followed by acid-catalyzed cyclization to yield the final simvastatin lactone.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this patented methodology offers compelling advantages that extend beyond mere technical feasibility. The simplification of the synthetic route directly correlates with enhanced manufacturing efficiency and reduced operational expenditures. By eliminating distinct deprotection steps and minimizing the number of isolation procedures, the overall cycle time for production is significantly compressed. This acceleration allows for increased throughput within existing facility footprints, effectively boosting capacity without the need for capital-intensive infrastructure expansion. Moreover, the reduction in unit operations leads to a corresponding decrease in solvent consumption and waste generation, aligning with modern green chemistry principles and lowering the costs associated with environmental compliance and waste disposal. For procurement managers, these efficiencies translate into a more competitive cost structure for the final API, providing a buffer against raw material price volatility.
- Cost Reduction in Manufacturing: The substitution of expensive tert-butyldimethylsilyl chloride with the more economical trimethylchlorosilane represents a direct material cost saving that scales linearly with production volume. Additionally, the elimination of the dedicated deprotection reaction step removes the requirement for specific acidic reagents and the associated neutralization chemicals, further driving down the bill of materials. The ability to proceed directly from the protection filtrate to methylation without intermediate washing also reduces solvent usage and labor costs associated with multiple extraction and drying phases. These cumulative savings contribute to a substantially lower cost of goods sold (COGS), enhancing the margin potential for generic manufacturers.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as TMSCl, THF, and cyclohexane ensures a robust and diversified supply base, mitigating the risk of shortages associated with specialized reagents. The simplified process flow reduces the number of potential failure points in the manufacturing chain, leading to higher batch success rates and more predictable delivery schedules. This reliability is crucial for maintaining continuous supply to downstream formulation partners and meeting regulatory commitments. Furthermore, the reduced processing time allows for more flexible production scheduling, enabling manufacturers to respond more agilely to fluctuations in market demand.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as filtration, distillation, and crystallization that are well-understood in industrial settings. The avoidance of harsh acidic conditions for deprotection enhances operational safety and reduces corrosion risks to equipment, extending asset life. From an environmental standpoint, the reduction in solvent volumes and the minimization of aqueous waste streams simplify effluent treatment processes. This alignment with sustainability goals not only reduces regulatory burdens but also enhances the corporate social responsibility profile of the manufacturing entity, a factor increasingly valued by global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this simvastatin synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind the process innovations. Understanding these details is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value proposition of this manufacturing strategy.
Q: What is the primary advantage of using trimethylchlorosilane over tert-butyldimethylsilyl chloride in this process?
A: Trimethylchlorosilane is significantly more cost-effective and readily available. Furthermore, the specific solvent system allows for automatic deprotection during workup, eliminating a separate acidic deprotection step required by traditional methods.
Q: How does the THF-cyclohexane composite solvent improve the reaction efficiency?
A: This solvent combination ensures complete solubility during the silylation reaction while allowing insoluble byproducts to precipitate upon cooling. This enables direct filtration and subsequent methylation without intermediate washing steps that could prematurely remove the protecting group.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process simplifies the workflow by reducing the number of unit operations and avoiding harsh acidic conditions for deprotection, making it highly scalable and safer for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Simvastatin Supplier
The technological advancements detailed in patent CN101381356A underscore the potential for significant optimization in statin manufacturing, yet realizing this potential requires a partner with deep technical expertise and proven scale-up capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this landscape, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and low-temperature reactions required by this process, ensuring that the theoretical benefits of the patent are fully realized in commercial output. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of simvastatin meets the highest international standards for potency and impurity control.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this efficient synthesis route for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain resilience.
